Q: Calculate the pH during the titration of 100.0 mL of 0.200 M HCl with 0.400 M NaOH. First what is…
A: PH of HCl before NaOH added is 1.69Explanation:Formula:Moles=molarity×volumen=M×VpH=−log[H+]Now ,…
Q: NH3 is a weak base (K₁ = 1.8 x 10-5) and so the salt NH4Cl acts as a weak acid. What is the pH of a…
A: NH4Cl is a salt of weak base NH3 and strong acid HCl. So in an aqueous solution of the salt the salt…
Q: Find the rate of reaction increased when the temperature coefficient of a reaction is 3.The…
A:
Q: 21. Which compounds below have both a cis and a trans isomer? 1 A. 1, 2, and 4 B. 2, 3 and 4 1 and 4…
A: Cis-trans isomers are a type of stereoisomerism in which atoms have different spatial arrangements…
Q: 1. Calculate the concentration of a solution of H₂SO, with a pH of 1.06.
A: 1) pH = - log [H+] Thus, [H+] = 10-pH
Q: 7. A 10.0 cm³ sample of 0.150 mol dm³ aminoethanoic acid with a pH of 5.3 was titrated with 0.100…
A: The objective of this question is to understand how the pH changes during the titration of…
Q: Can you show the stereoisomers and their stereochemistry?
A: Isomers having the same molecular formula and constitution but they differ in their space or…
Q: The fusion of one H-3 atom and one H-1 atom produces
A: The question is asking about the product of a nuclear fusion reaction between an atom of hydrogen-3…
Q: What reagent(s) can be used for the following transformation? H₂C Selected Answers: i) LAH, Et2O ii)…
A: We have to choose the correct reagent for the given transformation.
Q: In first image draw and name the following image The second question is 2. Draw and write down the…
A: “Since you have posted a question with multiple sub parts, we will provide the solution only to the…
Q: Identify the number of stereoisomers expected for the following: OH OH
A: The objective of the question is to identify the number of stereoisomers of the given molecule.
Q: Which of the following is considered to be a thermodynamic product of the reaction shown. DBr OA OB…
A: Diene shows the electrophilic addition reaction such addition of X2, HX etc. The addition of HX to…
Q: Assuming complete dissociation, what is the pH of a 3.09 mg/L Ba(OH)2 solution?
A: Concentration of Ba(OH)2 = 3.09 mg/Lmolar mass of Ba(OH)2 = 171.34 g/mol1 g = 1000 mg
Q: Benzoic acid, C6H5COOH, has a Ka = 6.3 x 10-5, What is the percent ionized in a 0.95 M solution of…
A: Final answer is given in explanation please see from there.Explanation:Approach to solving the…
Q: arrow. F Cl HS HC H SH Part 1 of 2 Draw the Newman projection for a staggered conformation of the…
A: The objective of the question is to find the Newman projection for one staggered and one eclipsed…
Q: This is the structure of 1-nonyne. Part: 0/2 Part 1 of 2 1-Nonyne reacts rapidly with NaH, forming a…
A: The given reaction is a reaction between an alkyne and sodium hydride (NaH). When alkyne reacts with…
Q: or the product, triphenylmethanol, what peaks should we see in 1) 1H NMR 2) 13C NMR, and 3) IR…
A: The objective is to assess the understanding of spectroscopic techniques () for identifying…
Q: 3. Draw the following compounds: a. cyclooctanamine b. triphenylamine c. N,N-dimethyl 2-butanamine…
A: Answer:-This question is answered by using the simple concept of writing the structure of the…
Q: ADD FACTOR *( ) ANSWER RESET > 1 6.99 110.98 4.21 x 1023 75.53 0.699 6.022 x 1023 77.6 3.79 × 1021 g…
A:
Q: Which of the structures below are drawn incorrectly using the conventions of line angle drawings?…
A: The proposed structure must adhere to the fundamental chemical rules and principles, such as the…
Q: Which Marijuana preparation has the highest content of THC? 1. Charas 2. Liquid hashish 3. Ganja 4.…
A: Answer: The most THC-rich form of marijuana is usually liquid hashish. Liquid hashish is a…
Q: PLS HELP ASAP ON ALL ASKED QUESTIONS AND SHOW ALL WORK
A: 3. c. the rate at which reactants are consumed 4. d. decreasing the activation energy of the…
Q: A key intermediate in the Wittig reaction is called an oxaphosphetane.Draw both products using…
A: The required answer is given below Explanation:Step 1: Step 2: Step 3: Step 4:
Q: PLS HELP ASAP ON ALL ASKED QUESTIONS AND SHOW ALL WORK
A: 9. d. mol/L.s 10. d. Both (a) and (b) are correctExplanation:9. The rate of reaction refers to the…
Q: 3. Complete the following table: Fill in the mL used of the two starting materials (from the…
A: The answers and calculations are as follows.Explanation:
Q: Which compound would be most reactive toward autooxidation? о
A:
Q: In the laboratory you are given the task of separating Ag* and Ba²+ ions in aqueous solution. For…
A: According to solubility rules, All sulfates are soluble except Ca+2, Ba+2, Ag+, Pb+2, Sr+2, Hg+2.…
Q: Homework: Construct a titration curve for 10 mL 0.01 M NaNH₂ titrated by 0.01 M HCl in NH3 solvent.…
A: As per the given question i was provided the solution i hope it will helps you.Explanation:Step…
Q: 9. Mechanism: Propose a mechanism for the following transformation. Br L Br H₂O
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: For the reaction below, Kc = 0.60 at 550 K. The reaction starts with a 0.050 M concentration of A2E…
A: For the equilibrium reaction:A2B2 (g) 2AB (g)Kc = 0.60Concentration of A2B2 = 0.050 MTemperature =…
Q: Decide whether this proposed Lewis structure is reasonable. proposed Lewis structure Is the proposed…
A: Lewis’s bonding theory is based on the octet rule. The Lewis structure is a simplified…
Q: Draw the major product of this reaction. Ignore inorganic byproducts.
A: LDA is a sterically hindered strong base and it is used in the formation of the kinetic enolate ion.
Q: Predict he MO ? & NACH Ca
A: Organic reactions can be defined as the reactions in which organic reactants react with each other…
Q: For the reaction 8 H₂S(g) 8H2(g) + S, (g) the equilibrium concentrations were found to be [H,S] =…
A: The equilibrium constant for a reaction is the ratio of the equilibrium concentrations of the…
Q: Draw the expected major elimination product and identify the mechanism. H3C CH3 CH₂OH I Select Draw…
A: Given reaction:We have to draw the expected major elimination product and identify the mechanism
Q: Choose the best classification for the molecule shown below. OH NH A) glycerophospholipid B) fatty…
A: The given molecule consist of a long-chain amino alcohol called sphingosine or dihydrosphingosine as…
Q: 3. Predict the product of the following multistep reaction. Br 1a) Mgo, ether b) Å c) H3O+ 2) DMP 3)…
A: The question is based on multiple steps synthesis in organic chemistry.
Q: Which two of the following compounds yield the same carbocation upon ionization? Check the two that…
A: -> If leaving group removed from molecule then there is formation of carbocation. If possible…
Q: Draw the major product of this reaction. Include stereochemistry if applicable. Use a dash or wedge…
A: It involves an elimination reaction - E2 (bimolecular elimination) reaction. This reaction proceeds…
Q: 1) The product of the reaction shown is which of the following? 1) NaBH4 CI 2) H3O+ a. An aldehyde…
A: C. An AlcoholExplanation:Approach to solving the question:The reaction is the reduction of carbonyl…
Q: How many chirality centers are in the following compound? 0 1 2 3 4
A: A chiral center, also known as a stereocenter, is the carbon atom within a molecule that is…
Q: Draw the mechanism of the acid catalyzed hydrolysis of the two following compounds. The products…
A: In the given question we have to do the acid catalysed hydrolysis of ester and amide functional…
Q: A compound containing Na, C and O is found to have 1.06 mol Na, 0.528 mol C, and 1.59 mol O. What is…
A: The empirical formula of a compound gives the simplest ratio of the number of different atoms…
Q: 0 NH2NH2, TSOH Drawing ૧
A: Aldehydes and ketones can be converted to a hydrazone derivative by reaction with hydrazine…
Q: Which alkyl bromide is necessary to complete the synthesis shown in the box? A A BB с с D D 1) Li 2)…
A: The reaction is three steps synthesis.
Q: 38.3 mL sample of a 0.475 M aqueous hydrofluoric acid solution is titrated with a 0.308 M aqueous…
A: We have to calculate the pH of the mixture.
Q: || CH3-C-NH2 amine H J CH3-C-CH3 I OH CH₂ ။ 0=CH–CH CH CH₂ CH=C=0=C=CH, O D
A: A functional group is a group of atoms present in the molecule with different chemical properties,…
Q: 4. Provide a mechanism for the following transformation: NaOH H2N Н20, д
A: The given question is based on nucleophilic addition reaction.
Q: Which alkyl bromide is necessary to complete the synthesis shown in the box?
A: The carbonyl compound reacts with alkyl lithium to form alcohol. Ketone reacts with alkyl lithium by…
Q: If the Ka for an acid is 0.010, what is the pK for its conjugate base? Give your answer to 2 decimal…
A: Given that,The Ka for an acid = Then,The pKb for its conjugate base =?
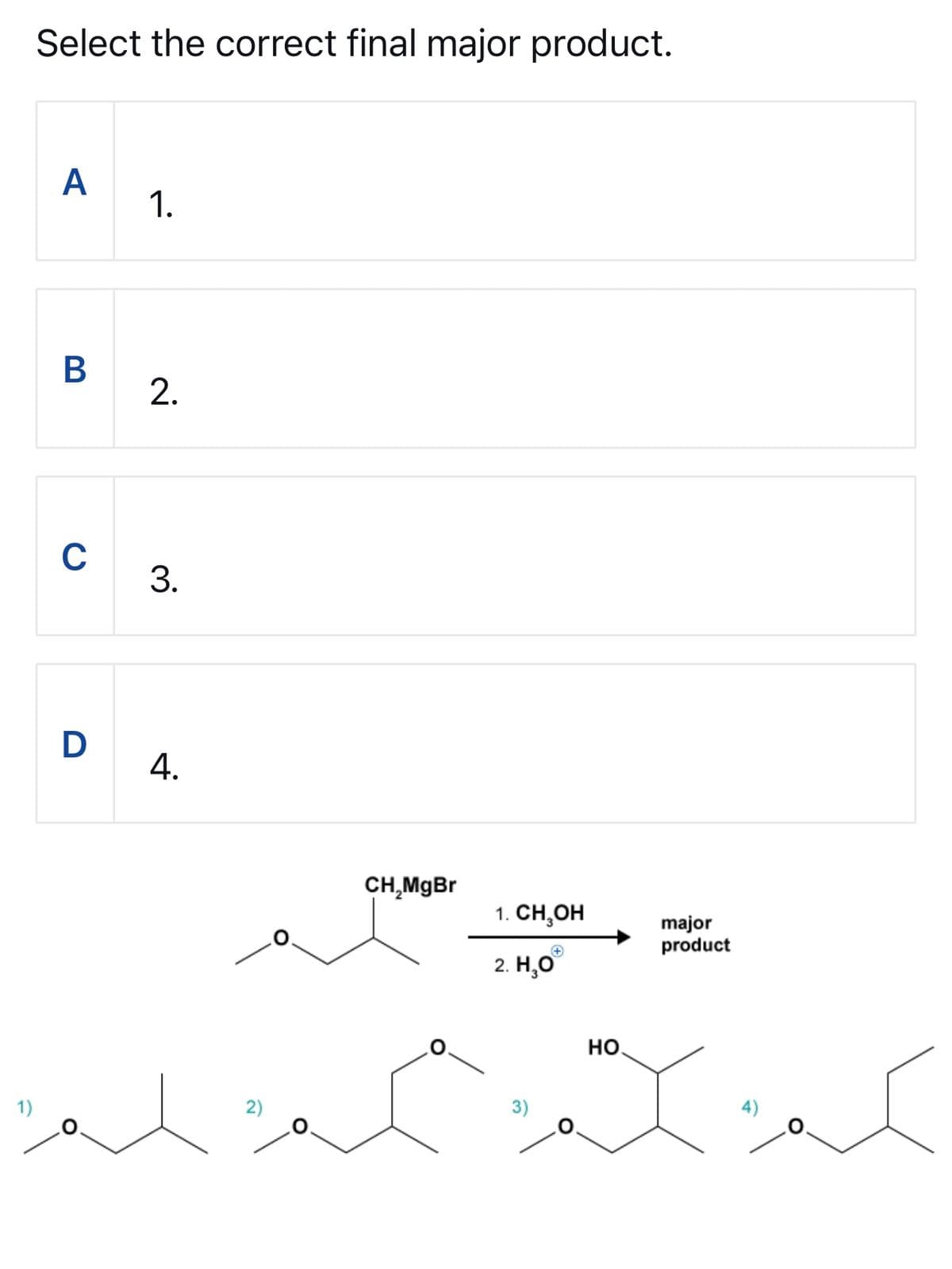
Step by step
Solved in 4 steps with 2 images

- Calculate △G° for the equilibrium reaction below. Given: CH3NH2(aq)+H2O(l)-->CH3NH3+(aq)+-OH(aq) Kb @25°C=4.4x10-4Orgonic Chemistry II: The answer is writtten as followed. But I Need Explanation. My question is that: Can I siwtch reagent 2 to reagent 1? for example reagent 1 is Cl2,Fecl3, can I changed it to reagent 2??? Why and why not???Pls ignore the highlighted part __________ is reduced in thefollowing reaction: Cr2O72- + 6S2O32-+ 14H+→ 2Cr3+ + 3S4O62- + 7H2O
- If there is no reaction from B-C that's okay. Could you do the rest of the reaction from A-E, and so on? Thank you!Consider compounds A-F. There can be more than 1 choice2A---> 2B + C (Kp1) 2D---> 2E + C (Kp2) Now find the corresponding Kp3= f(Kp1, Kp2) = ?for this reaction: D + B----> A + E elaborate throughly with every step and annotation
- Predict the product for each one clearly Please answer fast I give you upvotehelp me please ? all the reactions shall be covered in Chapter 12-15 from Organic Chemistry, 6th Edition by Marc Loudon and Jim Parise.Please help with this calculation of this, don't know to get the n(product) Page2. please help with it too Thanks in advance

