Q: What is the molar solubility of lead(II) bromide PbBr2 Ksp = 4.67 x 10-6.
A: The objective of this question is to calculate the molar solubility of lead(II) bromide (PbBr2)…
Q: 14 CH3OH H₂SO4
A:
Q: 5. Explain the relevance of the geometric isomers to our lives in terms of presence of cis and trans…
A: The objective of this question is to understand the importance of geometric isomers, specifically…
Q: (a) A 40 mL sample of 0.100 M HNO2 (pKa = 3.34) is titrated with 0.200 M NaOH. Find the pH after…
A: The objective of this question is to find the pH at different stages of a titration between a weak…
Q: What is the pH of a 0.030 M morphine (C17H19NO3) solution? (The pk of morphine is 5.79 at 25°C.)
A: Given:Concentration of morphine solution=0.030 MpKb of morphine=5.79We have to find the pH of the…
Q: Which of the following element(s) are typically added to gray iron to make it stronger and more…
A: Gray iron also called as gray cast iron has graphitic microstructure.Gray iron has high thermal…
Q: Draw the major organic product of the reaction. Indicate the stereochemistry via wedge-and-dash…
A:
Q: If wavelength of photon is 2.2 × 10-11m and h = 6.6 × 10-34Js, then momentum of photon
A:
Q: The main role of lipoproteins in a healthy human is to: Question 16 options:…
A: The question is asking about the primary function of lipoproteins in the human body. Lipoproteins…
Q: 5. Consider a molecular solid such as anthracene. It belongs to a monoclinic space group P2₁/a. a=…
A: The objective of the question is to understand the structure of the molecular solid anthracene,…
Q: HO .OH cat. H2SO4 H₂O cat. H2SO4 Complete both mechanisms above. BRIEFLY explain how the different…
A: The objective of the question is to understand the mechanisms of the reactions catalyzed by H2SO4,…
Q: What CO2 partial pressure (torr) is required to yield a velocity of 0.045 M-s1 for the reaction?…
A: The velocity is given as v = 0.045 M/sIt is required to find the pressure in torr.
Q: 74) Identify the reagents of product required. Add any Side products not shown. a) C b) AICI AICI…
A: Electrophilic aromatic substitution:Halogenation: Benzene on reaction with halogen in presence of…
Q: I am stuck on question 4, I found everything
A: The objective of the question is to determine the weight in grams and weight percentage of magnesium…
Q: Calculate the solubility (in g/L) of CaSO4(s) in 0.450 M Na, SO4(aq) at 25°C. The Ksp of CaSO4 is…
A: Molarity of Na2SO4 = 0.450 MKsp of CaSO4 = 4.9310-5molar mass of CaSO4 = 136.14 g/mol
Q: please help me with filling in the table below
A: Oxidative addition (i):Structure: [L-Pd(OTf)(X)]X-type ligands: 1L-type ligands: 3Oxidation state:…
Q: Given the following visual of the reaction below, how many of each species will be in the products…
A: The objective of the question is to determine the number of each species in the products box on the…
Q: 5. Identify compounds A, B, and C in the following synthetic sequence. CN H3O+ A CH3OH, H LIAIH4 B C…
A: Organic reactions can be defined as the reactions in which organic reactants react with each other…
Q: A 25.00 mL sample of 0.250 M LiOH is titrated with 0.750 M HCl at 25\deg C. Calculate the initial pH…
A: Molarity of LiOH = 0.250 MVolume of LiOH = 25.00 mL = 0.025 LMolarity of HCl = 0.750 MVolume of HCl…
Q: Using the two images attached please write one paragraph EXPLAINING THE ANALYSIS Please please…
A: The objective of this question is to understand the principles and procedures involved in the…
Q: A reaction is initiated by preparing an aqueous solution with 0.421 M silver ion, Ag+ (aq), and…
A: • So, The correct answer is : Kc = 6.02E1Explanation:Step 1:
Q: A 37.0 mL sample of a 0.380 M aqueous hydrocyanic acid solution is titrated with a 0.464 M aqueous…
A:
Q: 1. Which of the following is the correct name for the compound shown? Bubble your answer in…
A: The objective of the question is to find the correct name for the given compound.IUPAC naming is…
Q: Draw the mechanism of the acid catalyzed hydrolysis of the two following compounds. The products…
A: In the given question we have to do the acid catalysed hydrolysis of ester and amide functional…
Q: HO OH cat. H2SO4 H₂O cat. H2SO4 Complete both mechanisms above. BRIEFLY explain how the different…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: 9. Use an ethyl acetoacetate synthesis or malonic acid syntheses to prepare the following compounds.…
A: Active methylene compounds like Acetoacetic ester and malonic ester on treatment with base, the acid…
Q: At room temperature, what is the viscosity of drinking water, water with a 10% salt solution, and…
A: The objective of this question is to determine the viscosity of drinking water, water with a 10%…
Q: Gluconic acid is a substance in honey that prevents is from spoiling. The pKa of gluconic acid is…
A: Given that,The pKa of gluconic acid = The concentration of gluconic acid,The pH of the gluconic acid…
Q: 4. Provide a mechanism for the following transformation: NaOH H2N Н20, д
A: The given question is based on nucleophilic addition reaction.
Q: What is the major product? HNO3 Sn ? HC1 H₂SO NO2 NH2 NO₂ 8628 b C NO₂ d
A: ->Aromatic electrophilic substitution reaction in which an electrophile replaces a hydrogen atom…
Q: Draw the major product of this reaction. Ignore inorganic byproducts and the carboxylic acid side…
A: Given,The reaction:
Q: 1. Which of the following moldcules is the strongest? A. H-F B. H-Cl C. H-Br D. H-I
A: The objective of the question is to identify the strongest molecule among H-F, H-Cl, H-Br, and H-I.
Q: Select the best reagents for each of the following transformations. You should only use each set of…
A: 1.) G2.) C3.)I4.)BExplanation:1.) this is a Baeyer Villiger reaction in which aldehyde/ketone…
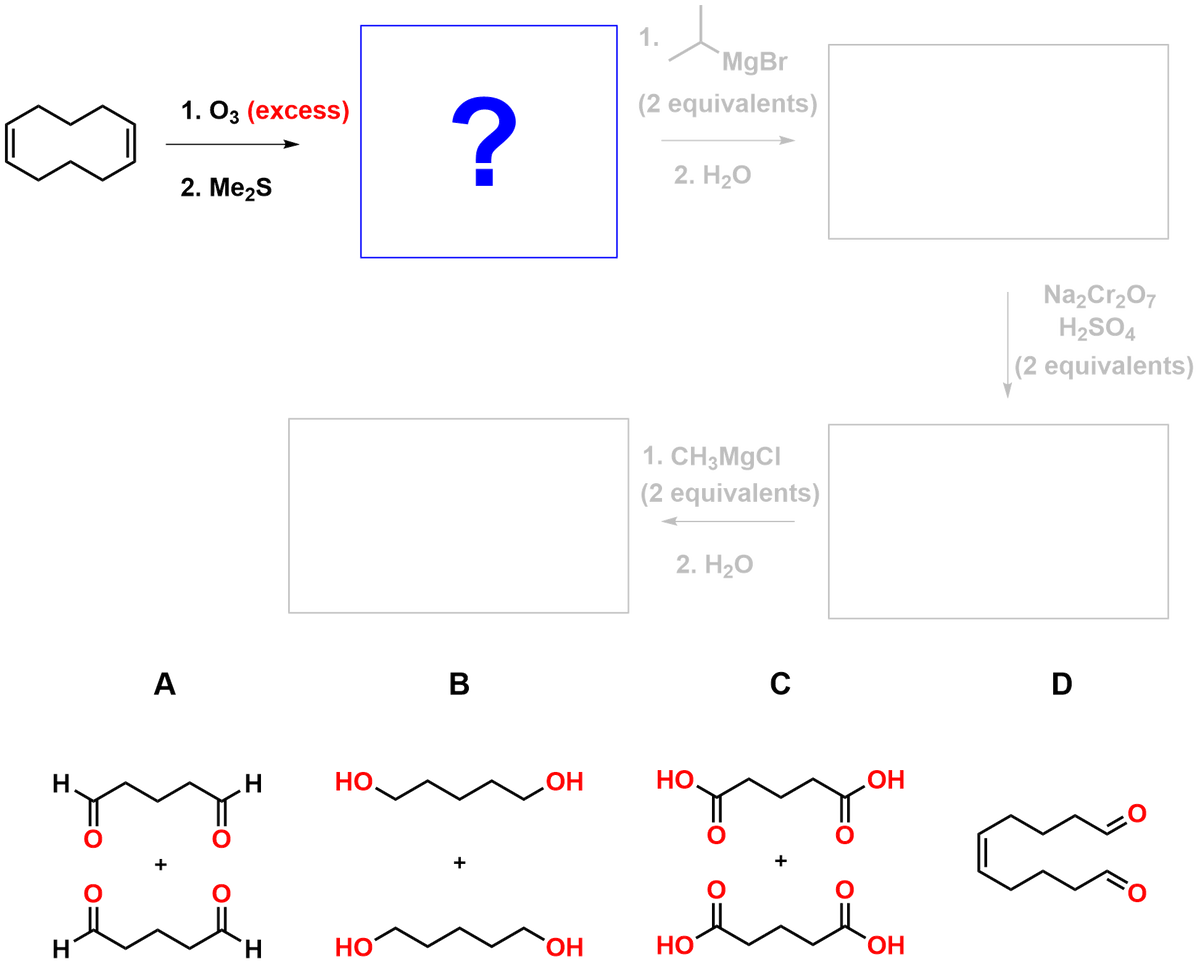
Q: -MgBr 1.03 2. H₂O 2. Me2S ? 1. LIAIH 2. H₂O 1. =MgBr 2. H₂O ? NaBH4 MeOH A H₂C 107
A: Ozonolysis is a reaction that breaks the double bond of an alkene and replaces it with two carbonyl…
Q: ИН H₂O (xs) H2SO4
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: onsider the following chemical equilibrium: 2H2(g) +0, (g) 2H₂O (1) ow write an equation below that…
A: Kp And Kc are the equilibrium constant of an ideal gaseous mixture. Kp is equilibrium constant used…
Q: Using chemdraw or a any chemical drawing program show the mechanism of the decarboxylation of 3,4-…
A: The given reaction is decarboxylation reaction. Decarboxylation reaction involves loss of carbon…
Q: 8. Use the following reactions and Draw the transition state in the following reactions: CH3 CH3 a)…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: Which of the following would you expect to NOT undergo a Jahn-Teller distortion? Select all that…
A: Each of the given compounds is an octahedral complex. According to CFT, the orbital splitting…
Q: Name the following compounds and draw their structures: (5) a) [Fe(OH)( H2O)5] Cl2 b) Li[Cr(OH)4] c)…
A: The objective of the question is to name the given compounds and draw their structures. The…
Q: 4. An enzyme (acetoacetate decarboxylase) catalyzes the decarboxylation of acetoacetate. The…
A: Decarboxylation is the chemical reaction by which CO2 is released through the removal of carboxylic…
Q: Use the reaction quotient to predict whether a precipitate will form. If 15.0 mL of 3.74×104 M Cal₂…
A: Given:Calculate concentrations of ions in solution after mixing solutions of different salts…
Q: Draw the expected major elimination product and identify the mechanism. H3C CH3 CH3OH
A: -> In presense of alcohol as reagent there occur elimination reaction which proceeds through E1…
Q: Bile is necessary for what process? production of HDL…
A: The question is asking about the role of bile in the human body. Bile is a fluid that is produced by…
Q: Some reactions are second order under low reactant concentrations, but are first order athigher…
A: All parts are solved below. In case you are having any problem feel free to discuss it with me in…
Q: (1) The element with an electron configuration of 1s22s22p63s23p64s23d3 is in group (2) The element…
A: Given are electronic configurations.In the electronic configuration, the principal quantum number…
Q: Which is the stereochemical relationship between this pair of molecules? L エ ... H CI A) enantiomers…
A: Given,The molecules:
Q: Suppose a 250. mL flask is filled with 1.2 mol of NO and 0.90 mol of NO 2. The following reaction…
A:
Q: 9. Draw the major product(s) for each reaction. Label all reactants and products (R,S,E,Z) when…
A: The question is based on organic reactions.We need to identify the product and explain its…
Q: Suppose a 500. mL flask is filled with 1.7 mol of OCl2, 0.70 mol of BrOCI and 1.1 mol of BrCl. The…
A: Answer:Value of equilibrium constant is always equal to the ratio of molar concentration of products…
Predict the final product for each of the following reaction or synthetic chain:
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

- A mixture of pure BaCO3 and pure Na2CO3 weighs 1.000 g and has the total neutralizing power of 15.37 meq of CaCO3. Calculate the percentage of combined CO2 in the mixture and the weight of Li2CO3 that has the same neutralizing power as 1.000 g of the above mixture. Note: include up to 4 decimal places Please explain each stepA mixture of pure BaCO3 and pure Na2CO3 weighs 1.000 g and has the total neutralizing power of 15.37 meq of CaCO3. Calculate the percentage of combined CO2 in the mixture and the weight of Li2CO3 that has the same neutralizing power as 1.000 g of the above mixture. Instructions: Include up to 4 decimal places See sample photo for the format of solutionA commercial product designed to prevent cut fruit from turning brown contains a mixture of ascorbic acid (HC6H7O6) and sugar (C12H22O11). Calculate the percent (w/w) of ascorbic acid in this product if a 2.1994 g portion of this product requires 36.15 mL of 0.2328 M NaOH to reach a phenolphthalein endpoint. MM HC6H7O6: 176.12 MM NaOH: 40.00
- It is known that acid content has a major effect on theflavor of vinegars, but most cheaper vinegars are diluted similarly to 5% acidity Wt./vol. % is equivalent to gsolute per 100mL solution (so 5% is equivalent to 5 g acid/100 mL solution). a.) First, calculate the approximate molar concentration of acetic acid in the 5% wt./vol vinegar. b.) Next, calculate the expected molarity of acetic acid in the solution upon dilution by a factor of 5. Thank you!You work with two amino acids. Tryptophan (MW=204 Da) and Methionine (MW=149 Da). You make varioussolutions of each amino acid separately and compare them When you compare two solutions at the same percentage concentration (e.g. 1%), there are moremoles of Tryptophan in solution than moles of Methionine because the molecular weight of Tryptophan ishigher. When you compare two 1-liter solutions containing these amino acids at a 10mM concentration, thereis the same number of amino acid molecules in each solutionDetailed calculations on how to prepare the solutions listed below Solution: 50 mL 1 M oxalic acid Oxalic acid Mw = 90.03 g mol-1 Oxalic acid purity = 98% Solution: 10 mL 3 M sulfuric acid Sulfuric acid Mw = 98.079 g mol-1 Sulfuric acid density = 1.84 g mL-1 Sulfuric acid purity = 98% Solution: 10 mL saturated potassium oxalate potassium oxalate (monohydrate used) Mw = 184.23 g mol-1 Solubility of potassium oxalate in water at 25°C = 360 mg mL-1 Solution :20 mL 3% hydrogen peroxide Hydrogen peroxide is commercially available as a 32% solution.
- A mixture of pure BaCO3 and pure Na2CO3 weighs 1.000 g and has the total neutralizing power of 15.37 meq of CaCO3. Calculate the percentage of combined CO2 in the mixture and the weight of Li2CO3 that has the same neutralizing power as 1.000 g of the above mixture.Q1/ prepare ( 0.3 M ) of HCl in 250 ml water, when you know Sp.Gr=1.18 and % = 36% . At.Wt / H=1 , Cl= 35.5A weight of 0.50 g was taken impure container containing sodium carbonate and bicarbonate. Dissolved in water and then crushed with hydrochloric acid (0.1 N), the burette reading game was at the endpoint of phenolphthalein of 10.5 ml and at the end point of the orange methylation point 30.1 ml. The percentage of sodium carbonate was in ................. knowing that the weights are: Na: 23, C: 12, O: 16
- Was wondering how to solve this hw problem? Calcium Flouride (dissociation) K@25 C = 3.94 *10^-11 Calcium fluoride is not very soluble in water. ** Its dissociation is: CaF2 (s) Ca²+ (aq) + 2 F¯ (aq) a. At 25°C, how many milligrams of CaF2 would dissolve in 2.0 L of water? b. What were your initial and final values of Q?Consider the Solubility Curve for this Problem: Analysis for KClO3 & NH4Cl MW (KClO3) = 122.55 g/mol , MW (NH4Cl) = 53.49 g/mol i) What is the temperature at which KClO3 & NH4Cl both have the same solubility? Round to nearest degree. +/- 1° T = °C ii) What is the solubility (g per 100g H2O) at this temperature. Round to nearest gram. +/- 1 g s (KClO3) = g/100g H2O s (NH4Cl) = g/100g H2O iii) What is the molar solubility for KClO3 at this temperature? Assume the density of the solution is 1.21 g/cc. Round off to 2 sig. figs. |s| KClO3 = M iv) Calculate the Ksp for each of these chemicals at this temperature. Ksp = v) What is the unit of Ksp for KClO3? Unit for Ksp =Part 1: Preparation of the Primary Citric Acid Standard Mass of Citric Acid: 4.05 g Volume of Citric Acid Solution: 7.5 (at the equivalence point) Moles of Citric Acid: 0211 mol (Molar Mass = 192.0 g/mol) Molarity of Citric Acid Solution: 0.2813 help me with part Part 2: Titration of the Sodium Hydroxide Solution Volume of Citric Acid at the Equivalence Point _____________ Moles of Citric Acid at the Equivalence Point _______________ Moles of NaOH at the Equivalence Point __________________ 3 NaOH + H3C6H5O7 à Na3C6H5O7 + 3 H2O Volume of NaOH _______________________________ 5. Calculated Molarity of NaOH _______________________



