Q: I1. IL Balance the following equatians. Shw your solution. 1. Al+ O2 AlzO KONS COHO 2. CaHa+ O2 3.…
A: in Balanced chemical equation number of atoms of each species are equal on both reactent and…
Q: What stereoisomers are obtained from the following reactions?
A: (a) 1. In this reaction NaNH3 is base. 2. Na is reducing agent. 3. It reduces alkynes to…
Q: In order to neutralize a sample of Sodium Hydroxide (NaOH) weighing 10.0 grams, how many moles of…
A:
Q: 4 HCI (g) O2 (g) 2 H20 (1) + 2 Cl2 (g) AH= +27 kcal +. -->
A: Le Chatlier's principle : If an equilibrium is disturbed by changing conditions, the position of…
Q: 1. ВНз, THF 2. NaOH, H202 OH HO OH OH OH HO II II IV V Select one: O II, II O II, IV O II, V O IV V
A:
Q: AG°F nol·K)] kJ/mol) _8 65.1
A:
Q: A 0.640 g sample of a metal, M, reacts completely with sulfuric acid according to M(s) + H,SO,(aq) –…
A: As per equation 1 mol of metal M gives 1 mol of H2 gas First find the moles of H2 gas by using…
Q: The equiVatent weight of Naz Pou is ? 98 54,6 B 32,6
A: Equivalent weight = Molar weight / valency factor For salt valency factor is either total cationic…
Q: Ba(OH)2(aq) + 2 HCI(aq) → BaCl,(aq) + 2 H,O(1)
A: The chemical name of Ba(OH)2 is barium hydroxide. It is a strong base. The chemical name of HCl is…
Q: Complete the equation: HNO3(aq) + KHCO3(aq) (a) H¿O(1) + CO2(8) + KNO3(aq) (b) KNO3(s) + H,CO3(aq)…
A: When two chemical species are allowed to react then, they will form the desired products. The…
Q: + H3O (aq) 3. HC,H7O4(aq) + H2Oa) 4 CH;O4 (aq) 4. CO; (aq) + H,Om S HCO3 (ag) + OH (aq)
A: Bronsted Lowry Theory - This theory states that acids release protons and bases take protons. When…
Q: (а) (b) NaN3 KCN ? H20 ? HOCH,CH,OH (с) (d) HCN 1. LIAIH4 ? HOCH,CH,OH ? 2. Н,о°
A:
Q: 3.37 g of an unknown triprotic acid is reacted with 2.00 M NaOH. It takes 50.4 mL of NaOH to…
A:
Q: (d)net lonic eqnation 18. NaoH CO(NO), (a)balanced equa tion (b)T01al ionic eonantion Cc) net 1onic…
A: Reactions of these compounds are known as double displacement reaction. In these reactions all the…
Q: D Click on the number to put it in your LabPad Calculator H₂SO, volume : 25 mL H₂SO4 diluted to :…
A:
Q: Pb(C2H3O2)2(aq) + H2O(l)+CO2(g)---PbCO3(s)+HC2H3O2(aq) suppose a aqueous solution containing 1.40…
A: Mass of lead (II) acetate = 1.40 g Mass of carbon dioxide = 5.80 g Molar mass of lead (II) acetate…
Q: Calcuate the volume
A:
Q: 6. (aq) (aq) BaCl, (aq) + H,O (1) +
A: Redox reaction is a simultaneous reduction of one compound and oxidation of another compound.…
Q: Zn(s) + 2HCI(aq) à ZnCl,(aq) + H,(g) 1) How many grams of Zinc are necessary to create 100 grams of…
A:
Q: How do you balance AlF3 + Na2SO4 ---> NaF + Al2 (SO4)3
A: Balanced chemical is that equation in which number of atoms on right hand side and left hand side…
Q: QUESTION 15 In which of the following equations is the outcome wrong? O a. O b. Oc CI d. Br. N₂+ CH…
A:
Q: Hello, I need help to balancep this equation. NiCl2(aq)+K2CO3(aq)= blank + blank Thank you.
A:
Q: camputely Nan cOs+ 2 HBr -> HaCOz How many mL of 0.677M HBr is needed to y retralize o.898g of Nag…
A:
Q: Q6\ How to prepare(10 MI/of 0.2 N of NaoH, m. wt NAOH =40
A:
Q: What volume of 3.00 M NaOH contains 1.75 mol NaOH? __?__ L
A: Molarity is defined as moles per litre.
Q: HCI Mg(OH)2 (aq) (aq) HCl (ag)-Mg(OH)2 (aq) MgCla (aq) + H:O (1) HCI (aq) Mg(OH): (aq) MgCI (aq) +…
A: In a chemical reaction the reactants get converted into the products. The number of moles of the…
Q: 3. Which set of reactants produces a gaseous product? 1. 6 M HNO3 (aq) + Cu (s) I only b. Il only…
A: The chemical reaction is the representation of reactants and products. The reactants and products…
Q: 2Al2O3 yields 4Al +3O2 delta H = 3351.5 kJ what is the delta H for the formation of 10.2 g of Al2…
A: Enthalpy of reaction is equal to the difference in enthalpy of product and reactants.…
Q: What is net ionic for CuSo4+NH4Cl ? Does CUCl2 Considered Insoluable ?
A: The reactants given are 1) CuSO4 (aq) 2) NH4Cl (aq) Since the 2 salts will react and undergo…
Q: 4. DECOMPOSITION INTO ELEMENTAL FORMS: NaN3(6) → ?? 5. POSSIBLE SINGLE DISPLACEMENT: Zn(s) +…
A:
Q: 1. O3 14. 2. (work-up) OH C. A. B. 15. A. В. C. O, NaHCO, NaHco,
A: -> ozonolysis of alkene produce carbonyl compounds. ->In presence of peroxy acid epoxidation…
Q: (c) Ba(CIO3)2 → BaCl2 + O2 Reaction type: (d) CrCl3+ AGNO3 → Cr(NO3)3+ AgCI Reaction type: (e) H2O2…
A: Balancing a chemical reaction aims at making the number of atoms equal on each side of the reaction.
Q: PbSO4 O Pb(NO3)2 (ag)+ Na2SO4 (ag)→PBSO4(s) + 2NaNO3(ag) O Pb(NO3)2(aq) + BaSO4 (s)→PBSO(s) +…
A: The precipitate of compounds form when two aqueous solution of one compound is mixed with aqueous…
Q: Br2 NaOH / H2O (ехcess) 1) LDA O LDA 2) Br
A:
Q: Q / How many grams is required to prepare BaS04 with a concentration of 0.25 M and a volume of 500…
A: Given Molarity = 0.25 M Volume = 500 mL 1000 mL = 1 Liter 500 mL = 5001000Liter…
Q: & or ob OEt oor DEt Cl₂ H₂O* NaOH, H₂O, A 1. NaOEt, EtOH 2. H₂O* 1. LDA, THF, -78 °C 2. 1₂ NaOH, H₂O…
A:
Q: give thumbs up if all are answered correctly. 1. Balance the following chemical equations.…
A:
Q: How many grams of water are produced in this reaction? NH4NO3(s) —> N2O(g) + 2H2O (I)
A: To calculate the gram of water produce by NH4NO3 , find the molar mass of both NH4NO3 and H2O . Then…
Q: N2 = 19 mg/L. s concentrations of N2 in water
A: Mass concentration of N2 : NOTE : 1mg = 10^-3 g mass concentration of N2 = 19 • 10^-3 g / L Mass…
Q: What type of reaction is represent by the equation: C,H12 + 902 → 6CO2 + 6H20
A: The reaction shown is burning of alkane in air to produce carbon dioxide and water .
Q: Manganese(IV) oxide reacts with aluminum to form elemental manganese and aluminum oxide. What mass…
A: Given Reaction 3MnO2 + 4 Al → 3 Mn + 2 Al2O3 Mass of MnO2 = 50 gram Mass of Al = ?…
Q: 4) + ? ? H20 HO,
A: Alkyl magnesium halide ( R-Mg-X ) is called Grignard reagent. Ketones reacts with Grignard reagent…
Q: C6H6 + H2 → C6H14 How many hydrogen atoms are on the product side of the equation? А. 1 В. 4 С. 6 D.…
A: Given: C6H6 + ____H2 ---------> C6H14
Q: A 102.7-g sample of nitric acid solution that is 70.0% HNO; (by mass) contains a. 4.53 x 10° mol…
A: Mass/volume percentage- is one of the ways to express concentration of solution. Formula is, Number…
Q: 3. 1. NaOEt, E1OH 2. Hзо* 4. NaCN HCI
A: Detail mechanistic pathway is given below to find out the product
Q: OH о не о то кон сон ? ? ? Br Br Br X
A: As per the bartleby expert guidelines, I am allowed to answer one question with its 3 sub parts…
Q: 3. A 2.00 g sample of BrxOy is converted to 2.936 g of AgBr. Determine x and y.
A: 2.00g of BrxOy gives 2.936g of AgBr % of Br= ( Mol mass Br/ mol mass AgBr).100
Q: 1. (NH4)2CO3 + H₂O H₂CO3 + NH4OH B. How many grams of ammonium hydroxide will be produced from 745.0…
A:
Q: Give the mayor product(s) of the following reaction. Brg (1 mole) ? heat Br Br Br Br Br Br I There…
A: The major product of the reaction has to be determined.
Can I please have help with this?
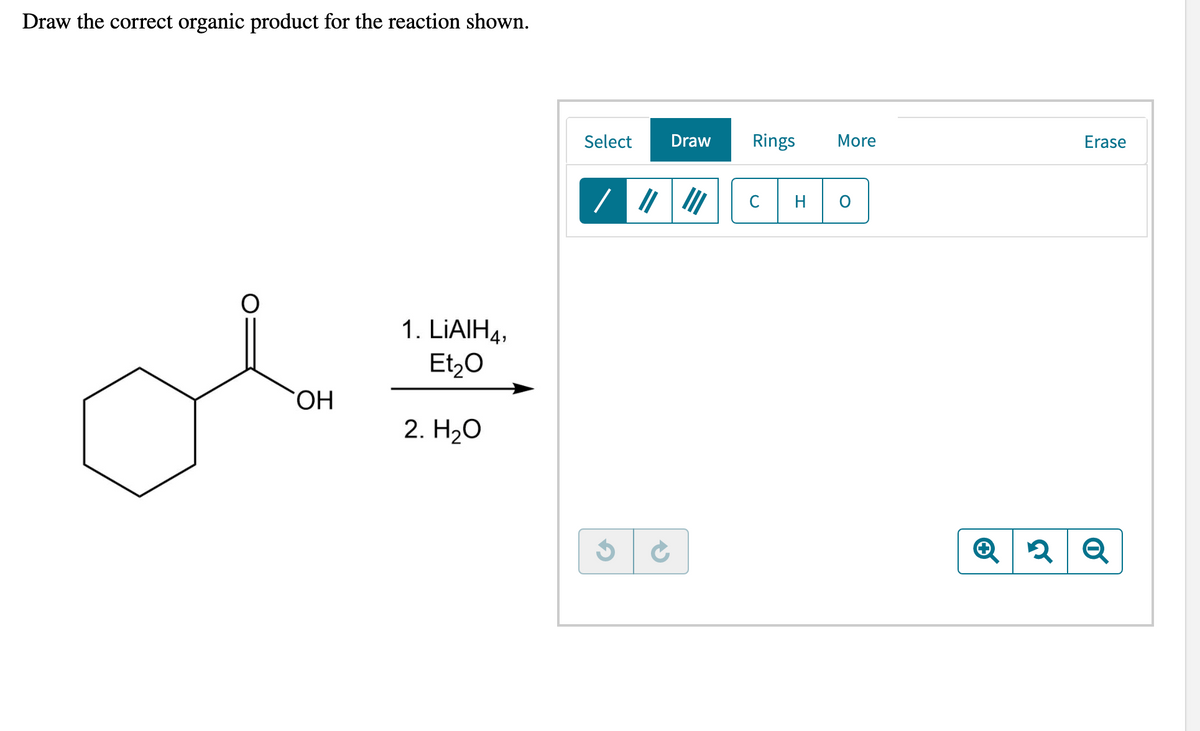
Step by step
Solved in 2 steps with 2 images

- Complete and balance the following methesis reaction in aqueous solution.Answer Entery Instructions• Parentheses are only required around polyatomic ions and only when more than 1 of those ions is present.K3PO4 (no parentheses required).Mg3(PO4)2 (parentheses required).• In each case one product is soluble (aq) and one insoluble (s). Use solubility rules to determine which product appears in each box.Answer123456other Fe(NO3)2 (aq) + Answer123456other (NH4)3PO4 (aq) ➝ Answer123456other Answer (aq) + Answer123456other Answer (s)Which compound is the main product of the reaction in Figure 22? * Compound A Compound B Compound C Compound D None of the above.For the following clauses reactions, draw the correct organic products
- Which compound is the main product of the reaction in Figure 22For 10.29, the trans product (major product) and the cis product (third product) are displayed as possible E2 products. However, for 10.30, only the trans product (major product) is shown. How come the cis product is not needed?can you help with quesiton 12
- Give the product to complete the reaction. Indicate NR if there is nor reaction.List the requested biological classification or organic functional group(s) for the reactant(s) first , predict the reaction type, and predict the product(s) classification or functional group(s). Use the information to draw the major product(s)What is product E?

