1. Alkaline phosphatase has a key serine residue in the active site that is critical for biological activity. Suggest a plausible four-step arrow-pushing mechanism for hydrolysis of phosphate Ser 0-00 Ser oo 0 0 0) phosphoserine intermediate
1. Alkaline phosphatase has a key serine residue in the active site that is critical for biological activity. Suggest a plausible four-step arrow-pushing mechanism for hydrolysis of phosphate Ser 0-00 Ser oo 0 0 0) phosphoserine intermediate
Chapter29: The Organic Chemistry Of Metabolic Pathways
Section29.SE: Something Extra
Problem 30MP
Related questions
Question
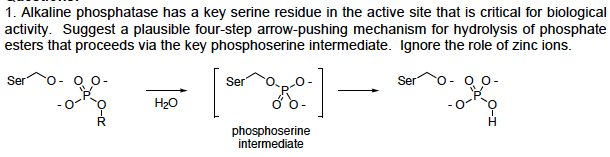
Transcribed Image Text:1. Alkaline phosphatase has a key serine residue in the active site that is critical for biological
activity. Suggest a plausible four-step arrow-pushing mechanism for hydrolysis of phosphate
Ser 0-00
Ser
oo
0
0 0)
phosphoserine
intermediate
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you


EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT



EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT
