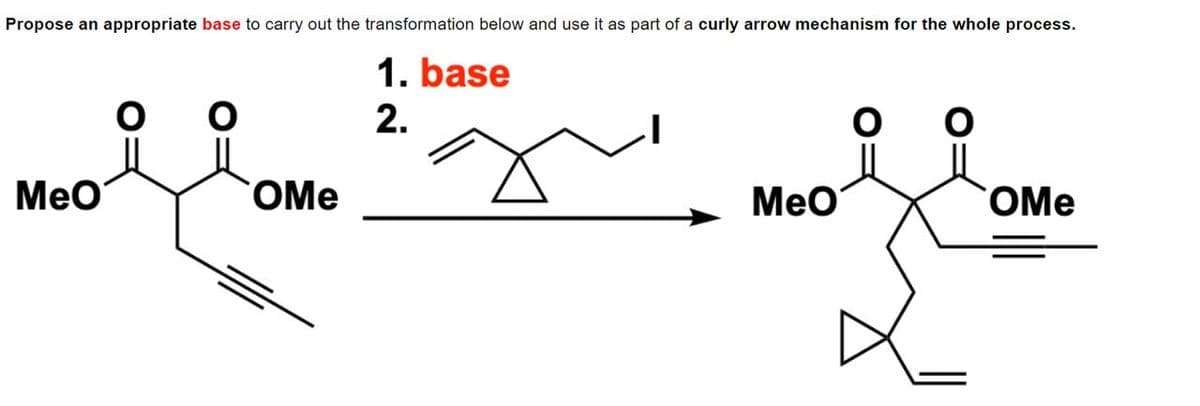
Propose an appropriate base to carry out the transformation below and use it as part of a curly arrow mechanism for the whole process. Meo ༠ OMe 1. base 2. ' ', Meo OMe
Q: Provide a curly arrow mechanism for this transformation, which is from a total synthesis of the…
A: This is an example of ketal formation. In a ketal formation reaction a ketone in the presence of…
Q: 1) For each pair of structures, determine if they are identical (I), resonance forms (RF),…
A: Note: Since you have posted a question with multiple sub parts, we will provide the solution only to…
Q: Give correct detailed Solution with explanation needed .don't give Handwritten answer..don't use Ai…
A: The objective of the question is to determine where the tail of the curved arrow would be placed in…
Q: P1A.12* Balloons are still used to deploy sensors that monitor meteorological phenomena and the…
A: (a) Volume of balloon is:
Q: Provide a synthesis of the following compound using the allowed starting materials: OH H of H OH H…
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: The most important commercial process for converting N₂ from the air into nitrogen-containing…
A: N2(g) + 3H2(g) ----> 2NH3(g)moles of N2 react = 3.0 molmoles of H2 react = 6.0 molmoles of NH3…
Q: Name the following molecule CH
A: Organic molecules can be defined as molecules that contain carbon and hydrogen atoms. We have been…
Q: The chemical formulae of some acids are listed in the first column of the table below, and in the…
A: Given ,Chemical formula of some acid.Objective : Determine the species present in solution when…
Q: Three reactions between a Grignard reagent and a carbonyl compound are given. Draw the main organic…
A: The objective of the question is to predict the product formed in the given reaction.
Q: Draw a structural formula of the S configuration of the compound shown below. CH₂CI
A: Given ,Compound : Objective : Determine the S configuration of the compound given.
Q: For the SN2 reaction, draw the major organic product and select the correct (R) or (S) designation…
A:
Q: How many grams of testosterone, C19 H28 O2, a nonvolatile, nonelectrolyte (MW = 288.4 g/mol), must…
A: The mass of testosterone is 15.38 g. Explanation:Given:…
Q: What are the effects of aerosols on climate change?
A: An aerosol is a colloid of fine solid particles or liquid droplets, in air or another gas.
Q: Using the two images attached please write one paragraph EXPLAINING THE ANALYSIS Please please…
A: The objective of this question is to understand the principles and procedures involved in the…
Q: Draw the major product of this reaction. Ignore inorganic byproducts.
A: LDA is a sterically hindered strong base and it is used in the formation of the kinetic enolate ion.
Q: What is the hybriziation of the carbon 1 atom
A: Hybridization is the concept of mixing atomic orbitals to form an equal number of hybrid orbitals…
Q: What is the major product? HNO3 Sn ? HC1 H₂SO NO2 NH2 NO₂ 8628 b C NO₂ d
A: ->Aromatic electrophilic substitution reaction in which an electrophile replaces a hydrogen atom…
Q: Draw the product of this reaction and account for its formation, providing a curly arrow mechanism…
A: Reaction of benzene ring containing halogen group attached leads to the formation of benzyne as…
Q: calculate the ph for each of the cases in the titration of 25.0 ml of .140 M pyrimidine with .140 M…
A: Since you have posted a question with multiple sub-parts, we will provide the solution only for the…
Q: Draw the mechanism of the acid catalyzed hydrolysis of the two following compounds. The products…
A: In the given question we have to do the acid catalysed hydrolysis of ester and amide functional…
Q: Gluconic acid is a substance in honey that prevents is from spoiling. The pKa of gluconic acid is…
A: Given that,The pKa of gluconic acid = The concentration of gluconic acid,The pH of the gluconic acid…
Q: How many molecules are in 79.6 moles of CO₂?
A: The objective of this question is to calculate the number of molecules in a given amount of moles of…
Q: How many different types of carbons does the following compound have?
A: 13C NMR spectroscopy is used to determine the number of distinct carbon atoms in a molecule and the…
Q: 1 & 2. Identify the steps necessary to prepare compound X. Show reagents and reaction conditions.…
A: To approach the product from cyclopent-1-en-1-ylbenzene to…
Q: Problem 206 of 36 Draw the major product of this reaction. Ignore inorganic byproducts. HCI, H₂O + a…
A: The given reaction is:Identify the major product of the reaction.The given reactant is enamine.…
Q: ADD FACTOR *( ) ANSWER RESET > 1 6.99 110.98 4.21 x 1023 75.53 0.699 6.022 x 1023 77.6 3.79 × 1021 g…
A:
Q: Provide the expected major organic product of the reaction sequence shown. 1. 03 2. (CH3)2S, (DMS) ?…
A:
Q: t ot Visited ball & stick✓ +labels Name the alkene from which the 1,2-diol above was made. Submit a…
A:
Q: Draw the organic product you would expect to isolate from the nucleophilic substitution reaction…
A: Nucleophilic substitution is a fundamental class of organic reactions in which a leaving group is…
Q: P.25 An HF molecule, with a bond length of 91.68 pm, absorbed on a surface rotates in two…
A: An 1H19F molecule with a bond length of 91.68 pm.It is absorbed on a surface that rotates in 2…
Q: Which alkyl bromide is necessary to complete the synthesis shown in the box? A A BB с с D D 1) Li 2)…
A: The reaction is three steps synthesis.
Q: Among the molecules below, which one should have the highest boiling point?
A: Organic molecules are molecules that contain carbon and hydrogen atoms. We have been asked to choose…
Q: 1. Predict the product and provide the mechanism for the following reversible nucleophilic addition.…
A: Since you have asked multiple question, we will answer the first question for you. If you want any…
Q: A 1.0 L buffer is prepared that is 0.85 M in formic acid, HCOOH and 1.4 M in sodium formate. Find…
A: The objective of the question is to calculate the initial pH of the buffer solution and the pH after…
Q: Choose the correct chemical structure for every single acronym used below:
A: Acronyms are a type of abbreviation that is used to abbreviate chemical structures , these…
Q: What quantity in moles of CH NHCI need to be added to 200.0 mL of a 0.500 M solution of CH NH2 (Kb…
A:
Q: Malic acid, (HO2C)CH2CH(OH) (CCH) has two acidic protons. What is the pKa for the following…
A: The pKa value is a measure of acidic strength of a solution. The higher the pKa value, the lower the…
Q: Which is the stereochemical relationship between this pair of molecules? OH OH A) enantiomers B)…
A: The given structures are:Identify the stereochemical relationship between these two pairs of…
Q: Calculate the pH for each of the cases in the titration of 25.0 mL. of 0.220 M pyridine, CH,N(aq)…
A:
Q: Predict the product of the following Wittig reaction. Be sure your answer accounts for…
A: cyclopent-1-en-1-ylbenzene will formExplanation:
Q: The rate of a reaction increases by 4 times when temperature of the reaction is raised from 340K to…
A:
Q: 0 When 4.18 g of a certain molecular compound X are dissolved in 45.0 g of benzene (C6H6), the…
A:
Q: Match the lipid pathway with its description Question 27 options: dietary…
A: The objective of the question is to match the lipid pathway with its corresponding description.
Q: er Write the mass balance for a K2CO3 solution if the species in the solution are K+, CO32-, HCO3,…
A: The species in the K2CO3 solution are:K+CO32-HCO3-H2CO3 H+OH-We need to calculate the mass balance…
Q: A 20.0 mL sample of 0.150 M ethylamine CH3CH2NH2 is titrated with 0.0981 M HCl. What is the pH after…
A: The objective of this question is to calculate the pH of a solution of ethylamine before any HCl is…
Q: A molecule phosphoresces with a single peak wavelength of 700 nm. The single fluorescence peak is at…
A: The answer is given in attachment below.Explanation:Step 1:Step 2: Step 3: Step 4:
Q: Write the formula correctly for the following like shown in the image below.…
A: Find the highest priority functional group.Determine and name the longest continuous carbon chain…
Q: If 35.0 g of NaOH is added to 0.750 L of 1.00 M Cd(NO₃)₂, how many grams of Cd(OH)₂ will be formed…
A: Answer: 64.1 g 64.1 g of Cd(OH)2 will be producedExplanation:Step 1:Step 2:Step 3:
Q: 4. An enzyme lowers the activation energy barrier for a chemical reaction. In addition, it cha the…
A: The objective of the question is to estimate the given statement about working and affect of enzyme…
Q: How many kJ of heat is needed to raise the temperature of 0.250 L of water in t liquid phase from…
A: Volume of water V = 0.250 LT1 = 24.6 oCT2 = 46.2 oC
Step by step
Solved in 3 steps with 1 images

- If the structure below is subjected to retrosynthetic analysis, which reagent is least useful?1.Explain why pyridine ( Kb=2.3x10-9) is a much stronger base than pyrrole (Kb=2.5x10-14), 2.Explain and illustrate, why it is difficult to perfom Friedel-Craft reactions on unactivated pyridine.Do a retrosynthetic analysis on the following compound, ending with the given starting material:
- Design a synthesis that would lead to the formation of the desired product. Please provide arrow pushing mechanism and provide reasoning for each step.Can someone help with the synthesis of this problem please?Show an actual arrow-pushing mechanism for the transformation below, and briefly explain the observed regioselectivity.
- Please perform a retrosynthesis for the image below including all the steps and reagentsMolecule 4 was prepared via molecule 3. Provide a complete curved arrow mechanism for the most straightforward conversion to 4. You can use HB+ as your acid and B: as your baseClassify following solvent as protic or aprotic CH3NO2
- Do a retrosynthetic analysis on the following compound, ending withavailable starting material.Perform a retrosynthetic analysis and suggest a synthesis of the target molecule (on the left) from the given starting material (on the right). (any reagents maybe usedShow how HC≡CH, CH3CH2Br, and (CH3)2CHCH2CH2Br can be used to prepare CH3CH2C≡CCH2CH2CH(CH3)2. Show all reagents, and use curved arrows to show movement of electron pairs.

