2) You have just finished a GC experiment using the standard additions method to determine the amount of toluene in a gasoline sample. The following results were obtained. % toluene toluene internal reference Peak added peak area peak area Ratio 28.918 8.988 1 37.142 9.034 45.364 9.502 3 53.590 9.598 4 61.812 9.139 70.038 9.189
2) You have just finished a GC experiment using the standard additions method to determine the amount of toluene in a gasoline sample. The following results were obtained. % toluene toluene internal reference Peak added peak area peak area Ratio 28.918 8.988 1 37.142 9.034 45.364 9.502 3 53.590 9.598 4 61.812 9.139 70.038 9.189
Chapter8: Sampling, Standardization, And Calibration
Section: Chapter Questions
Problem 8.27QAP
Related questions
Question
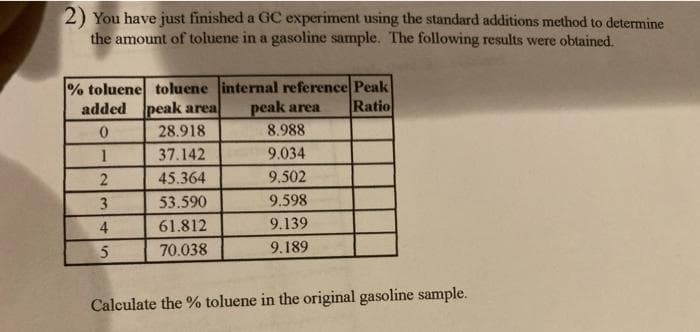
Transcribed Image Text:2) You have just finished a GC experiment using the standard additions method to determine
the amount of toluene in a gasoline sample. The following results were obtained.
% toluene toluene internal reference Peak
added peak area
peak area
Ratio
28.918
8.988
37.142
9.034
45.364
9.502
3
53.590
9.598
4
61.812
9.139
5
70.038
9.189
Calculate the % toluene in the original gasoline sample.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning