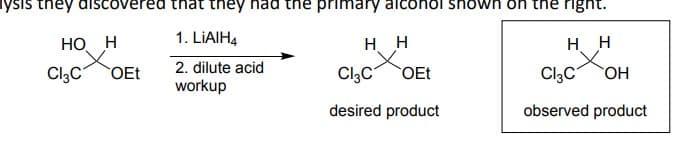
lysis they discovered that they had the primary alcohol shown on the right. HO H 1. LiAlH4 H H Cl3C OEt 2. dilute acid workup Cl3C OEt HH Cl3C OH desired product observed product
lysis they discovered that they had the primary alcohol shown on the right. HO H 1. LiAlH4 H H Cl3C OEt 2. dilute acid workup Cl3C OEt HH Cl3C OH desired product observed product
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter8: Addition Via Carbocation
Section: Chapter Questions
Problem 9CTQ
Related questions
Question
Explain what happens so as indicated in the picture( why the desired product wasn't formed but instead the observed product only formed)
What chemistry could we form to get the desired product ?
Transcribed Image Text:lysis they discovered that they had the primary alcohol shown on the right.
HO H
1. LiAlH4
H H
Cl3C
OEt
2. dilute acid
workup
Cl3C
OEt
HH
Cl3C OH
desired product
observed product
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 1 steps

Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning
