22-23 The two optically B-keto acids below were decarboxylated using the conditions typically used for the acetoacetate synthesis. Will the ketone products also be optically active? Provide the complete mechanism to explain your answer. (a) Нао* ОН Нeat СНз СНз (b) Нзо* ОН Нeat H CH3
22-23 The two optically B-keto acids below were decarboxylated using the conditions typically used for the acetoacetate synthesis. Will the ketone products also be optically active? Provide the complete mechanism to explain your answer. (a) Нао* ОН Нeat СНз СНз (b) Нзо* ОН Нeat H CH3
Chapter22: Carbonyl Alpha-substitution Reactions
Section22.SE: Something Extra
Problem 30MP: One of the later steps in glucose biosynthesis is the isomerization of fructose 6-phosphate to...
Related questions
Question
hi i want to help me
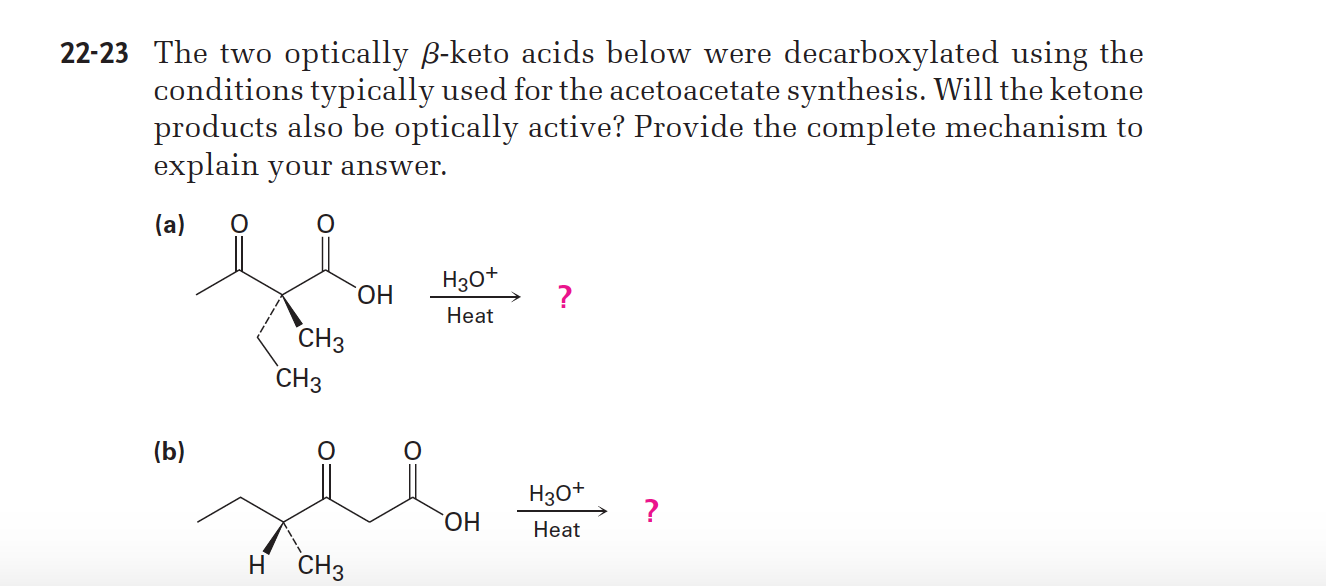
Transcribed Image Text:22-23 The two optically B-keto acids below were decarboxylated using the
conditions typically used for the acetoacetate synthesis. Will the ketone
products also be optically active? Provide the complete mechanism to
explain your answer.
(a)
Нао*
ОН
Нeat
СНз
СНз
(b)
Нзо*
ОН
Нeat
H CH3
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning


Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning