1. Carefully examine the following table, which represents a process in multiple ways-words, sketches, a graph, and an equation. Also see the graph at the right. The process starts with 0.10 kg of ice at - 40°C and ends with 0.10 kg of steam at + 150°C. Note: c stands for specific heat. AU (kJ) 319.1 309.3 83.7 41.9 8.4 +T(°C) -40 150 Words Sketch the initial and final Mathematical parts of the process. Q3mcAT 8.4 kJ = 0.1 kg*c*40 degrees C A. Ice starts at -40°C and warms to its melting temperature at O°C. 20 Q = mLmelting B. Energy provided to ice causes the solid ice to convert slowly to liquid at 0 °c. C. After the ice has melted completely, more energy causes the liquid water to warm from 0°C to 100°C, its boiling temperature. BUC = mC Lighid D. Now, energy provided causes the water to convert slowly from the liquid to the gaseous state while still at 100°c. AUE mCgas (150E Tool) E. Additional energy causes the gas to warm from 100°C to 150°C 2001 Are the equations for parts B and D consistent with the corresponding parts of the graph? Why does temperature not show up in these equations?
1. Carefully examine the following table, which represents a process in multiple ways-words, sketches, a graph, and an equation. Also see the graph at the right. The process starts with 0.10 kg of ice at - 40°C and ends with 0.10 kg of steam at + 150°C. Note: c stands for specific heat. AU (kJ) 319.1 309.3 83.7 41.9 8.4 +T(°C) -40 150 Words Sketch the initial and final Mathematical parts of the process. Q3mcAT 8.4 kJ = 0.1 kg*c*40 degrees C A. Ice starts at -40°C and warms to its melting temperature at O°C. 20 Q = mLmelting B. Energy provided to ice causes the solid ice to convert slowly to liquid at 0 °c. C. After the ice has melted completely, more energy causes the liquid water to warm from 0°C to 100°C, its boiling temperature. BUC = mC Lighid D. Now, energy provided causes the water to convert slowly from the liquid to the gaseous state while still at 100°c. AUE mCgas (150E Tool) E. Additional energy causes the gas to warm from 100°C to 150°C 2001 Are the equations for parts B and D consistent with the corresponding parts of the graph? Why does temperature not show up in these equations?
Chapter5: Temperature And Heat
Section: Chapter Questions
Problem 1MIO: In Section 5.2, we discussed the phenomenon of thermal expansion. Concept Map 5.4 was to have been...
Related questions
Question
help with the Q and with sketches (drowing) thank you
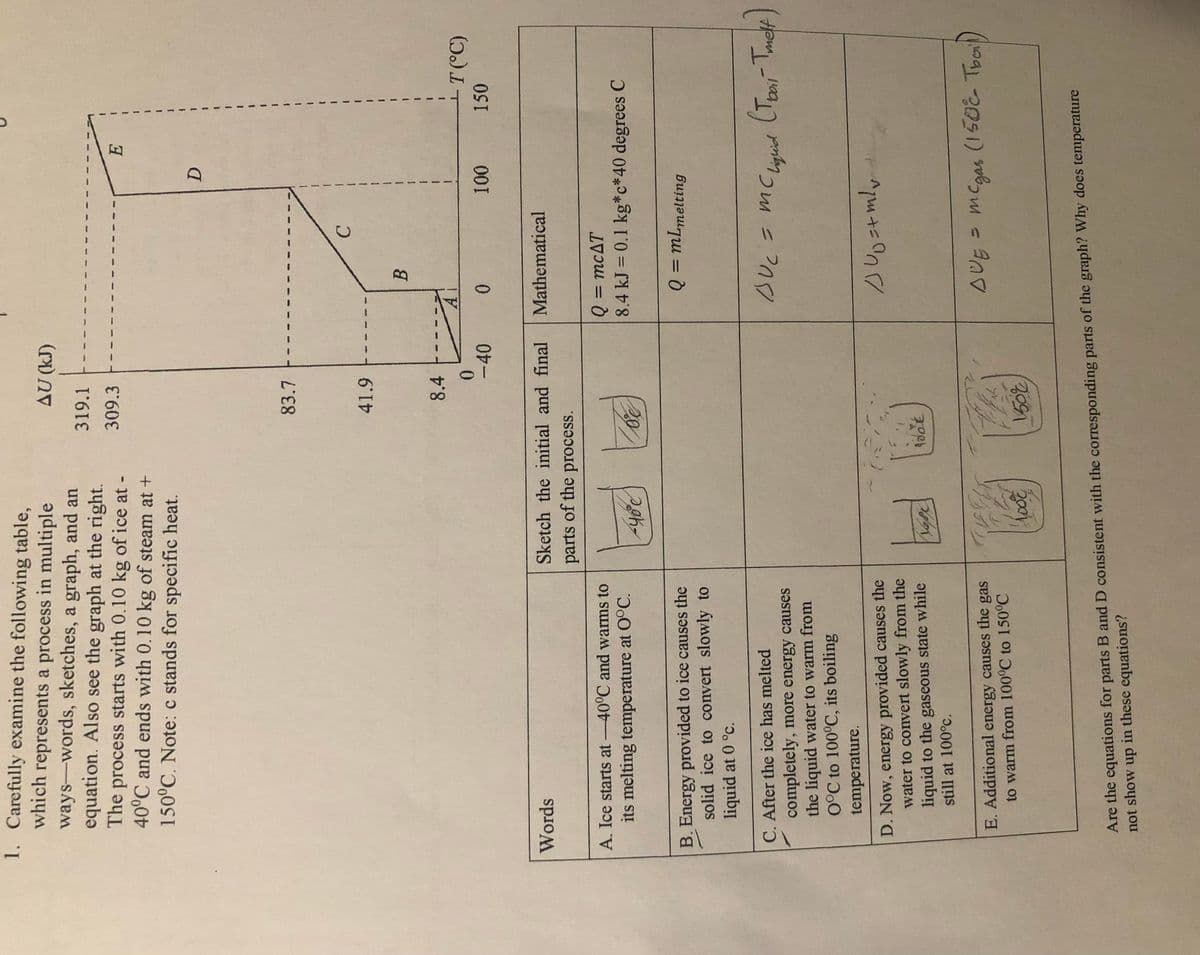
Transcribed Image Text:1. Carefully examine the following table,
which represents a process in multiple
ways-words, sketches, a graph, and an
equation. Also see the graph at the right.
The process starts with 0.10 kg of ice at -
40°C and ends with 0.10 kg of steam at +
150°C. Note: c stands for specific heat.
(r1)
319.1
309.3
83.7
41.9
C.
一
B.
8.4
T(°C)
150
-40
0.
Words
Sketch the initial and final
Mathematical
parts of the process.
Q = mcAT
8.4 kJ = 0.1 kg*c*40 degrees C
A. Ice starts at -40°C and warms to
%3D
its melting temperature at O°C.
Q = mLmelting
B. Energy provided to ice causes the
solid ice to convert slowly to
liquid at 0 °c.
%3D
C. After the ice has melted
completely, more energy causes
the liquid water to warm from
0°C to 100°C, its boiling
BUc= mC
Liguid
temperature.
D. Now, energy provided causes the
water to convert slowly from the
liquid to the gaseous state while
still at 100°c.
sUE MCgas (150E Tooil)
E. Additional energy causes the gas
to warm from 100°C to 150°C
Are the equations for parts B and D consistent with the corresponding parts of the graph? Why does temperature
not show up in these equations?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you


College Physics
Physics
ISBN:
9781938168000
Author:
Paul Peter Urone, Roger Hinrichs
Publisher:
OpenStax College

College Physics
Physics
ISBN:
9781285737027
Author:
Raymond A. Serway, Chris Vuille
Publisher:
Cengage Learning


College Physics
Physics
ISBN:
9781938168000
Author:
Paul Peter Urone, Roger Hinrichs
Publisher:
OpenStax College

College Physics
Physics
ISBN:
9781285737027
Author:
Raymond A. Serway, Chris Vuille
Publisher:
Cengage Learning

An Introduction to Physical Science
Physics
ISBN:
9781305079137
Author:
James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar Torres
Publisher:
Cengage Learning