19. Answer the following questions about the aldohexoses: a. Are D-glucose and D-allose enantiomers? b. Are D-idose and D-mannose C-2 epimers?
Q: An aldohexose would have the functional group A. See first image B. See second image C. -CH3 D.…
A: Functional group is a molecule or group of atoms that have its own characteristic property…
Q: How is each compound related to the simple sugar D-erythrose? Is it an enantiomer, a diastereomer,…
A:
Q: . Determine the number of stereoisomers possible for a ketopentose
A: Stereoisomerism can be defined as the isomerism in which molecules have the same molecular formula…
Q: There are four d-aldopentoses (Table 25.1). If each is reduced with NaBH4, which yield optically…
A: The four aldopentoses are D-Ribose, D-Arabinose, D-Xylose and D-Lyxose. When these aldopentoses are…
Q: 2) Part 1: Draw D-glucose in its proper Fischer Projection (using the template below). Part 2: Draw…
A: Given
Q: 1) a) For the following 4 carbohydrates, identify chiral carbons by putting a* near each one. b) For…
A: Chiral carbon is the optically active center of a molecule, it has four distinct group of atoms…
Q: What is the configuration of each of the asymmetric centers in the Fischer projection ofa.…
A: If carbon is bonded with four different substituents, then that center is called a chiral center or…
Q: 26. This compound is L-glyceraldehyde. Draw a stereochemically correct representation of C-1 and C-2…
A: 1.carbohydrate with aldehydes group called aldose And with keto group called ketose. 27.a.ketose…
Q: A 2-ketohexose is reduced with NABH4 in CH3OH to form a mixture of D-galactitol and D-talitol. What…
A: To find: The structure of 2-ketohexose
Q: Draw the products formed when B-D-galactose is treated with each reagent. a. Ag,0 + CH3I b. NaH +…
A: “Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: Stereoisomer of D-altrose that ONLY different is Carbon #3 a. d-glucose b. d-mannose c.…
A: we need to determine the C-3 isomer of D-altrose
Q: Given the Fisher projection
A:
Q: What reaction is responsible for the conversion of linear d-glucose to cyclic d-glucose? A.…
A: Solution-Hemiacetal reaction - reaction is responsible for the conversion of linear d-glucose to…
Q: Allose is the C3 epimer of glucose. Draw the cyclic hemiacetal form of d-allose, first inthe chair…
A: The structure of glucose is given as shown below. The structure of allose is given as shown below.
Q: The specific rotation of α-d-galactose is 150.7 and that of β-d-galactose is 52.8. When an aqueous…
A: When monochromatic light is passed through a solution of the chiral compound, the light is reflected…
Q: Identify the following as an a or ß form and draw the Haworth structure of the other anomer. CH2OH…
A:
Q: The monosaccharides shown below are… (MARK ALL THAT APPLY) Both α sugars Both D-form sugars…
A: Given that : We have to identify which of the following are true for the above given monosaccharides…
Q: D-Tagatose is epimeric of D-Fructose at C4. What is the structure of α-D-Tagatofuranose?
A: To determine the structure of alpha-D-Tagatofuranose, we would first draw the open chain structure…
Q: Convert attached aldohexose to the indicated anomer using a Haworth projection.
A: Given aldohexose is
Q: What D-aldohexose forms the same osazone as D-glucose?
A:
Q: Answer the following questions about monosaccharide A.a. Draw the α anomer of A in a Haworth…
A: a) The α anomer of A in a Haworth projection has to be given, The given compounds, The structural…
Q: 1. Is sophorose a reducing or a non-reducing sugar? 2. Is monosaccharide Y: an aldose or a ketose?…
A: Since you have posted a question with multi sub-parts, we will solve first three sub-parts for you.…
Q: 5-Deoxyglucose will close to a? O a. thanose O b. furanose O c. maranose O d. none of the other…
A: Glucose is a sugar molecule. in deoxy-glucose, one of the OH is replaced by hydrogen. hence if it is…
Q: 26. The Fischer projection for glucose is shown below. CHO но H- H HOH HO-H H- -HO- ČH2OH a. Is…
A:
Q: Draw
A:
Q: Is the following carbohydrate a D or an L sugar? CHO HO- O- HO- 1. H OH CH2OH H HHH
A: In the sugar, we see the last asymmetric carbon to determine whether the sugar is D or L If the last…
Q: The enantiomer of D-glucose is Oa there is none Ob. D-galactose Oc L-glucose Od. D-mannose Oe…
A:
Q: A 2-ketohexose is reduced with NaBH4 in CH3OH to form a mixture of D-galactitol and Dtalitol. What…
A:
Q: Draw Fischer projections for the product(s) formed by reaction of d-ribose with the following. In…
A: In presence of strong oxidizing agent HNO3, D-galactose is oxidized to form aldaric acid. Because of…
Q: How to Assign R,S designations to stereogenic center in glucose ?
A: Glucose is a monosaccharide, having formula C6H1206. It is an important simple sugar that circulates…
Q: What is the name of the carbohydrate compound C? но CH2OH C A. D-xylose В. D-lyxose C. L-threose D.…
A: We have to tell the name of the carbohydrate compound C.
Q: galactose
A: when D glucose is reduced with sodium borohydride glucitol is formed which have no plane of…
Q: 15. Are mannose and galactose epimers? Allose and altrose? Gulose and talose? Ribose and arabinose?…
A: a) Mannose and galactose: D- Mannose and D- galactose is not epimer because their configuration…
Q: ouTube Maps 4 6 5 CH2OH ОН ОН ОН 3 The Haworth projection of D-mannose is shown. 1 Question 10 of 37…
A:
Q: How many stereoisomers are possible for a. a ketoheptose? b. an aldoheptose? c. a ketotriose?
A: Asymmetric(chiral) centre- It is a molecule having a special arrangement of ligands which is not…
Q: A 2-ketohexose is reduced with NaBH4 in CH3OH to form a mixture of D-galactitol and D-talitol. What…
A: Given that a mixture of D-galactitol and D-talitol is formed by the reduction of a 2-ketohexose…
Q: How many stereogenic centers are present in α-D-galactose?
A: The chiral carbon atom bearing four different groups is called as stereo-centre. The position of the…
Q: Convert attached aldohexose to the indicated anomer using a Haworth projection.
A: Carbohydrates are the component of food. They are also called sugars. Aldohexoses are the…
Q: h. Draw a diastereomers of D-Glucose.
A:
Q: Answer the following questions about the eight aldopentoses: Which are enantiomers?
A: Among the eight aldopentoses which are enantiomer has to be given,
Q: 22. How many stereogenic centers are present in fructose, a simple sugar? CH,OH =0 но- H- -OH H- -OH…
A: Answer - The correct option is (c) = 3 Explanation - A stereocenter or stereogenic center is any…
Q: What products are obtained from the reduction of a. d-idose? b. d-sorbose?
A: The structure of D-idose is as: The reduction of D-idose gives
Q: The following trisaccharide derivative is important to human health. The A ring is a deoxy…
A: The structure of ring A is:
Q: Carbohydrates classification.
A: "Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: (b) The optical rotation of a- and B-anomers of D-glucose are [a]p = +112.2° and [a]p = +18.7°, %3D…
A:
Q: Assign R, S designations to each stereogenic center in glucose.
A: The carbon atom which is bonded to four different groups is known as stereogenic or chiral center.…
Q: What sugar is the C-3 epimer of d-xylose? . What sugar is the C-5 epimer of d-allose? What sugar is…
A: Hello. Since your question has multiple sub-parts, we will solve first three sub-parts for you. If…
Please answer question 19 a and b
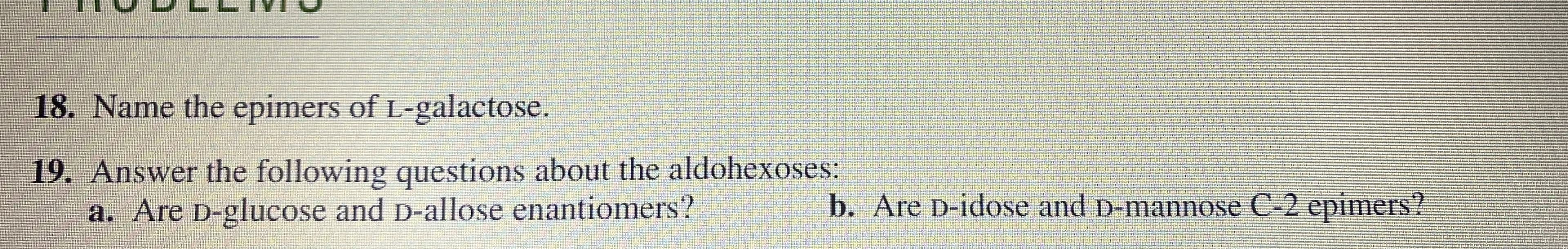
Step by step
Solved in 5 steps with 3 images

- Ribulose is a ketopentose with the anomeric C on C-2. Will this monosaccharide reduce Benedict’s reagent?Answer the following questions about the eight aldopentoses: a. Which are enantiomers? b. Which are C-2 epimers? c. Which form an optically active compound when oxidized with nitric acid?1. Carbohydrates classification. 2. Write down the reactions: a) α,D-Glucopyranose + C2H5OH → b) D-Glucose + [Ag(NH3)2]+ → c) D-Glucopyranose + (CH3CO)2 O → d) D-Glucopyranose + CH3I → e) D-Glucose + HNO3 → f) D-Glucose + H2 → g) Lactose formation h) Sucrose hydrolysis 3. Write down the formula of β,D-galactopyranose
- 26. This compound is L-glyceraldehyde. Draw a stereochemically correct representation of C-1 and C-2 of D-glucose. CHO | HO—C—H | CH2OH 27. Categorize each of the following as an aldose, a ketose, or neither. 28. Define each in 20 words or less: (a) anomeric carbon; (b) enantiomers; (c) furanose and pyranose; (d) glycoside; (e) aldose and ketose. 29. A) Draw the structure of any aldohexose in the pyranose ring form. B) Draw the structure of the anomer of the aldohexose you drew above. C) How many asymmetric carbons (chiral centers) does each of these structures have? D) How many stereoisomers of the aldohexoses you drew are theoretically possible? 30. A) Define "reducing sugar." (b) Sucrose is a disaccharide composed of glucose and fructose (Glc(1 → 2)Fru). Explain why sucrose is not a reducing sugar, even though both glucose and fructose are.D-Tagatose is epimeric of D-Fructose at C4. What is the structure of α-D-Tagatofuranose?What is the configuration of each of the asymmetric centers in the Fischer projection of a. D-glucose? b. D-galactose? c. D-ribose? d.D-xylose? e.D-sorbose?
- 5-Deoxyglucose will close to a?For D-lyxose i. Draw an epimer at C3. ii. Draw a diastereomer that is not an epimer. iii. Draw the anomers of -D-lyxofuranose and -D-lyxopyranose. iv. Draw the product as Haworth projection when reacted with methanol catalysed by acid.What is the difference between1) an aldose and a ketose ?2) a pentose and a hexose ?3) D- and L-forms of sugars (in Fischer projection)?4) a furanose and a pyranose ? 5) α and ß-forms of sugars (in Haworth projection)?
- Draw the α-d-tagatopyranose sugars using Haworth projections:What reaction is responsible for the conversion of linear d-glucose to cyclic d-glucose? A. Reduction reaction B. Oxidation reaction C. Hydrolysis reaction D. Hemiacetal reactiona. How many stereogenic centers are present in α-D-galactose? b. Label the hemiacetal carbon in α-D-galactose. c. Draw the structure of β-D-galactose. d. Draw the structure of the polyhydroxy aldehyde that cyclizes to α- and β-D-galactose.

