Chapter24: Amines And Heterocycles
Section24.SE: Something Extra
Problem 40MP
Related questions
Question
100%
Please help me

Transcribed Image Text:SPECTROSCOPY DRY LAB
General Formula to calculate the degree of unsaturation:
#C atoms - (#H atoms/2) + 1
Example: Benzene (C6H6) 6 - (6/2) + 1 = 4
Rules for calculating unsaturation with organic compounds that contain heteroatoms
(halogen, O, S, N)
Rule 1) Replace any halogens with hydrogens
Example: CH3B change to CH4 and then calculate 1- (4/2) + 1 = 0
Rule 2) Ignore any O or S atoms in the compound
Example: Phenol (C6H5OH) calculate for C6H6 6- (6/2) + 1 = 4
Rule 3) Subtract a H for every Nitrogen present
Example: Aniline (C6H5NH2) calculate for C6H6 6- (6/2) + 1 = 4
For the following problems given spectral data and the chemical formula:
1) Determine the degree of unsatuaration using the unsaturation rules above and list it in the
appropriate box:
2) Label important absorption bands directly on the IR spectra
3) Identify and label all signals directly on the 1H NMR and or 13C NMR spectra (make sure to
use Ha, Hb, Ca, Cb, etc...
Remember TMS shows up as a singlet at 0 ppm for both the 1H and the 13C NMR
CDCI3 (solvent) sometimes shows up as a triplet around 77pm in a 1°C NMR
4) Propose a structure for the compound that matches 1-3 above (If your structure has more
than four degrees of unsaturation it's quite likely to have an aromatic ring). Draw the
SKELETAL structure in the proposed structure box.
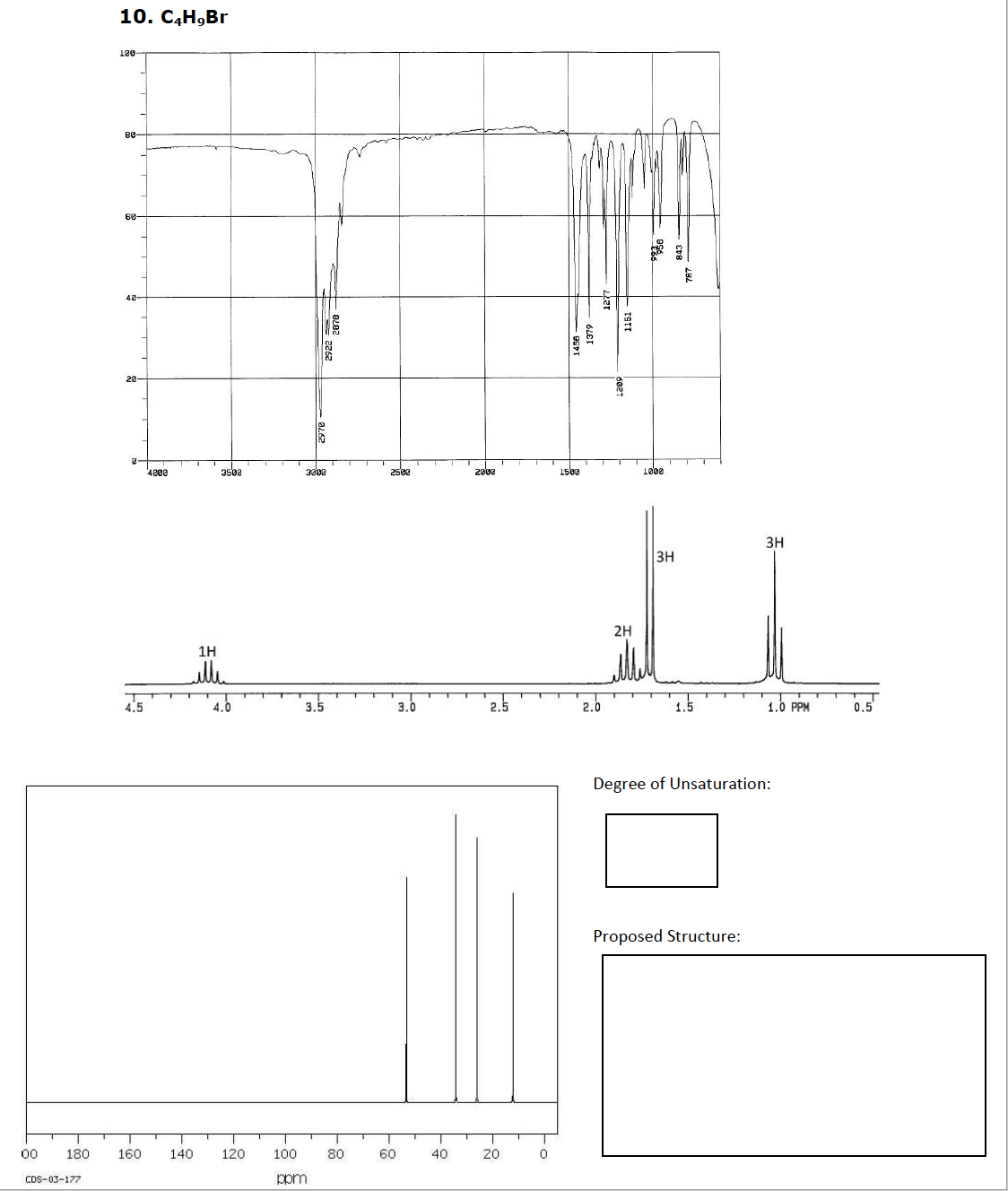
Transcribed Image Text:10. СН,Br
10-
80-
60-
40-
20-
4200
3508
302
2508
2002
1500
1000
3H
3H
2H
1H
elle
4.5
4.0
3.5
3.0
2.5
2.0
1.5
1.0 PPM
0.5
Degree of Unsaturation:
Proposed Structure:
00
180
160
140
120
100
80
60
40
20
CDS-03-177
ppm
ISTT
GLET
- 95T
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning