1. Draw the predominant forms of glutamic acid with increasing pH. The pKas for the ionizable groups are amino terminus = 9.67, carboxy terminus = 2.19, and side chain carboxylic acid = 4.25. C D A Charge: Charge: Charge Charge Lowest pH Highest pH
1. Draw the predominant forms of glutamic acid with increasing pH. The pKas for the ionizable groups are amino terminus = 9.67, carboxy terminus = 2.19, and side chain carboxylic acid = 4.25. C D A Charge: Charge: Charge Charge Lowest pH Highest pH
Anatomy & Physiology
1st Edition
ISBN:9781938168130
Author:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Chapter2: The Chemical Level Of Organization
Section: Chapter Questions
Problem 24RQ: Ty is three years old and as a result of a stomach bug has been vomiting for about 24 hours. His...
Related questions
Question
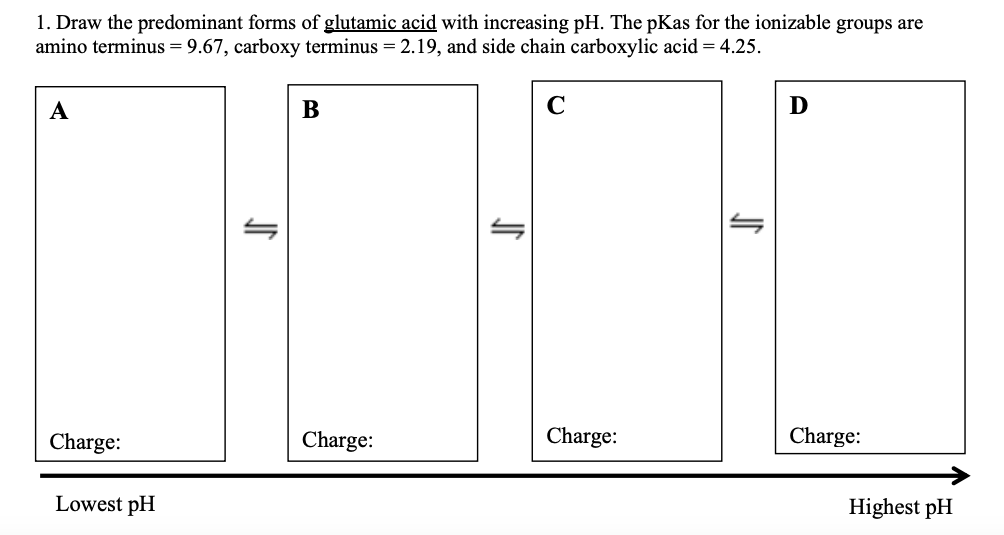
Transcribed Image Text:1. Draw the predominant forms of glutamic acid with increasing pH. The pKas for the ionizable groups are
amino terminus = 9.67, carboxy terminus = 2.19, and side chain carboxylic acid = 4.25.
C
D
A
Charge:
Charge:
Charge
Charge
Lowest pH
Highest pH
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

Anatomy & Physiology
Biology
ISBN:
9781938168130
Author:
Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:
OpenStax College

Anatomy & Physiology
Biology
ISBN:
9781938168130
Author:
Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:
OpenStax College