Q: A student suggests that the molecule on the right can be made from a single molecule that doesn't…
A: The proper structure is drawn out in section below -Explanation:Here is the correct structure which…
Q: OCH3 E NH2 -B(OH)2 NiCl2(PCY3)2 Na2CO3, THF NH2 ала OH 1) SOCI 2, Et₂O 2) EtOH NaOH, H₂O O₂N SnCl2…
A: Suzuki coupling reactionAmide formationAmide formation Ester formationEster hydrolysisReduction of…
Q: Please help me , I need fast solution. Please don't parovide handwritten solution ....
A: The objective of the question is to complete the given chemical reaction involving CH2I2, Zn(Cu) and…
Q: Suppose a 500. mL flask is filled with 1.7 mol of H2 and 0.90 mol of NH3. This reaction becomes…
A:
Q: 1. Calculate the concentration of a solution of H₂SO, with a pH of 1.06. 2. A solution of NaOH has a…
A: The pH value of a solution can be used as a measure of the concentration of hydronium ion (H3O+)…
Q: Determine the normal boiling point of a substance whose vapor pressure is 75.1 mm Hg at 35°C and has…
A: Clausius-Clapeyron equation pertains to the relationship between pressure and temperature. It is…
Q: Use the following data, AHfus AHvap Metal Tm (K) Tb (K) (kJ/mol) (kJ/mol) Li 454 2.99 1615 134.7 Na…
A: The objective of the question is to calculate-The entropy change for the fusion of Rubidium.The…
Q: Only typed solution
A: The objective of the question is to understand the reasons behind the increase in pH and PO2 in the…
Q: A chemical reaction is spontaneous
A: A spontaneous reaction occurs when favorable conditions allow for the production of products,…
Q: What would be the major product of the following reaction sequence? 1.03 2. (CH3)2S ? O by H مہد H H
A: Ozonolysis is an organic reaction where the unsaturated bonds are cleaved with ozone (O3). Multiple…
Q: A 0.2415 g sample of solid magnesium is burned in a constant-volume bomb calorimeter that has a heat…
A: for given magnesium sample Heat on changing given temperature difference is,1. Heat = 1.44 KJ/g2.…
Q: arrow. F Cl HS HC H SH Part 1 of 2 Draw the Newman projection for a staggered conformation of the…
A: The objective of the question is to find the Newman projection for one staggered and one eclipsed…
Q: Which of the following Lewis structures will have resonance form H | H - с O-H H H-CEN - H C H ==0
A: Resonance structure : It is the group of more than one Lewis structure , that is representing a…
Q: Assuming complete dissociation, what is the pH of a 3.09 mg/L Ba(OH)2 solution? pH =
A:
Q: Which of the following strategies is NOT expected to effect the reduction of 4-tert-butanol to…
A: The Wolff-Kishner reduction is a method used to convert a carbonyl group (C=O) in an aldehyde or…
Q: QNo: treatment treatment of compound F Pylamide followed by Cyclohexane with lithiu gives
A: The objective of this question is to elucidate the mechanisms behind the formation of ethers via…
Q: Give the products of periodic acid oxidation of each of the following. How many moles of reagent…
A: The objective of the question is to determine the products of periodic acid oxidation of the given…
Q: Draw and write down the expanded formula (see previous question for expanded formula) for the…
A: In an expanded molecular formula, all the atoms and bonds within the molecule are shown.
Q: Draw the major product of this reaction. Ignore inorganic byproducts and the alcohol side product.…
A: LiAlH4 (Lithium aluminium hydride) is a strong reducing reagent which reduces carboxylic acid into…
Q: Part 1 Select the carbons which contain hydrogen atoms that could be eliminated in an electrophilic…
A: In the given question we have to predict the hydrogen atoms that will be eliminated in an…
Q: An aluminum cup weighing 60.0 g is filled with 30.0 g of water. How much energy is needed to raise…
A: FINAL ANSWER: How much energy is needed to raise the temperature of the water filed cup from…
Q: CHEM 372-Problem Set 13 - Ester and amide hydrolysis 4. Esters undergo hydrolysis to the…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: 1. Calculate the concentration of a solution of H₂SO, with a pH of 1.06.
A: 1) pH = - log [H+] Thus, [H+] = 10-pH
Q: Why is the electon-pair geometry for NH, tetrahedral but the electron- pair geometry for BH, is…
A: Given compound and electron pair geometry,1) 2) Valence eelctron in Valence electrons in Valence…
Q: For the following chair flip, the equatorial position of the methyl group is favored over the axial…
A: The value of standard free energy change (ΔGo) indicates the spontaneity of a reaction. A negative…
Q: Positronium is an atom composed of an electron and a positron (mp = me Q = +e). Calculate the…
A: The reduced mass of the positronium is The distance between the particles is inversely proportional…
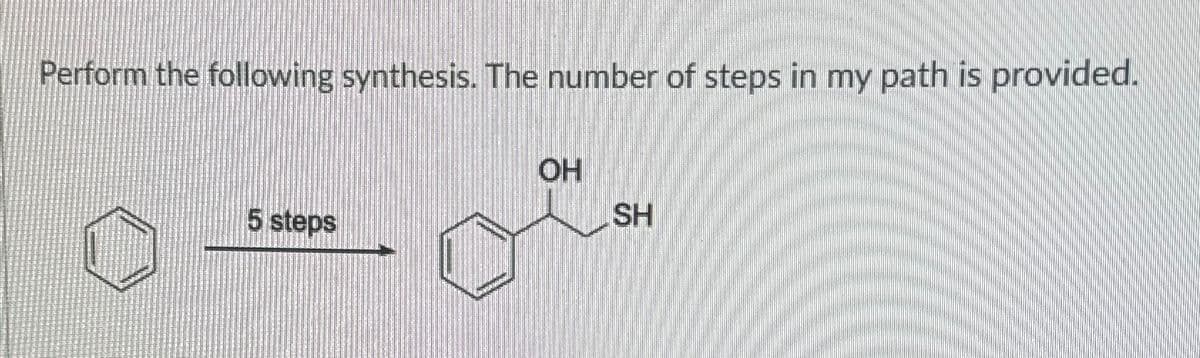
Q: Hi can someone please show the synthesis steps using the grinard reagent and oxidation, etc? from
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: For the reaction 8 H₂S(g) 8H2(g) + S, (g) the equilibrium concentrations were found to be [H,S] =…
A: The equilibrium constant for a reaction is the ratio of the equilibrium concentrations of the…
Q: Part A OH Spell out the full name of the compound. hept-2-en-4-ol Submit Previous Answers Request…
A: (2E)-hept-2-en-4-olExplanation:
Q: When a solid dissolves in water, heat may be evolved or absorbed. The heat of dissolution…
A: Given,The mass of K2SO4 = 9.07 gThe mass of water = 110.50 gInitial temperature of the solution =…
Q: Paylben
A: In the given reaction, there is an aldehyde, a ketone, and a secondary amine group which react to…
Q: Only typed solution
A: The detailed explanation given belowExplanation:Step 1:To calculate the heat required to vaporize…
Q: Find △°Arxn and △°Grxn at 298 from (table) enthalpies of formation and the absolute entropies of…
A: The answer in the explanation part.Explanation:
Q: Consider the following reaction. aA +bB→ CC whose first and second half-lives are 22.3 and 11.15…
A: The objective of the question is to calculate the concentration of reactant A at a given time in a…
Q: A 510 mL buffer solution is 0.105 M in HNO2 and 0.165 M in KNO Identify whether each addition would…
A: We can calculate moles of any substance using the formula below: n=MV, where n is the number of…
Q: 11. Treatment of compound F with lithium diisopropylamide followed by cyclohexanone gives either G…
A: The objective of this question is to explain the processes behind the aldol condensation and…
Q: Draw the major product of this reaction. Ignore inorganic byproducts. CI H₂O pyridine 0
A: The objective of the question is to find the major product of the given reaction substituted acid…
Q: You will dilute ~1.00 g of solid acid in a flask. To what volume in mL will you dilute it? Type…
A: To calculate the volume of solution needed to dilute 1.00 g of solid acid, you can follow these…
Q: A monoprotic acid, HA, with a Ka of 3.65 × 10-5 has a distribution constant (KD) of 2.51 (favoring…
A: The distribution ratio (D) of a monoprotic acid can be calculated using the Ka value, distribution…
Q: Ammonia will decompose into nitrogen and hydrogen at high temperature. An industrial chemist…
A: The equilibrium reaction can be written as follows:The given moles of ammonia = 6.2molThe volume of…
Q: Which of the following element(s) are typically added to gray iron to make it stronger and more…
A: Gray iron also called as gray cast iron has graphitic microstructure.Gray iron has high thermal…
Q: 2. 13.9 g of ammonium nitrate is dissolved in a total volume of 225 mL in a coffee cup calorimeter.…
A: The objective of this question is to find the final temperature of the solution when ammonium…
Q: Only typed solution
A: The objective of the question is to understand the reasons behind the increase in pH and PO2 in the…
Q: Macmillan Learning What is the IUPAC name for the compound shown? HICH HICH H H H H C. CIH HICH…
A: The objective of the question is to determine the IUPAC (International Union of Pure and Applied…
Q: Click on all the atoms that are EXPLICITLY shown in this molecule that have a positive partial…
A: In the given question we have to identify the atoms that have a partial positive charge.The covalent…
Q: A beaker with 1.00×102 mL of an acetic acid buffer with a pH of 5.000 is sitting on a benchtop. The…
A:
Q: 1. Predict the product and provide the mechanism for the following reversible nucleophilic addition.…
A: Since you have asked multiple question, we will answer the first question for you. If you want any…
Q: An organic chemistry Teaching Assistant (TA) suggested in your last discussion section that there is…
A: The objective of this question is to draw the product obtained from the given reaction.
Q: A B D Fraction of acetic acid reacted Attempt 4 Which of the following statements are true about…
A: Acetic acid is CH3COOH. It is a weak acid.The degree of dissociation of an acid is determined by its…
Q: PLS HELP ASAP ON ALL ASKED QUESTIONS AND SHOW ALL WORK
A: Final answer is given in explanation please see from there.Explanation:Approach to solving the…
Step by step
Solved in 4 steps with 2 images


