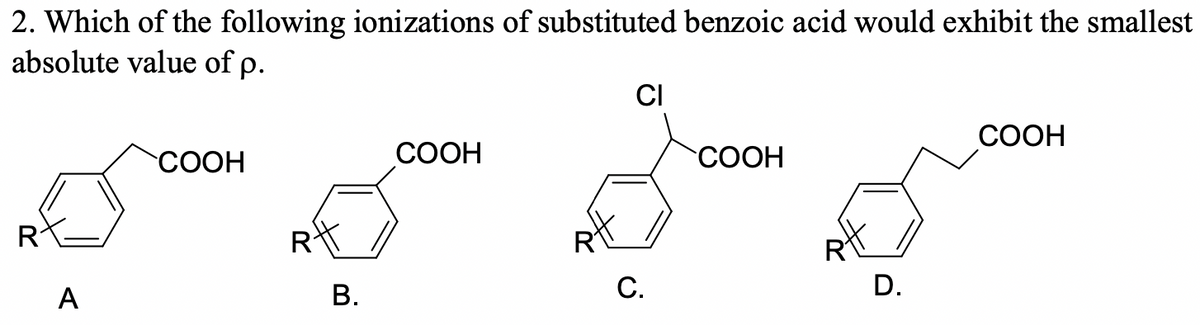
2. Which of the following ionizations of substituted benzoic acid would exhibit the smallest absolute value of p. CI СООН COOH СООН COOH А С. D. B.
Q: Rank the following ions in order of increasing basicity. a. CH,ČH2, CH,0", CH,ÑH c. CH;CO0",…
A: Introduction: According to Bronsted Lowry's concept, an acid is a substance that can donate protons…
Q: Rank the following compounds in the order of INCREASING acidity. но но он II II IV
A: Ans) 2<4<3<1
Q: изон CH3 OH CH3 OH
A:
Q: Rank the following in order of increasing acidity. HS. H3C° НО. A
A:
Q: [References] Arrange the compounds in order of increasing base strength. Consult the table for the…
A: A substance which can give hydroxide ions or can accept protons are called as base. The strength of…
Q: Write the acid dissociation expression (kb) for the following reaction below: a) NH3 (aq) +…
A:
Q: Arrange in order of increasing acidity НО. Но. HO. Но HO, Arrange in order of decreasing acidity HO,…
A: I) When carboxylic acids are dissolved in water they dissocitates into carboxylate ion and the…
Q: 8. Which of the following compounds is the weakest base? (consider stability of conjugate acid) Me D…
A:
Q: Provide an explanation for your chosen order. Explain why pyrrole is a much stronger acid ammonia.…
A:
Q: Arrange the following compounds in order of decreasing their acidity (most first) I. H20 II. C2H6…
A: The acidic strength of any compound depends on the stability of their conjugate bases. with the…
Q: Which indication of relative acid strengths is INCORRECT? Select one: O a. CF3CH2COOH > CH3CH2COOH O…
A: Acidic strength of more for the acid which have more tendency to donate H+ ion in aqueous solution.
Q: NH2 NH2 NH2 NO2
A: The given molecules are: All of these compounds are basic in nature and basicity is inversely…
Q: Rank the labeled protons (Ha–Hc) in mandelic acid, a naturally occurring carboxylic acid in plums…
A: The Hc will the most acidic hydrogen because the conjugate base formed by the deportation of this…
Q: Arrange the compounds in the order of increasing boiling point (lowest first) H3C' `CH3 H2O CH;CH,OH…
A: We have to arrange the following given compounds in order of increasing boiling point as follows in…
Q: 2-Hydroxybutanedioic acid occurs naturally in apples and other fruits. Rank the labeled protons…
A:
Q: Arrange the following in order of decreasing basicity (most basic first). I. CH3CH2M8BR II. HC°CMgBr…
A: Concepts of acids and bases.
Q: Answer questions a-c about the Bronsted acid-base reaction below using the identifying letters A-D…
A: Since you have asked multiple questions, I am solving first question for you in details. You can see…
Q: | Rank the following compounds from strongest acid to weakest acid. `OH F3C *OH A D
A: Carboxylic acids and ketones both belong to the class of carbonyl group. But carboxylic acids are…
Q: Answer questions a-c about the Bronsted acid-base reaction below using the identifying letters A-D…
A:
Q: Arrange each group of compounds in order of increasing acidity.(c) benzoic acid, o-nitrobenzoic…
A:
Q: The following reaction runs as written therefore... 1HClO2 +1NH3 ==>1ClO2- +1NH4+ (a)HClO2 is a…
A: The reaction given is, => HClO2 + NH3 -------> ClO2- + NH4+ Given : The reaction runs in the…
Q: 2. Which carboxylic compound of each pair would you expect to be stronger acid? Indicate (a) or (b)…
A: The compound in which negative charge will be more stable will be more acidic in Nature. Hence…
Q: F ONLY- ( STRUCTURAL ANSWER ONLY)
A:
Q: Rank the following compounds in order of increasing acidity of their mostacidic hydrogen atom. (See…
A: The compound C is the least acidic because the electronegativity of nitrogen is less than that of…
Q: Rank the following compounds in order of decreasing acidity. CH4 F3C-S HO. H3C-S- HO- HO, A C A: […
A: Solution : The acid-base reaction involves the donation of a proton from an acid to a base. This…
Q: OEt EtO OEt
A: In this question, we have to find out the correct answer of given problem by the help of the…
Q: Arrange the given compounds based on their relative Brønsted acidities. H-F H-Br H-SH H-CH3…
A: By Bronsted theory, a species that is capable of giving away a proton are termed as Bronsted acids.…
Q: Since each molecule of acetic acid which ionizes produces 1 H+ ic concentration in the solution are:…
A: Weak acid ionized partially. Degree of ionization (a) = 1.34 % = 0.0134 initial concentration of…
Q: What is the correct rank of the following compounds in order of decreasing acidity? NH2 HO II IV…
A: Acidity of any molecule is directly proportional to the stability of -ve charge formed after…
Q: Which solution has the highest concentration of H3O+: a 0.50 M solution of acetic acid (CH3CO2H),or…
A: ∝=Degree of Dissociation HA⇌ H+ + A-t=0 C 0 0t=eq…
Q: Identify the strongest acid O H2Te O H2S O H2Se O H2O O not enough information is available
A: The hydrides of group 16 are weakly acidic. The strength of the acidity of these hydrides can be…
Q: 1.3. Using concepts learned from acid-base theory in class, choose species from the list below that…
A: 1.3. More the electronegative difference, stronger the interaction. Out the given anions, F- has the…
Q: СООН OH CH3 СООН NO2 CI I II III IV
A: Acid are those species that donate H+ ion or accept a pain of an electron. Acidic strength depends…
Q: Select the weaker acid from each of the following pairs: I. HI or HBr II. H3AsO3 or H2SeO3 III.…
A: In this question we will compare the acidic character between two compounds in each case and will…
Q: Rank the labeled protons (Ha–Hc) in mandelic acid, a naturally occurring carboxylic acid in plums…
A: The stability of a conjugate base formed after the removal of a proton decides the acidic strength…
Q: Rank these hydrogens as to acidity. H;C HO. нн A C A most, C least O C most, B least O A most, B…
A:
Q: Amphetamine is a powerful stimulant of the central nervous system.Draw the products formed (and…
A: We have to predict the products and flow of electrons.
Q: -NH3 NH3 но- -NH3 H3C- -NH3 I II III IV
A: Acidity completely depends on the stability of conjugate base of that acid. If it is most stable…
Q: 4. Watter verbinding (i of ii) is die sterker basis? Bespreek jou antwoord volledig deur o.a. h suur…
A: Strong base:- A Lewis base have capacity to donate its lone pair electrons.
Q: Using the Identity of X in HX to Determine Relative Acidity Without reference to a pKa table, decide…
A: Since F is more electronegative than O, the H-F bond is more polar than H-O bond. The H-F bond…
Q: How to determine the Relative Acidity of Protons ? Explain all steps ?
A: Relative acidity of protons is calculated when different types of protons are attached in molecules.…
Q: [References] Answer questions a-c about the Bronsted acid-base reaction below using the identifying…
A: In general, smaller the pKa value of an acid, stronger is the acid. And according to Bronsted…
Q: 27. Rank the following conjugate bases in order of decreasing basicity, putting the most basic…
A: Since you have posted multiple questions, as per guidelines we can answer 1 per session . If you…
Q: Rank the compounds in each group in order of increasing acidity.
A:
Q: Which of the following bases are the STRONGEST? pyridine, C5H5N urea, NH2CONH2…
A: Bases are the substance that gives hydroxide ion in the solution. Lower the value of pkb, higher…
Q: Rank the following species in order of increasing acidity. Explain your reasons for ordering them as…
A: Species in order of increasing acidity : NH3 < H2O < CH3OH < CH3COOH < HF <…
Q: IV. Stability O2N- CF3 D A B H3CO- G H E F V. Acidity он HO- O2N- B A ON F3C он (H3C)2N- OH D E F…
A:
Q: Solve the Attachment
A: Acids tend to lose H+ and form conjugate base. Stronger the acid, more is the Ka value and smaller…
Step by step
Solved in 3 steps with 1 images

- According to the Evan's pKa table, phenol has a pKa of 9.95. p-Nitro and m-nitrophenol have pKas of 7.14 and 8.35 respectively. Using resonance structures, explain the observed trends in the acidity of these molecules.Identify the correct ranking of the structures from highest acidity to lowest acidity for the bolded hydrogen. The correct answer is D, please explain why that is correct and why other options are wrong.1.Explain why pyridine ( Kb=2.3x10-9) is a much stronger base than pyrrole (Kb=2.5x10-14), 2.Explain and illustrate, why it is difficult to perfom Friedel-Craft reactions on unactivated pyridine.
- Compare these two reactions attached, with different Lewis bases and the same Lewis acid. include, at least, kinetic aspect and thermodynamic aspect along with the detailed structural features. show the final products in Fisher projections.based on this video: on Hinsberg Test Tests for Amines - MeitY OLabs (8.5min) https://www.youtube.com/watch?v=j5jgMUWri8U Write the reaction (two-step, in skeletal) of each test amine when tested in the Hinsberg Test.The Ka of acetic acid (CH3CO2H) is 1.8x10-5 and the Kb of methylamine (CH3NH2) is 4.4x10-4. Complete the following equilibrium reaction (acid-base reaction) equation and predict the side of the equilibrium that is favored. Explain. Show all your work.



