Q: The table below gives the cmc in H2O and 0.05M aq KCI as well as the aggregation number and Krafft…
A: 1.1. CMC refers to critical micelle concentration it is the minimum concentration after which…
Q: You are asked to do quality control and quality assurance during the determination of lead…
A: The limit of quantitation is given by the expression: Limit of quantitation=10 sm where,…
Q: Chloroform is an internal standard in the determination of the pesticide DDT in a polarographic…
A:
Q: kindly prepare an assignment on Application of HPLC in Drug Analysis: one page show name of the…
A: In high-performance liquid chromatography (HPLC) we inject the sample, which is in solution form,…
Q: In a spectrophotometry experiment, (34.1±0.2)% of the light (=I/Io) was transmitted through the…
A: -log10II0=εbcc=-log10II0εb
Q: The mass transfer term "C" depends on which of the following factors? A.type of column B.length of…
A: Chromatography:
Q: Please draw a calibration curve from these numbers and get the epselon(molar absorptivity) and…
A: The concentration v/s absorbance data given is,
Q: Example: A GC-FID analysis was conducted on a soil sample containing pollutant X. The following…
A: Answer -
Q: a. Find the equilibrium concentrations of SA in the aqueous and the non-aqueous phases. b.…
A: The Beer-Lambert law states that the amount of light absorbed by any substance dissolved in a…
Q: > A standard sample was prepared containing 10.0 ppm of an analyte and 15.0 ppm of an internal…
A:
Q: The following data were obtained by gas-liquid chromatography using a 40.0 cm packed column: Air i.…
A: Since you have asked multiple questions, we will answer the first one for you. To get the remaining…
Q: Calculate the pl values after addition of the following volume 0.0500 M NH41 to 30.00 mL 0.0500 M…
A: The volume of AgNO3 = 30.00 mL The molarity of AgNO3 = 0.0500 M The moles can be calculated as…
Q: Pure water containing no arsenic was spiked with 0.40 µg arsenate (AsO43) / L. Seven replicate…
A: The given information is represented as follows:
Q: Column resolution is an indicator to identify the selectivities and efficiencies of the column…
A:
Q: A/ The following results were obtained in the replicate determination of the lead content of a blood…
A:
Q: A chromatogram gives an ideal Gaussian peak with tr = 8.70 min and win = 0.19 min. How many…
A: 1. The number of theoretical plates (N) = 11616 2. The plate height of the column (H) = 0.00938 mm
Q: A chromatogram gives an ideal Gaussian peak with tR = 9.40 min and win = 0.19 min. How many…
A: Chromatography is a techniques where separation of components takes places depending on various…
Q: How long is a macrocentury, if a lecture period (50.0 min) is close to 1.00 microcentury?
A: Given: 1.00 microcentury = 50.0 min approx.
Q: A quality control (QC) sample was run in replicates of six to produce the following concentrations…
A:
Q: Alfred was tasked to synthesize at least 250.0 ppm of his novel gold nanoparticles (AuNP’s) to be…
A: The concentration of the solution is parameter that describes the amount of a substance in solution…
Q: The following data are for a liquid chromatographic column: column length 24.7 cm volume of the…
A:
Q: All of the following are classified as an instrumental method of analysis EXCEPT: O A.…
A: Answer: Different methods are used In chemical analysis and they can be classified into two…
Q: What are the benefits of Zeeman correction in electrothermal atomization (ETA)? a. Zeeman background…
A: The method used for the determination of the chromium present in the low traces and ultraraces…
Q: For indirect iodometric analysis of copper oxide, why did you not need to accurately weight out ki…
A:
Q: 7-2: Why do quantitative and qualitative analyses often require different monochromator slit widths?
A: 7-2 The monochromator is a wavelength selector that is used to select a particular wavelength from a…
Q: What was the most important parameter change that lead to the development of modern HPLC? A.…
A: answer- What was the most important parameter change that lead to the development of modern HPLC -…
Q: ) -SH -OCH₂ -NO₂ silica column 20% CHCI₂ 6 Time (min) 8 -CIN 10 -OH -CO₂CH₂-COCH, CHO 12 -NH₂ 400…
A: Silica Column is eluted by 29% CHCl3. Now in the given graph elution time of different compounds are…
Q: Choose the best answer. What are the factors that need to be considered in HPLC to ensure the two…
A: Introduction : HPLC means High pressure liquid chromatography it is used to find out the drug…
Q: Alfred was tasked to synthesize at least 250.0 ppm of his novel gold nanoparticles (AuNP’s) to be…
A: Given the absorbance of the blank solution = 0.200 First, we have to find the concentration (in…
Q: A molecular exclusion column has a diameter of 7.8 mm and a length of 30 cm. The solid portion of…
A: (a)
Q: prepare an assignment on Application of HPLC in Drug Analysis(Amiloride): one page show name of the…
A: We have to prepare an assignment on application of HPLC in Drug Analysis showing name, dosage form,…
Q: Consider a separation performed on a 45.0 mm long open tubular column with a 0.50 mm diameter and a…
A:
Q: What is the experimental purpose of constructing a standard curve? a. To achieve an appropriate…
A: The experimental purpose of constructing a standard curve.,
Q: A gel filtration column has a radius (r) of 0.80 cm and a length (l) of 20.0 cm. Calculate the total…
A:
Q: (5) If 200 g of polymer A, 300 g of polymer B, 500 g polymer C, and 100 g of polymer D are mixed,…
A: Molecular mass of polymer is a most important parameter and directly related to the physical…
Q: Under what wavelength (in nm) should a spectrophotometer be set to be able to measure the optical…
A: 1. Answer - At 600 nm wavelength (in nm) should a spectrophotometer be set to be able to measure…
Q: c Na(g) d. Nalg) The efficiency of chromatograpnic separation increases with: 2 a. Increase the…
A: The answer to the following question is-
Q: 4. For the system in question 2, estimate tlGo for micellization if the molecular weight of the…
A: Gibb’s free energy (∆G)– Formula is, ∆G=∆H-T∆S Where, ∆G= Gibb’s free energy ∆H=change in…
Q: The following data were obtained for four compounds separated Compound on a 20-m capillary column.…
A: Hello, For calculating selectivity factor, the value of tM that is the elution time of an…
Q: How long is a microcentury, if a lecture period (50.0 min) is close to 1.00 microcentury?
A:
Q: Column resolution is an indicator to identify the selectivities and efficiencies of the column…
A: Coloumn resolution is an indicator to identify selectivities and efficiencies of coloumn ability to…
Q: Based on the given preparation procedure, identify THREE mistakes that were made.
A: High-performance liquid chromatography (HPLC) is a more enhanced chromatography with a large…
Q: The following data are for a liquid chromatographic column: Length of Packing 24.7 cm Flow rate…
A:
Q: A water sample was analyzed for Fe content using the iron-phenanthroline method. The following data…
A: Volume of Stock Solution (mL) Total Volume of Standard Solution (mL) Concentration of Standard…
Q: The following data are for a GLC column: L= 35.5 cm, flow rate 0.303 mL/min A chromatogram of a…
A: Hello. Since the question contains multiple subparts, the first three parts are solved. In case the…
Q: The relative displacement (r/ro) of bovine serum albumin was observed as a function of time: t (s)…
A:
Q: What are the parameters that can be used for analytes identification and quantification using Cyclic…
A: Cyclic voltammetry can be applied to determine many reaction mechanisms when electron ttransfer is…
Q: Compound Unretained solutes Tr (min) W (min) 1.19 8.04 0.15 B. 8.26 0.15 8.43 0.16
A: Chromatography resolution implies how well two peaks gets separated. It depends on coloumn length.
-
In the following figure, which parameter of van Deemter equation is involved in these variations. Which curve of them would provide the best separation of analytes and at what linear velocity?
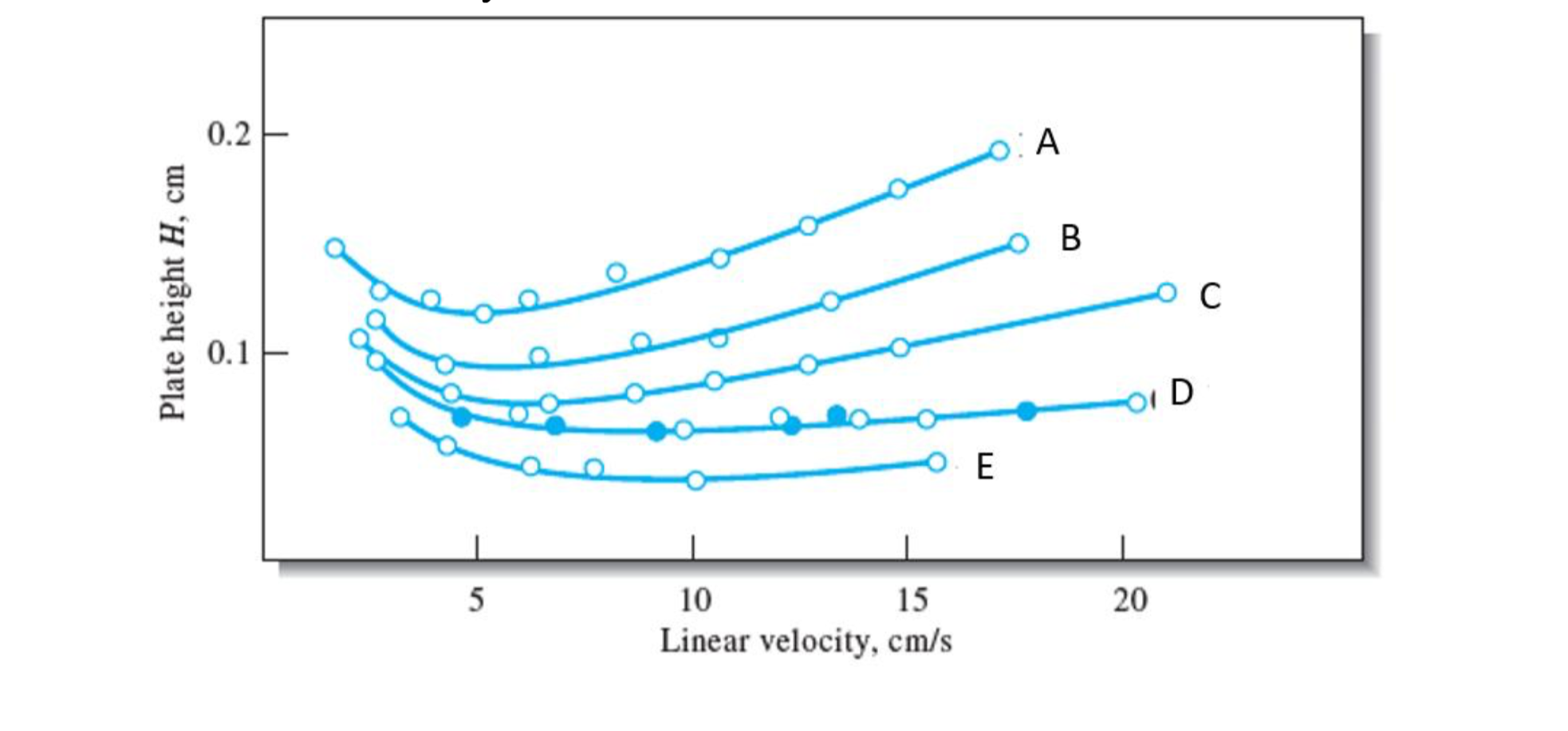
Step by step
Solved in 3 steps with 1 images

- Calculate the following exercise with correct sig figs: ((2.9365mL - 3.00mL)/3.00mL)x100%A capillary viscometer was used to measure the viscosity of a fluid at room temperature. At a pressure drop per unit length of 0.28 mmHg cm−1, i.e., ΔP/L, the measured flow rate of the fluid through the capillary viscometer was found to be 0.0125 cm3 s−1. When the value of ΔP/L was increased to 0.79 mmHg cm−1, the flow rate of the fluid was found to be .047 cm3 s−1. Under these conditions, is the fluid Newtonian or non-Newtonian? Be sure to justify your answer.For a regular cuboid particle of dimensions 1.00 x 2.00 x 4.00 mm, calculate the ff diameters: (a) the equivalent volume sphere diameter; (b) the equivalent surface sphere diameter; (c) the surface-volume diameter (the diameter of a sphere having the same external surface to volume ratio as the particle)
- What effect will the following have on plate height (H). Make sure to explain your full and complete reasoning. Increasing stationary phase thickness. Reducing the rate of sample injection. Increasing mobile phase flow rate. Decreasing temperature. Reducing stationary phase particle size.For a disc-shaped particle of diameter 2.00mm and length 0.500 mm, calculate the following diameters:a. the equivalent volume sphere diameter;b. the equivalent surface sphere diameter;c. the surface-volume diameter (the diameter of a sphere having the same external surfaceto volume ratio as the particle) d. What is the sphericity of the particle?A steel ball of density 12.45 g/mL and 2 mm diameter requires 30 sec to fall a distance of 1 meter through a liquid of density 0.789 g/mL. Calculate the viscosity of the liquid in Pa-sec.
- 5 0.0030 0.0015 0.0005 6 0.0030 0.0057 0.0089 7 0.0086 0.0057 0.0089 Show the Calculations of Qc and Kc for Exp. 5-7 Please help me out. ThanksPlot the following data and perform linear regression to obtain the value of the slope, the value of the y-intercept, and the correlation coefficient r. x- axis y-axis ln(0) 4.6052 0.0000 3.8712 0.6931 3.4012 1.0986 2.7081 1.3863 2.1972 1.6094 1.7918 1.7918 1.0986 1.9459 0.0000The thermal conductivities of human tissues vary greatly. Fat and skin have conductivities of about 0.20 W/m · K and 0.020 W/m · K respectively, while other tissues inside the body have conductivities of about 0.50 W/m · K. Assume that between the core region of the body and the skin surface lies a skin layer of 1.0 mm, fat layer of 0.50 cm, and 3.2 cm of other tissues. (a) Find the R-factor for each of these layers, and the equivalent R-factor for all layers taken together, retaining two digits. Rskin m2 · K/W Rfat m2 · K/W Rtissue m2 · K/W R m2 · K/W (b) Find the rate of energy loss when the core temperature is 37°C and the exterior temperature is 0°C. Assume that both a protective layer of clothing and an insulating layer of unmoving air are absent, and a body area of 2.0 m2. W
- y= mx+ b Slope () = -4E-09 y-intercept () = 0.4021 and OD= 0.540 , Calculate the concentration (x)If light machinery oil has a viscosity of 5E-2 Pa-s and density of 970 kg/m³ at 25°C, how long will it take for a sample to pass through a viscometer if water under this condition takes 1 min? Density of water is 1000 kg/m³ and its viscosity is 8.9E-4 Pa-s.A. 36.5 minB. 47.7 minC. 57.9 minD. 64.4 minHelp with q=mcchange in temp ?



