Q: [OH-]=3.34×10-3 M pH рон
A: The objective of the question is to calculate the pH of a solution given the concentration of…
Q: Please help me , i need fast solution. Please don't provide handwritten solution ...
A: The objective of this question is to calculate the pH of a solution of NH3 after the addition of…
Q: Draw the electron flow arrows and the resulting resonance mechanism. CH3 CH3 H3C- H3C CH2
A: The objective of this question is to draw the resonance structure for the carbocation generated in…
Q: Perform the following synthesis. The number of steps in my path is provided. 5 steps OH SH
A: The given reaction is based on multiple steps synthesis to achieve targeted product.
Q: Write the formula correctly for the following like shown in the image below.…
A: Find the highest priority functional group.Determine and name the longest continuous carbon chain…
Q: A monoprotic acid, HA, with a Ka of 3.65 × 10-5 has a distribution constant (KD) of 2.51 (favoring…
A: The distribution ratio (D) of a monoprotic acid can be calculated using the Ka value, distribution…
Q: Suppose a 250. mL flask is filled with 0.10 mol of NO2, 1.1 mol of NO and 0.80 mol of CO2. The…
A:
Q: 20. For the following chair flip, the equatorial position of the methyl group is favored over the…
A: The value of standard free energy change (ΔGo) can be calculated using the following data-Here, R is…
Q: Consider a set of 0.100 M weak acid solutions with various pk, values. Which one is the least…
A: According to Arrhenius concept, an acid is a substance i.e. when dissolved in water gives H+(aq)…
Q: What is a major difference between acid and base catalyzed hydration of a carbonyl?
A: Given question is based on organic chemistry.The given reaction is hydration of ketone.The hydration…
Q: For the mechanism, draw the curved arrows as needed. Include lone pairs and charges in your answer.…
A: The objective of the question is to predict a suitable synthesis mechanism for the given reaction.
Q: 2. 13.9 g of ammonium nitrate is dissolved in a total volume of 225 mL in a coffee cup calorimeter.…
A: Given:Mass of ammonium nitrate dissolved = 13.9 gTotal volume = 225 mLInitial temperature of the…
Q: Draw C4H8Br2 that has the following 'H NMR spectrum: 1.97 (s, 3H), 3.89 (s, 1H). Hint: Draw C4H10…
A: Given :Molecule : Objective : Determine the structural formula.
Q: Propose a synthesis for the following molecules, starting from benzene or a named varian
A: The objective of this question is to propose syntheses for specific molecules starting from benzene…
Q: The heat of vaporization of water at 100°C is 40.66 kJ/mol. Calculate the quantity of heat that is…
A: Given,mass of steam water = 5.00 gThe heat of vaporization of water at 100 oC = 40.66 kJ/mol
Q: 4. Extraction: a) ( ) In what order (from lowest to highest R,) would you expect to find compound X,…
A: Thin layer chromatography (TLC) is a method of separating components of the mixture. It is based on…
Q: 3. (Sections 19.6 and 19.7) Draw the mechanism for the Wolff-Kishner reaction in a., being sure to…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: 2. 13.9 g of ammonium nitrate is dissolved in a total volume of 225 mL in a coffee cup calorimeter.…
A: The objective of this question is to find the final temperature of the solution when ammonium…
Q: Determine the number of atoms of O in 79.8 moles of Cr₃(PO₄)₂..
A: Atoms are made up of electrons, protons and neutrons. Protons are positively charged particles,…
Q: Draw and write only the main product of the reaction (C) Only typed solution
A: Organic reactions are reactions in which organic reactants react with each other to produce organic…
Q: At the same concentration, which of the following aqueous solutions has the lowest pH? O HNO3…
A: pH stands for power of hydrogen. pH is defined as negative of logarithm of hydrogen ions.…
Q: When 15.0 mL of a 2.53×10-4 M lead nitrate solution is combined with 15.0 mL of a 1.91×10-4 M sodium…
A: No precipitate will be formed. Q value is 1.21x10-8.Explanation:Given:…
Q: Gases in a Salt Marsh: You measure appreciable CH 4(g) andCO 2(g) partial pressures in the sediment…
A: The objective of the question is to calculate the redox potential (Eh) from the ratio of the…
Q: IUPAC name: 이 2. 30 H2N ₂N H
A: IUPAC (International Union of Pure and Applied Chemistry) naming, also known as systematic naming or…
Q: A 360.0 mL buffer solution is 0.170 mol L-¹ in HCOOH and 0.170 mol L-¹ in HCOOK. K.(HCOOH) = 1.8 ×…
A: The mass of NaOH that could be added before the pH rises above 4.00 is approximately 2.45…
Q: Identify the correct dipole for the C-N bond in the Lewis structure of hydrogen cyanide shown using…
A: The objective of the question is to identify the correct dipole for .
Q: Predict the FINAL (?) product for each of the following reaction or synthetic chain: +MgBr 1. 03 ?…
A: Given,The reaction:
Q: STARTING AMOUNT Determine the number of moles in 4.21 x 103 molecules of CaCl2
A: The objective of the question is to find out the number of moles in a given number of molecules of a…
Q: Draw the product of this reaction and account for its formation, providing a curly arrow mechanism…
A: Reaction of benzene ring containing halogen group attached leads to the formation of benzyne as…
Q: 1) For each pair of structures, determine if they are identical (I), resonance forms (RF),…
A: We have to choose the correct relationship for each pair.
Q: What substitution product are you expecting from the reaction below? OH CI Хосно NaOCH 3 This…
A: The leaving group should be good or moderate
Q: Determine the shape of the complex, [MnBr2(en)2] Select one: O a. linear O b. either tetrahedral or…
A: The objective of the question is to determine the shape of the given complex [MnBr2(en)2].
Q: For the reaction below, Kc = 0.60 at 550 K. The reaction starts with a 0.050 M concentration of…
A: The chemical equilibrium constant, often denoted as Kc for concentration equilibrium, is a…
Q: What is the hybriziation of the carbon 1 atom
A: Hybridization is the concept of mixing atomic orbitals to form an equal number of hybrid orbitals…
Q: What quantity in moles of precipitate will be formed when 104.0 mL of 0.150 M LiBr is reacted with…
A: The objective of this question is to determine the quantity in moles of precipitate (PbBr2) that…
Q: A voltaic cell using Pb2+/Pb and Ni²+/Ni half-cells is set up at standard conditions, and each…
A: Answer:Voltaic cell is a type of electrical cell that converts chemical energy into electrical…
Q: Draw step 1 of the mechanism by completing the starting materials. H₂C R NH + H₂C CH3 CH3 CH3 Edit…
A: The question is based on Acid-Base reaction in the organic chemistry.
Q: STARTING AMOUNT How many atoms of vanadium are in 1.28 grams of vanadium ?
A: We have to find how many atoms of vanadium are in 1.28 grams of vanadium.
Q: Balance the following reaction KMnO4(aq) + KOH(aq) + KI()+ KMnO4(aq) + KIO(aq) + H2O(I) Express your…
A: Given ,Balanced reaction : Objective : Determine the atom economy based on as the desired…
Q: Please don't provide handwritten solution ....
A: The objective of the question is to calculate the equilibrium constant in terms of concentration…
Q: What is the molar solubility of lead(II) bromide PbBr2 Ksp = 4.67 x 10-6.
A: The objective of this question is to calculate the molar solubility of lead(II) bromide (PbBr2)…
Q: 14 CH3OH H₂SO4
A:
Q: What is the formal charge in this nitrogen atom?
A: The formal charge can be calculated using the expression:Formal charge = [Total no. of valence…
Q: Which of the following molecules represents the most oxidized state? a. LOH b. C. ΝΗ d. Select an…
A: The term "oxidized state" refers to the condition of an atom or molecule when it loses electrons…
Q: Complete this table. name sodium chloride empirical formula ☐ lead(II) iodide ☐
A: An ionic compound is formed by the electrostatic interaction between the cation and the anion. A…
Q: Part C MISSED THIS? Watch KCV: The Titration of a Weak Acid and a Strong Base, IWE: Weak Acid-Strong…
A: The question is based on the concept of the pH of the solution.It is defined as a negative logarithm…
Q: Check the box next to each molecule on the right that has the shape of the model molecule on the…
A:
Q: 0: What is the major organic product obtained from the following reaction? 1)NaCN H 2) H* Heat
A: When ketone react with NaCN / H+ form cyanohydrin .
Q: 4. A 500 ml solution of KOH was found to have a pH of 12.55. Calculate the concentration of [OH] in…
A: The pH value of a solution can be used as a measure of the concentration of hydronium ion (H3O+)…
Q: What is the main product of the following reaction? (pay attention to regiochemistry and…
A: The question is based on pericyclic reaction.The Diels-Alder reaction is a type of cycloaddition…
Please give the following structures IUPAC names, provided with an explanation
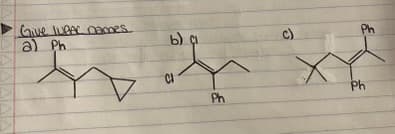
Step by step
Solved in 1 steps with 2 images

- Give both IUPAC names and common names for the following compounds PhCO2KWhat is CH3CH2NH3+ classifed as? Group of answer choices strong acid weak acid strong base weak base negligible salt spectatorIn a 0.28 mM aqueous solution of trimethylacetic acid (C4H9CO2H), what is the percentage of trimethylacetic acid that is dissociated?

