15.23 The equilibrium constant for the reaction 2 NOBr (g) 2 NO(g) + Br2(g) 2 is K 1.3 x 10 at 1000 K. (a) At this temperature does the equilibrium favor NO and Br2, or does it favor NOBR? (b) Calculate K for 2 NOBr(g) = 2 NO(g ) + Br2(g). (c) Calculate K, for NOBr(g) NO (g) +Br2(g).
15.23 The equilibrium constant for the reaction 2 NOBr (g) 2 NO(g) + Br2(g) 2 is K 1.3 x 10 at 1000 K. (a) At this temperature does the equilibrium favor NO and Br2, or does it favor NOBR? (b) Calculate K for 2 NOBr(g) = 2 NO(g ) + Br2(g). (c) Calculate K, for NOBr(g) NO (g) +Br2(g).
Chemistry by OpenStax (2015-05-04)
1st Edition
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Chapter13: Fundamental Equilibrium Concepts
Section: Chapter Questions
Problem 60E: At a temperature of 60 C, the vapor pressure of water is 0.196 atm. What is the value of the...
Related questions
Question
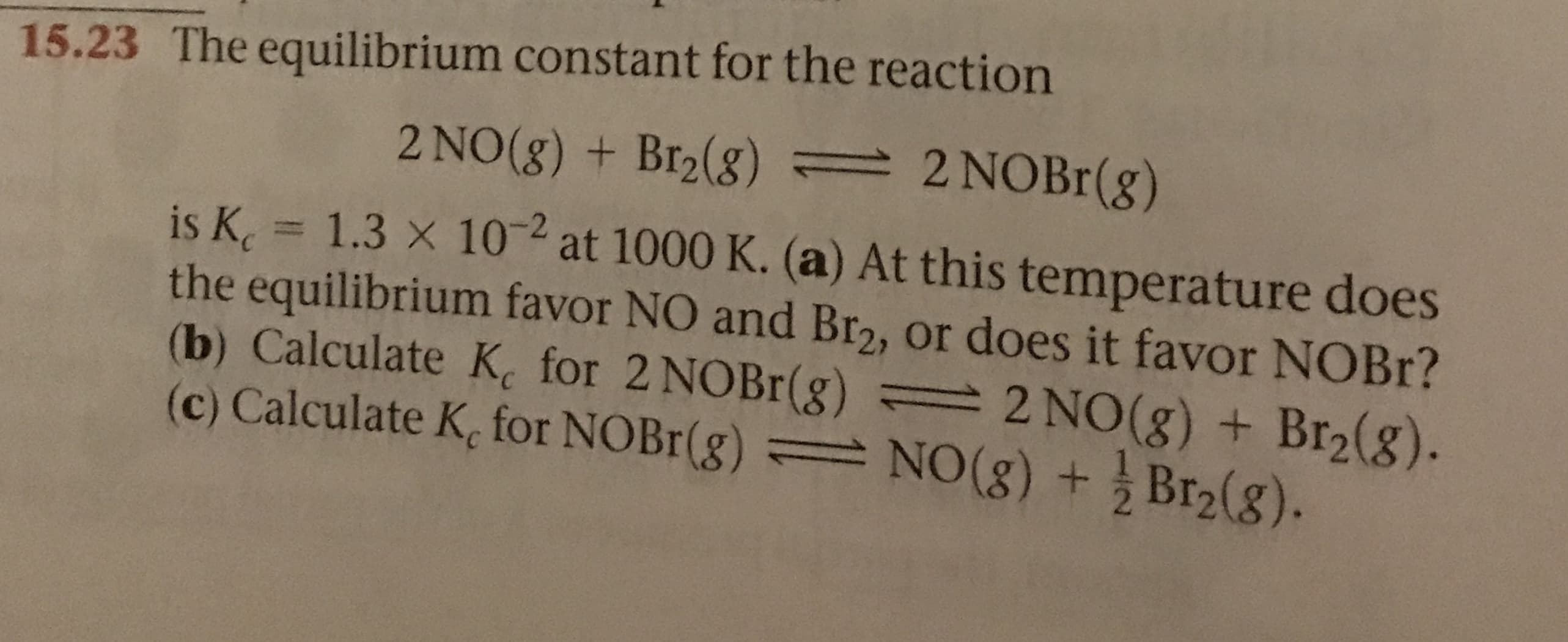
Transcribed Image Text:15.23 The equilibrium constant for the reaction
2 NOBr (g)
2 NO(g) + Br2(g)
2
is K 1.3 x 10 at 1000 K. (a) At this temperature does
the equilibrium favor NO and Br2, or does it favor NOBR?
(b) Calculate K for 2 NOBr(g) = 2 NO(g ) + Br2(g).
(c) Calculate K, for NOBr(g) NO (g) +Br2(g).
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 4 images

Recommended textbooks for you

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781285199030
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning