3) The rate of change of molar concentration of methyl radicals CH₂ in reaction: 3 2 CH3 (g) → CH₂CH₂ (g) was reported as d[CH₂]/dt = -1.0 mol dm³¹ s¯¹. Find: a) the rate (formula and numeric value) of overall reaction v; ú) the rate (formula and numeric value) of formation of ethane CH CH
3) The rate of change of molar concentration of methyl radicals CH₂ in reaction: 3 2 CH3 (g) → CH₂CH₂ (g) was reported as d[CH₂]/dt = -1.0 mol dm³¹ s¯¹. Find: a) the rate (formula and numeric value) of overall reaction v; ú) the rate (formula and numeric value) of formation of ethane CH CH
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter11: Chemical Kinetics: Rates Of Reactions
Section11.1: Reaction Rate
Problem 11.2CE: Instantaneous rates for the reaction of hydroxide ion with Cv+ can be determined from the slope of...
Related questions
Question
Question 3
![3) The rate of change of molar concentration of methyl radicals
CH, in reaction:
2 CH₂ (g) →>> › CH₂CH₂ (g)
was reported as d[CH₂]/dt = -1.0 mol dm³¹ s²¹.
Find:
a)
the rate (formula and numeric value) of overall reaction v;
b) the rate (formula and numeric value) of formation of ethane
CH₂CH₂.](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2Fbc2a6021-039e-42ef-ade4-d4f55d9b1f3f%2F59f31d87-6722-4d37-9f13-75a7054ef0f2%2Fp9rd8l_processed.jpeg&w=3840&q=75)
Transcribed Image Text:3) The rate of change of molar concentration of methyl radicals
CH, in reaction:
2 CH₂ (g) →>> › CH₂CH₂ (g)
was reported as d[CH₂]/dt = -1.0 mol dm³¹ s²¹.
Find:
a)
the rate (formula and numeric value) of overall reaction v;
b) the rate (formula and numeric value) of formation of ethane
CH₂CH₂.
Expert Solution
Step 1
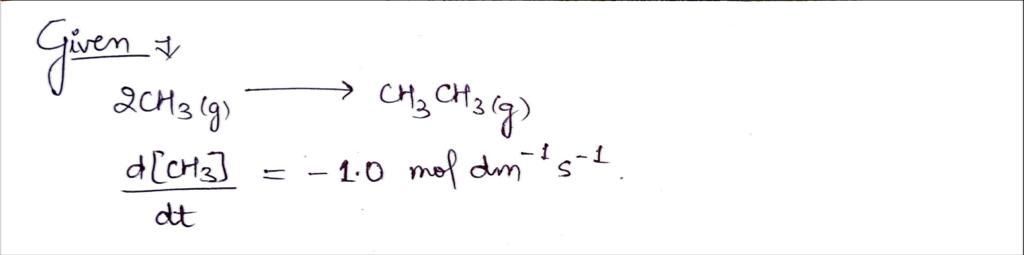
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning