Q: During the synthesis of the antiasthmatic drug montelukast (Singulair), a silyl ether protecting…
A: Given reaction,
Q: Which of the following can convert bromobenzene to a carboxylic acid? Mg, ether, CO2…
A: This can be done by grignard reagent.l
Q: 6. perform a retrosynthetic analysis by working backwards two steps in the synthesis below.
A: Retrosynthesis is a technique for solving problems in planning the organic synthesis. It is a…
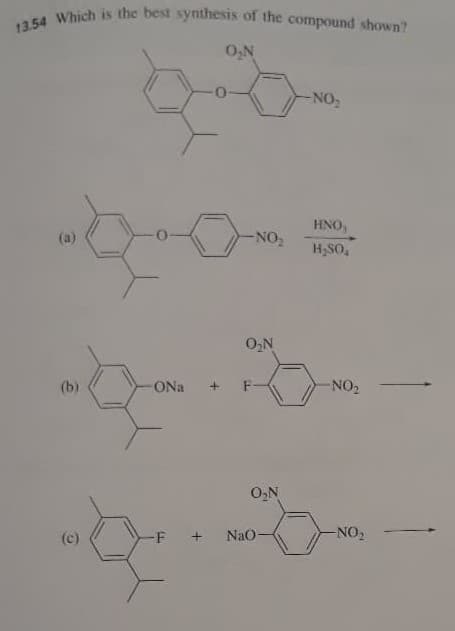
Q: 17) Which of the following reaction(s) is(are) incorrect? a. Ortho Para Meta CH3 CH, CH, NO, CH,…
A: The question is based on the concept of organic reactions. We have to identify which of the given…
Q: Find the products (A and B) for the following reaction sequence: Br 1. t-BUOK, t-BUOH NaOMe, MEOH 2.…
A: Different reagents give different products with the same reactant. These reagents are used so that…
Q: Predict the product of the following reaction. 1. LDA, -78°C H. 2. H. HDJCMD но TH.
A: When butanal treated with LDA (lithium di iso propyl Amide ) then in first step enolate ion is…
Q: Which combination of reagents A-D is suitable for the synthesis of the following compound: Br + Li B…
A:
Q: 11. Which of the following compounds is most basic? IV E. All of these A. I B.I C. II D. IV A В D E
A: Conjugate base of weak acid is more basic and less stable in nature. So more stable negative charge…
Q: Show that you understand the concept of retrosynthetic analysis by working backwards two steps in…
A: We have to synthesize the given compound C from compound A and B using retro synthetic analysis.
Q: 7. Tramadol (Ultram8) is an opioid painkiller used to treat moderate to severe pain. Tramadol may be…
A: Concept: Grignard reagent: Grignard reagents(RMgX) react with the carbonyl group, C=O, in aldehydes…
Q: Two products are formed in the following [1,7] sigmatropic rearrangement, one due to hydrogen…
A: Given reaction with [1, 7] sigmatropic rearrangement.
Q: 1. 2. NaOEt EtOH i) mechanism: ii) most important determining factor(s): iii & iv) detailed,…
A: E2 elimination is favored by a strong base and strong leaving group. Also, E2 elimination is…
Q: 3. For each reaction below identify the electrophile and the nucleophile. Na + CH,CI CH,N3 a. NO,…
A: A Nucleophile Is A Reactant That Provides A Pair Of Electrons To Form A New Covalent Bond. And An…
Q: What is the most likely product distribution for the reaction shown below? SH CH3-1 (excess)…
A: Thiol Being nucleophile Attacks electrophilic Carbon and iodine leaves as Leaving Group.
Q: the product expected for the reaction wn. 1) N₂OEt 2) 4CH₂Br H₂ C EtO₂C CO₂Et EtO₂C. CO₂Et CH₂ H₂…
A: Organic reactions are those in which organic reactant react to form organic products.
Q: 6-The PKa of p-chlorobenzoic acid is 3.98, and that of benzoic acid 4.19. Calculate o-value of…
A:
Q: 2.3 Draw correlation diagrams for conrotatory and disrotatory paths for reactions (a) and (b). Label…
A:
Q: Provide the major product for the following reactions 1. A,B,C,D,E,F 2. A,B,C,D,E,F
A:
Q: What is the percent composition of a mixture of (S)- (+)-2-butanol, [a] = +13.52°, and…
A: The first step to solve this problem is to find the percentage of the excess isomeric compound by…
Q: Predict the product(s) of the following reaction: with HBr at 80°C
A:
Q: Name the major organic product which results when benzenesulfonic acid reacts with Cl2 in AICI3,…
A: Benzene sulphonic acid is meta directing group and its gives chlorination reaction on meta…
Q: Which is not an intermediate or resonance species in the following reaction? A a B b C d Your answer…
A:
Q: Predict the major product for the following reaction: HN Na, CH3OH NH3 HN HN HN HN HN O equal…
A: An organic reaction takes place in the presence of a regent, or a catalyst. The reaction can proceed…
Q: Which of the structures below (A - D) is the final product (Y) of the two reactions shown below?…
A: The given reaction is an example of Wolf-Kishner reaction. In this reaction an aldehyde or ketone is…
Q: CH,CCHCCH,CH, CH3CCHČCH,CH3 + "NH, =t NH, CH,CCHCCH,CH, DK,- 38 Kea Equilibrium position=
A:
Q: What is the most probable product when the compound below reacts with one mole of CH3OH in the…
A: Aldehyde are more reactive than ketone . Nucleophile will attack aldehyde in presence of ketone.…
Q: 5) Identify which of the following reactions are unfavorable and explain why: A) B) C) D) :o: CH3 +
A: In nucleophilic substitution reaction, Nucleophile has to attack to the electrophilic centre . Here…
Q: Practice Problem 13.32 When ethylene glycol is treated with sulfuric acid, 1,4-dioxane is obtained.…
A: The mechanism for reaction between ethylene glycol and sulphuric acid is shown below, First, the…
Q: Identify the major product in the following reaction „CH. !CH.CH,MgBr II. H,O LO, H,SO,(conc.)…
A:
Q: In the following reactions substituent X was varied between electron donating and electron…
A: The effect of the substituents on the rate (n) of a chemical reaction is expressed by the Hammet…
Q: Ignoring stereochemistry, what is the major organic product of the reaction of 2,4-dimethylpentane,…
A: Chlorination reaction are proceeds via radical mechanism, in presence of light.
Q: What compounds will make up the product distribution for the reaction below? .CI Ch hv A O Compounds…
A: Chlorination reaction follows free radical mechanical. More the stability of free radical more will…
Q: 7. Provide the major organic product of the following reaction. X 50% H₂SO4
A: Alkenes react with H2SO4 and give alkene hydrogensulphates. After hydrolysis these give alcohols.
Q: Select the reagents and the appropriate sequence to prepare the product shown below from benzene,…
A:
Q: Draw a curly arrow mechanism for the reaction below and hence predict the structure of K. (c) 1) PC,…
A: Organic transformation:
Q: CHM 2 1 2 5 LABORATORY MANUAL V. Draw the intermediates for the attack of the nitronium ion (NO2")…
A: The delocalization of pi electrons in a conjugated system giving rise to different hybrid structures…
Q: 13. Identify the compounds A to E in each of the following reactions. conc. H2SO4 a) CH3CH,CH,OH A +…
A: conc H2SO4 is a dehydrating agent. It takes away water molecule from any compound making a double…
Q: 1 A What is the expected product A for the following transformation? N-CH₂ H-N HN 19. F1 N N-CH3 CH3…
A:
Q: Give explanation too
A: The sequential double dehydrohalogenation gives the diene.
Q: What is the AHrxn for the reaction 2B 3C given the following: A- 2B AHrn = AH1 A - 3C AHrxn = AH2 A.…
A:
Q: 9) Predict the major organic product in the following reaction. heat or H,02 NH2 (1) (II) (III) (IV)
A: The given reaction is an example of cope elimination reaction.
Q: Propose a mechanism for the following Cope rearrangement. OH 320"C H.
A: Mechanism for the given Cope rearrangement to be proposed.
Q: Ignoring stereochemistry, what is the major organic product of the reaction of 2,4- dimethylpentane,…
A: This is photolytic chlorination reaction of alkane. This reaction follows radical mechanism and…
Q: What is the most probable product when the compound below reacts with one mole of CHOH in the…
A: Carbonyl compound (aldehyde and ketone) reacts with an alcohol to form hemiacetal in the presence of…
Q: Two products, A and B, are obtained from the reaction of 1-bromobutane with NH3. Compound A reacts…
A: IR spectroscopy is mainly used for the functional group determination. We cannot obtain much…
Q: Rank the following compounds in order of increasing pKa: (A) para-chlorobenzoic acid (B)…
A: Strength of acidity means how easily a compound can give hydrogen ions
Q: Tropone is an unusually basic carbonyl (C-0) compound. When it is treated with one equivalent of the…
A: Tropone accepts a proton and acts as a base. The basic nature of the tropone can be understood by…
Q: HO opro CH;CH3 CH,CH, CH,CH-CH, CH, anol 0-CH2CH,CH3 CH, CH,CH,-O-CH,CH3 2. Complete the following…
A:
Q: 1. Draw the products for the following base-catalyzed E2-elimination. What are their relative…
A: The reaction given are,
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Please provide a correct aanswer Answer the following and Identify the reagent (and its concentration) that is used to confirm the presence of each given: a. CO32- b. S2- c. I-In the following three compounds(1,2,3) arrange their relative reactivity towards the reagent CH3Cl / AlCl3. and justify it.Provide a reasonable arrow-pushing mechanism for Reaction 5b, and explain the the stereochemical outcome. 5d below
- Predict the product C of the following reaction below and briefly explain youranswer.[PtCl3(C2H4)]¯ + NH3 → CBenzene is one of the compounds used as octane enhancers in unleaded gasoline. It is manufactured by thecatalytic conversion of acetylene to benzene: 3C2 H2(g) ⇌ C6 H6(g). Which value of Kc would make this reactionmost useful commercially? Kc ≈ 0.01, Kc ≈ 1, or Kc ≈ 10. Explain your answerWhich is more acidity; 4-nitrobutanol or 2-nitrobutanol and how?
- Rank the following compounds in order of increasing pKa: (A) para-chlorobenzoic acid (B) meta-chlorobenzoic acid (C) ortho-chlorobenzoic acid (D) benzoic acid a. D, A, B, C b. D, A, C, B c. D, C, B, A d. A, B, C, D e. C, B, A, DButter yellow is a dye once used to color margarine. It has since been shown to be carcinogenic, and its use in food is no longer permitted. Outline a synthesis of butter yellow from benzene and toluene. N(CH)2 Butter yellowGrignard reagents react with oxirane (ethylene oxide) to form 1° alcohols but can be prepared in tetrahydrofuran solvent. Why is this difference in behavior observed? A. There is a better leaving group in the oxirane molecule. B. Steric hindrance in the case of tetrahydrofuran precludes reaction with the Grignard. C. It is easier to obtain tetrahydrofuran in anhydrous condition. D. Oxirane is a cyclic ether, while tetrahydrofuran is a hydrocarbon. E. The oxirane ring is the more highly strained.
- Tunicates are marine animals that are called "sea squirts" because when they are taken out of water, they tend to contract and expel seawater. Lepadiformine is a cytotoxic agent (toxic to cells) isolated from a marine tunicate. During a recent synthesis of lepadiformine, the investigators observed the formation of an interesting by-product (3) while treating diol 1 with a reagent similar in function to PBr3 (J. Org. Chem. 2012, 77, 3390–3400):Provide the major product for the following reactions 1. A,B,C,D,E,F 2. A,B,C,D,E,F9. Product of the reaction of nitrobenzene with sulfuric acid in the presence of oleum at 60 °C:a) o-nitro benzenesulfonic acidb) m-nitro benzenesulfonic acidc) p-nitro benzenesulfonic acidd) None of the above 10. Due to their ability to move through ducts or pipes, they are generally called fluids:a) Solidsb) Solid mixturesc) Gasesd) Liquids and gases

