1. Cr20aq) + CI Cr3+ (aq) + Cl2c9) in acidic solution (aq) 2. Cus) + NO3 (aq) Cu aa) + NO2) + NO29) in acidic solution 3. CN(ag) + Мп04 (аg) CNO (aq) + Mn02(s) in basic solution
Q: In practice the standard solutions are made by pipetting a known amount of a stock solution with a…
A: Given: Concentration of stock solution = 900 µM And the solution's data given is,
Q: wing mixtures has the highest buffering cap 2-10.90 M PO43- 2-10.10 M PO43- e the same buffer…
A: Buffer solution is the mixture of weak Acid/base with its conjugate base/acid. Buffer which have…
Q: Solid CO2 (0.42 mol) is reacted with CH3MGB (0.29 mol) in ether solvent. The product of the reaction…
A: pH is used to determine the concentration of hydronium ion.
Q: you think? Write in “BULLET POINTS”. An alkaline solution was prepared with LiOH in such a way that…
A: Here are some limitations of modern periodic table: 1. There is no proper place for hydrogen in the…
Q: The molar absorptivities of the indicator weak acid HIn (Ka = 1.42 x 10-5) and its conjugate base…
A: The absorbance of the mixture of two species can be written as: A=εHInbCHIn+εIn-bCIn-
Q: A student mixes 40.mL40.mL of 0.10MHBr(aq)0.10MHBr(aq) with 60.mL60.mL of 0.10MKOH(aq)0.10MKOH(aq)…
A:
Q: a) Cr.0,2- + Fe* → Fe3* + Cr3+ (in acidic solution) b) Mn* + BiO3 → MnOq¯ + Bi³* (in acidic…
A: a) Cr2O72-+Fe2+→Fe3++Cr3+ The oxidation state of Cr is +6. b) Mn2++BiO3-→MnO4-+Bi3+ The oxidation…
Q: A 300.00 mL solution of 0.00165 M A2B5 is added to a 230.00 mL solution of 0.00380 M C2D3. What is…
A: The volume of 0.00165 M A2B5 solution is 300.00 mL. The molarity of 230.00 mL solution of C2D3 is…
Q: Your research advisor asks you to prepare a buffer solution at pH = 12.5 with capacity of about…
A: To make a buffer of desired pH/pOH with best buffer capacity, that acid /base should be used which…
Q: Arthur Wen, a CHEM 18.1 student, was tasked to make a 250mL solution of NaCH3COO with pH = 9.022 for…
A: Solution : pH is a quantitative measure that is used for the determination of hydrogen ion…
Q: lem Set #6 (Ch 16) eise 16.53 - Enhanced - with Feedback SED THIS? Watch KCV 16.6, IWE 16.3; Read…
A: Since you have asked a question with multiple sub-parts, we will solve first three sub-parts for…
Q: e 1. Volumes of NaOH solution needed to titrate 25.00mL of HCI Concentration Initial of NaOH(M)…
A: Given: Concentration of NaOH = 0.0963 M Volume of HCl solution used = 25.00 mL. And HCl is titrated…
Q: The standardization of iodine solution was conducted using 10.0 mL of a standard 1.00 mg/mL ascorbic…
A: Molarity of ascorbic acid can be calculated using its given strength in mg/ml by dividing it with…
Q: Question: calculate the pH of 50.0 mL H20 + 1.00 mL 0.6 M NaOH (below is the data to answer the…
A: Formula
Q: A 25.0025.00 mL solution of 0.033000.03300 M Na2CO3Na2CO3 is titrated with 0.026300.02630 M…
A: Using molarity equation as shown below- M1: molarity of Sodium Carbonate. V1: volume of Sodium…
Q: he Affect of Metal Degradation Based on Acid Type Four acids are put into varying test tubes and…
A: To determine which acid provides the highest reaction rate. Test Tube-1 (H2SO4 3M) time: 3.68seconds…
Q: Calc. the alkaline strength of a sample of impure K2CO3 in terms of percent K2O from the ff. data:…
A: Given : Wt. of sample = 1.000 g HCl used = 55.90 ml NaOH used = 0.42 ml…
Q: Please give
A: Given,Molarity of NaOH = 0.248M.Volume of NaOH = 18.71mL = 0.01871L.Volume of H2SO4 = 10.00mL =…
Q: 4. The molar absorptivities of the indicator weak acid HIn (K_{a} = 1.42 * 10 ^ - 5) and its…
A:
Q: Titration of unknown acid with NaOH. Determine the pKa of unknown acid: mass H2A=0.5 g H2O=50 mL…
A: Titration of unknown acid with NaOH. Mass of H2A=0.5 g H2O=50 mL Volume at equivalence point: 23.7…
Q: s. Given the following selectivity data (kij) for this ISE, à Chat activity = concentration). erant…
A: Given: Econst=0.197 V0.5M Ca+,0.2MCa2+
Q: 1. Determine the Ecell values for the following: a. TI (s) | TI* (aq) || Sn2+ (aq) | Sn(s) b. Zn(s)|…
A: Ecello is Cell potential value of cell reaction under standard pressure and temperature. It is…
Q: Step 2: Phosphate buffer Monobasic (mM) Dibasic (mM) pH 50 5.37 40 10 6.08 30 20 6.48 20 30 6.83 10…
A: Solution : Monobasic acids contain one replaceable hydrogen atom per acid molecule while…
Q: Table 3-1. Absorbance Readings of Beet Solutions after Exposure to Various pH Buffer Solutions pH of…
A: A question based on Beer-Lambert law, which is to be accomplished.
Q: Solid CO, (0.38 mol) is reacted with CH3MGB (0.21 mol) in ether solvent. The product of the reaction…
A: The reaction is:
Q: a. NiO2 + 2 H2O + Fe→Ni (OH)2 + Fe(OH)2 in basic solution b. CO2 + 2 NH2OH→CO + N2 + 3 H20 in basic…
A: NiO2 + 2H2O →Ni(OH)2 + 2OH- Fe + 2OH- → Fe (OH) 2 On combining both the reactions, OH- will be…
Q: Complete the given data sheet in determination of total alkaline strength in soda Show your…
A: Alkaline strength is the the strength of solution is the ability of a solution to neutralize an…
Q: RT E = E°. In Q nF Zn(s) + Cu²+(aq) – Zn2+(aq) + Cu(s) E° = +1.10 V Show Annotation After Cu2+ ion…
A: Nernst equation: The Nernst equation relates to the reduction potential of an electrochemical…
Q: Fe (s) | FESO4(0.30 M) || KIO3(0.50 M), 12(s), pH 4.0 | Pt(s) Given: Fe2+ +2 e Fe (s) EO - 0.44 %3D…
A: Firstly half cell reactions are set. Reaction with highest value of reduction potential will undergo…
Q: meyer flasSK and oceeded to make 250-mL of a 0.18 M NaOH(ag) solution in another 250-mL lenmeyer…
A:
Q: otentiometry Topic Briefly discuss the following: 1.different types of membrane indicator…
A: 1. Solution - According to the question - membrane electrodes that produce an electrical potential…
Q: What is the Eh of a solution with 0.05 M Fe2+ and 0.0000002 M Fe3+ ? What is the Eh of a solution…
A: The expression of Nernst equation is shown below: Ecell = E0cell - 0.0591n log Q Where; Ecell= cell…
Q: C: Determination of Ka of a Weak Acid: 25 mL of 0.01 M acetic acid solution is mixed with 25 mL of…
A: We would use Henderson Hasselbalch equation to calculate pKa and using pKa we can calculate Ka.
Q: MasteringChemistry…
A: Hi there, as there are multiple questions posted. We are answering the first question(question…
Q: Fe2+ (aq) + Cr207 2- (aq) Fe3+ (aq) + Cr 3+ (aq) (acidic solution) CN (aq) Fe3+ (s) Fe2* (aq) CNO…
A: to balance the reaction using oxidation number. 1st one in acidic medium 2nd in basic medium
Q: First one is my data and second is what i need help with, please include calculations. a. name of…
A: Given, Concentration of stock solution ,M1=200×10-6M
Q: . NaI + Cl H2O + n hexane ------- _____________ bownish solution with dark violet layer on top…
A: It is a single displacement reaction. When NaI reacts with Chlorine, chlorine displaces iodine and…
Q: Ca2(aq) + Na2C2O4(aq) -> CaC2O4(s) + 2Na(aq) Suppose that a solution is known to contain…
A: From the given reaction: It is clear that the reaction takes place in equimolar ratio. So that, 1…
Q: 2,5 ml volume has taken from an “hypothetic” solution which includes (3+) Sb and (3+) Fe and at the…
A: Fe3+ ion is stable to KMnO4 solution so, only Sb3+ ion undergoes oxidation to Sb5+ ion. The chemical…
Q: Commercial phosphate buffers are sold to maintain fish tanks at a pH of 7. Use a table of acid…
A: Acid dissociation equilibrium constants for H3PO4 are : Ka1 = 7.2 x 10-3 Ka2 = 6.8 x 10-8 Ka3 =…
Q: The value of Ksp for Mg3(AsO4)2 is 2.1 x 10-20. TheAsO43 - ion is derived from the weak acid H3AsO4…
A: (a) Mola solubility of Mg3(AsO4)2 in water; Ksp for Mg3(AsO4)2 =2.1 x 1020. Mg3(AsO4)2 ⇋ 3Mg2+…
Q: How would you prepare 1.00 L of a buffer solution at pH 6.00 from 0.500 M Na3AsO4 and 0.400 M HCl?
A:
Q: The voltage measured by the pH meter is linearly proportional to the "H+ concentration" of the…
A: Concept is based on pH.
Q: Pls help ASAP.
A: to calculate : concentration of free Ag+ in the solution
Q: Which of the following shows the correct dissolution reaction for CaCO3 A.…
A: CaCO3 is an ionic compound. Hence it will dissolve and produce ions
Q: Under standard conditions, the number of moles of M* oxidized when one mole of X is converted to Y…
A: The electrode potential of a cell is calculated by using the Nernst equation, which is:…
Q: EDTA: 0.2 M solution KH2PO4: 0.4 solution Sodium Nitrate MW: 84.9947 g/mol A 150 mL buffer solution…
A: Volume of buffer solution = 150 mL…
Q: Fe*(aq) + KSCN(s) →FESCN²+(aq) + K*(aq) To determine the moles of Fe3*(aq) in a 100.0 mL sample of…
A: From the curve, at 0.20 absorbance, the concentration of FeSCN2+ is 4×10-5 M that mean 1000 mL of…
Q: Calculate AG for the following reaction. 2 Cr(s) +3 Cl2(aq) → 2 Cr** (aq) + 6 CI (aq) Hall-Reaction…
A: The expression for the standard Gibbs free energy change can be given as follows:…
Q: Molarity of titrant (NaOH): 0.4550 M HC2H3O2 (aq) + NaOH (aq) → NaC2H3O2 (aq) + H2O (l) Trial #…
A: Given: Molarity of titrant (NaOH): 0.4550 M Trial # First Second Third Fourth…
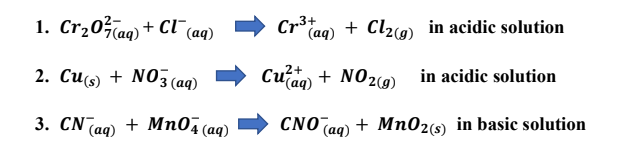
complete and balance this equation by the method of half reaction
Step by step
Solved in 2 steps with 2 images

- A 370.00 mL solution of 0.00185 M A3B4 is added to a 390.00 mL solution of 0.00100 M C3D4. What is pQsp for A3D4?A pH probe/meter uses the following equations: Ecell = L + 0.0592 log a1 = L - 0.0592 pH Where L = L1 + EAg/AgCI + Easy= constants L1 = - 0.0592 log a2 a1 = activity of analyte solution a2 = activity of internal solution Questions: How will measured pH value be affected vs “real” pH if the temperature of the sample is 30C when pH was measured? How will measured pH value be affected vs “real” pH if HCl in pH electrode, became 0.15M instead of 0.1M? How pH value will be affected vs “real” pH if the glass of the pH electrode is not fully hydrated? Please answer all questions and provide a brief explanationC8This is for my reviewer please help me with the step by step solution and answer, thank you
- Sodium carbonate is a reagent that may be used to standardize acids in the same way that you have used KHP in this experiment. In such a standardization it was found that a 0.498-g sample of sodium carbonate required 24.8 mLmL mL of a sulfuric acid solution to reach the end point for the reaction. Na2CO3(aq)+H2SO4(aq)→H2O(l)+CO2(g)+Na2SO4(aq)Na2CO3(aq)+H2SO4(aq)→H2O(l)+CO2(g)+Na2SO4(aq) What is the molarity of the H2SO4H2SO4?You collected the following data from a titration experiment using a 0.129M standardized NaOH solution to titrate a 26.55 mL solution with an unknown Molarity concentration (M) of sulfuric acid (H2SO4). Initial Burette Reading (mL) Final Burette Reading (mL) Vol Delivered (mL) Trial 1 0.44 19.69 ?? Trial 2 0.18 17.2 Trial 3 0.50 19.94 For just Trial 1, determine the amount of NaOH delivered for the titration with appropriate significant digits. Do not include units.3I- + OCl- + 2H+ ----> I3- + Cl- + H2: A 25.00 ml sample of liquid bleach was diluted to 1000 ml in a graduated flask. A 25 ml portion of the diluted sample was pipetted into an Erlenmeyer flask and treated with excess KI to oxidize OCl- to Cl- and I3- was produced at the end of the reaction. The released I3 was titrated with 0.09892 M Na2S2O3 and 8.96 ml was spent to reach the turning point besides the starch indicator. What is the percent by weight/volume of NaOCl in the bleach sample? (NaOCl:74.44 g/mol)
- 0.1724g of a mineral containing MnO₂ was dissolved and then treated with excess iodide according to the following unbalanced reaction: MnO2(s) + H+ → Mn²+ + I2 + H₂O The I₂ released was titrated with a solution of Na₂S2O3 0.07320 mol/L, requiring 14.65 mL to reach the point end of degree a) Determine the percentage of MnO₂ in the ore. b) When iodine solutions are used as titrants these are prepared by dissolving 1₂ in concentrated KI. Explain the reason for the need for KI in the preparation of these solutions?The solubility of borax, which is made up of sodium tetraborate (Na2B4O5(OH)4 8H2O), was analyzed. The dissolution of borax is: Na2B4O5(OH)4 • 8H2O(s) ⇌ 2 Na+(aq) + B4O5(OH)42–(aq) + 8 H2O(l) A 50 mL saturated solution was prepared. After filtration of solution, 5 mL aliquot was transferred to a flask and titrated using 0.432 M HCl. The endpoint was found to be 4.73 mL of the titrant. Tetraborate anion (B4O5(OH)42-) is a weak base which reacts with HCl like the following reaction: B4O5(OH)42–(aq) + 2 H+(aq) + 3 H2O(l) ⇌ 4 H3BO3(aq) What is Ksp expression for the dissolution? What is the tetraborate ions concentration in the filtrate? What is the molar solubility and Ksp of borax if the titration was done at room temperature (298 K)?The SO2 present in air is mainly responsible for the phenomenon of acid rain. The concentration of SO2 can be determined by titrating against a standard permanganate solution as follows: 5SO2 + 2MnO4- + 2H2O ---> 5SO42- + 2Ms2 + 4H+ Calculate the number of grams of SO2 in a sample of air if 4.90mL of 0.00700M KMnO4 solution are required for the titration. Be sure your answer has the correct number of significant digits.
- For full contact with the acid, nitrile gloves with at least 0.4 mm thickness are required. The thickness of the nitrile gloves in our labs is 6 mil (1 mil = 0.001 in). Will our gloves protect you from full contact with concentrated HCl? Show a calculation to support your conclusions.Show complete solution. Round-off your answers to four decimal places The amount of copper in a wire was determined by a redox titration using KMnO4 as the titrant. A 0.4185-g sample was dissolved in acid and the liberated Cu2+ quantitatively reduced to Cu+, using a reductor column. Titrating with 0.0250 M KMnO4 requires 41.27 mL to reach the endpoint. Determine the % w/w CuO in the sample of meteorite. Answer: % CuO = ______Make a scheme with this procedure. Write the result. Procedure 1. Place 20 drops of each of the following aqueous solutions to separate centrifuge tubes: 0.1M Cr (NO3)3, 0.1M Al (NO3)3, 0.1M Co (NO3)2, 0.1M Zn (NO3)2, 0.1M Mn (OH)2, 0.1M Ni (NO3)2, 0.1 M Fe (NO3)3. Make each solution basic by adding few drops of 6M NH4OH. Confirm using a litmus paper. 2. Add 5 drops of freshly prepared 6M (NH4)2S to each centrifuge tube. Place the samples in the centrifuge machine for 3 mins. After centrifuge record results. Decant the supernatant liquid of all the samples. 3. Add one drop of NH4OH in each centrifuge tubes. Add 20 drops of distilled water in each centrifuge tubes. Then add a few drops of 6M HCl in each solution. Place the samples in the water bath for 10 mins. After water bath, centrifuge the samples for 3 mins. 4. After centrifuge, add a few drops of 6M NH4Cl in each sample. Decant the supernatant liquid in each sample. 5. To the centrifuge containing Al+3 and Cr+3, slowly add…



