Q: Calculate the percentage by mass of oxygen in the following compounds. 4 Part B methadone, C21H27 NO…
A:
Q: What are some identifying characteristics (reactants, products, formats) for each of the 8 reaction…
A: The eight types of chemical reactions are synthesis, decomposition, single displacement, double…
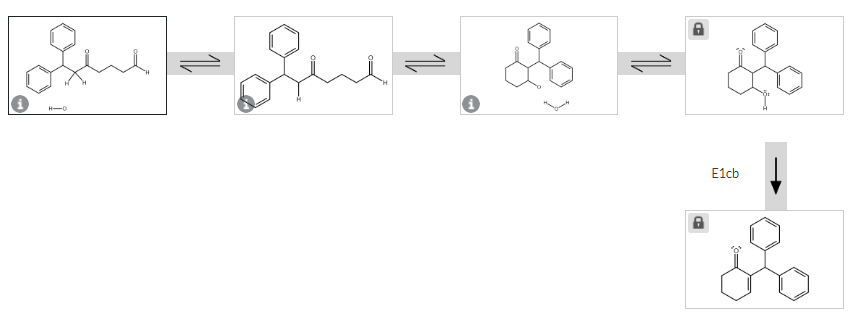
Q: Draw all the intermediates formed for the reactions shown. (Hint: There are five intermediates for…
A: The first reaction is the esterification reaction,at first presence of acid protonation occurs at…
Q: Balance the redox reaction: H, S + NO H2S+NOS + NO 3 8
A: A balanced chemical reaction occurs when the number of the atoms involved in the reactants side is…
Q: Which group r compo Ma Mag This
A: magnesium reacts with oxygen and forms magnesium (II) oxide Charge on Mg = +2Charge on O = -2
Q: 2. IN HNO3 H2SO4 % o/p/m?
A: Information about the question
Q: In the laboratory a student combines 29.9 mL of a 0.426 M ammonium phosphate solution with 17.6 mL…
A:
Q: What is the formal charge on a chlorate ion where chlorine is single bonded to each oxygen? +1 +2 +3…
A: The difference between an atom's valence electron in its elemental state and the number of electrons…
Q: C2O4 (aq) + H2O2 (aq) + 2 H+ (aq) → 2 CO2 (g) + 2 H₂O (1) Use the experimental data in the table to…
A: Rate law,Rate=k[C2O4-2][H2O2]2[H+]Explanation:Where k=rate constant
Q: second order reaction starts out with a concentration of F2 as .064 M. If k=.03, after a time 150s,…
A: Integrated rate equation for second order reaction is,A0 - Initial concentration of reactant AAt -…
Q: Draw the appropriate Newman projection which leads to the major E2 ELIMINATION product. Hint: Review…
A:
Q: 9.64 Draw the products of each reaction, and indicate the stereochemistry where appropriate. a.…
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: The following H-NMR spectrum (showing all available signals) is for one of the possible products…
A: A triplet at 1.1 ppm and a quartet at 2.4 ppm suggests that the molecule has -CH2CH3 group. The…
Q: A piece of aluminum foil 1.18 cm2 and 0.590-mm thick is allowed to react with bromine to form…
A: The area of aluminum foil = The thickness of aluminum foil = The density of aluminum foil =
Q: When the following skeletal equation is balanced under basic conditions, what are the coefficients…
A: A redox reaction is balanced by balancing the mass and charge. Hence the number of atoms of all the…
Q: CH3 H3C-C- C H₂ Br CH3 H₂O
A: Answer:S in sn1 reaction is unimolecular nucleophilic substitution reaction.
Q: Draw major E2 ELIMINATION product. Hint 1: Review Chapter 10.3, the elimination can only occur if…
A: Based on your question we need to find out the product by using beta elimination mechanismI'm…
Q: 10.52 Draw the products of each reaction, including stereois a. H₂O H₂SO4 b. C. Cl₂ HI d. e. Cl₂ H₂O…
A: The question is based on organic reactions.We need to identify the product and explain its…
Q: An analytical chemist is titrating 165.1 mL of a 1.100M solution of acetic acid (HCH3CO2) with a…
A: The volume of acetic acid (HCH3CO2) = 165.1mL = 0.165LThe Molarity of acetic acid( HCH3CO2)…
Q: Each row of the table below describes an aqueous solution at 25 °C. The second column of the table…
A: We have to complete the chart.
Q: i) Ph2PH ii) Nal iii) PhCH2OH iv) CH3SH v) Pr2CuLi At what letter in the picture will each above…
A: The reaction between substrate and reagent depends on the soft-hard acid base concept. A soft…
Q: Consider the reaction: CO2(g) + CCl4(g) ⇌ 2 COCl2(g) Calculate ΔG for this reaction at 25 °C under…
A: CO2 (g) + CCl4 (g) → 2COCl2 (g)∆G° = 2∆G° (COCl2) - [ ∆G°f (CO2) + ∆G°f (CCl4)] =…
Q: Provide an acceptable name for the following compound. 0- CH-N-CH2CH3 Spell out the full name of the…
A: RULES for IUPAC nomenclature; In first step, we identify functional groups present. We make longest…
Q: Helium present inside a spherical balloon with a diameter of 7.5 m is found to be at a pressure of…
A: Given that,Diameter = 7.5 mPressure = 200 kPa.Surronding air temperature = 22 degrees Celsius.
Q: For the given SN2 reaction, draw the organic and inorganic products of the reaction, and identify…
A:
Q: In the provided boxes, provide synthetic sequences (including reagents used and the compound…
A: The objective of this question is to convert a given compound into a specified target compound…
Q: Q, Rotate your molecule so that you can see what the rat is looking at below. Draw the Newman…
A: The dot represents the front carbon and circle represents the back carbon.
Q: Refer to the Ksp values in table pasted below to calculate the molar solubility of each compound in…
A: Information about the question
Q: Consider the following equilibrium system: 2CrO4 2-(aq)+2H+(aq) = Cr2O72 (aq)+H2O(1) In what…
A: As per Le-Chatelier's principle, when a stress is applied to a system at equilibrium, the…
Q: 22. Propose a synthesis of the desired product from the given starting material(s). Starting…
A: The question is based on the concept of organic synthesis.. We need to synthesize the product using…
Q: Assuming the reaction shown below takes place via an SN2 mechanism, which of the compounds shown…
A: Organic reactions can be defined as the reactions in which organic reactants react with each other…
Q: Is there a pattern to the way pH, amount, K, pressure increases decreases or has no change?
A: To determine or predict the changes in the equilibrium, use the Le Chatelier's…
Q: Limestone stalactites and stalagmites are formed in caves by the following reaction: Ca2+(aq) +…
A: The balanced chemical equation is:Ca2+(aq) + 2HCO3-(aq) CaCO3(s) + CO2(g) + H2O(l) The reactant…
Q: 9.66 Prepare each compound from cyclopentanol. More than one step may be needed. a. CI b. OCH 3 C.…
A: Answers given below Explanation:
Q: Predict the major products of the following organic reaction: + NC Δ ? Some important Notes: • Draw…
A: The Diels–Alder reaction is a pericyclic reaction (cycloaddition) between a conjugated diene and a…
Q: The preparations of two aqueous solutions are described in the table below. For each solution, write…
A:
Q: A 0.200 M weak base has a pH of 9.96. What is the K, value of this base? K = 4.715e-8
A: The objective of this question is to find the K value of a weak base given its molarity and pH. The…
Q: The green band consisted of chlorophylls. What does it tell you about the relative polarity of the…
A: Here, the elution behavior of compounds in the chromatography is influenced by the polarity of the…
Q: Draw the possible 1,2- and 1,4-addition products formed when this diene reacts with Br2. Do not…
A: 1,2 addition product formed at low temperature ,also know as kinetic product .1,4 addition products…
Q: QUESTION 6 Identify the salts that will not hydrolyze water. The solution will be neutral. NaCl NH41…
A: Answer:Salts of strong acid and strong base doesn't get hydrolyzed in water. So, in this problem we…
Q: Draw Zaitsev and Hofmann products that are expected when each of the following compounds is treated…
A: In an E2 elimination reaction, an alkyl halide when treated with strong base, it can produce two…
Q: 18. If 1.00 mL KMnO4 solution will react with 0.125 g Fe2+ and if 1.00 mL KHC2O4 H2C2O4…
A: Given data:Determine the volume of NaOH.Mol is a measurement unit for substance amounts. It is a…
Q: Draw structural formulas for the a,ß-unsaturated aldehyde or ketone and the lithium diorganocuprate…
A: Organic reactions can be defined as the reactions in which organic reactants react with each other…
Q: Determine the relationship between the two compounds: O enantiomers same compound constitutional…
A: Enantiomers: Non-superimposable mirror images are called enatiomers If we move from one enantiomer…
Q: 6) What is the product of the following reaction? A) CHOCH D) LOCK CHOCH OCH CHO
A: The given reaction proceeds through the addition-elimination mechanism. As the leaving group present…
Q: 1. Explain why the protonation of pyrrole occurs at C2 to form A, rather than the N atom to form B…
A: pKa = 0.4pKa =5.3
Q: Draw one of the two enantiomers of the major product from this reaction. Use a dash or wedge bond to…
A: The addition of ICl to unsymmetrical alkene is takes place according to Markovnikovs rule. That…
Q: Find the pH of 0.150 M nitrous acid, HNO2, solution at 25o Also determine the percent ionization of…
A: pH = 2.10 % ionization = 5.35 %Explanation:
Q: Question 19 Identify the Major and ALL Minor product(s) that are expected for each of the following…
A: Alkyl halides undergo E2 elimination reactions in the presence of a strong base. The elimination…
Q: Part D Η Spell out the full name of the compound.
A: Find out the name of this compound
Step by step
Solved in 1 steps with 2 images


