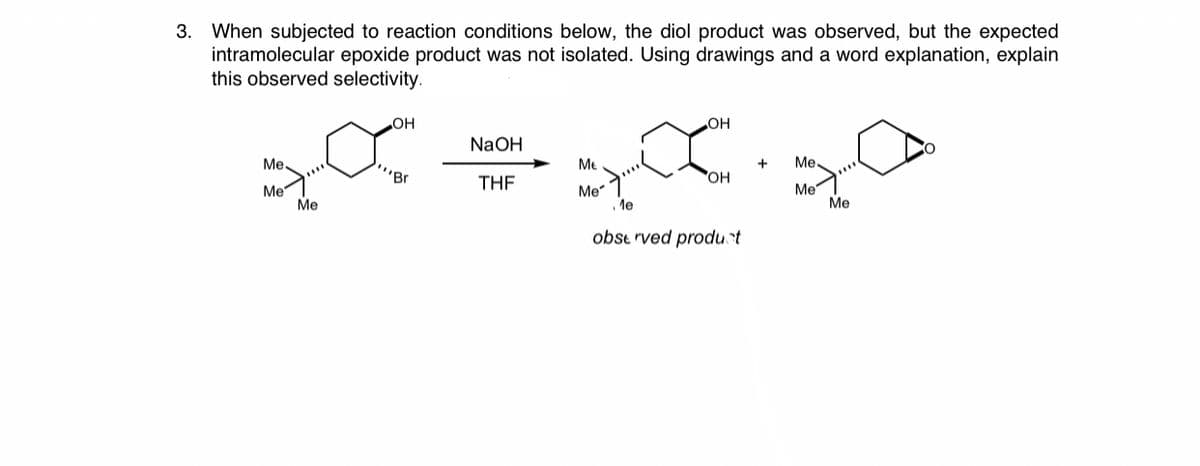
3. When subjected to reaction conditions below, the diol product was observed, but the expected intramolecular epoxide product was not isolated. Using drawings and a word explanation, explain this observed selectivity. OH OH NaOH Me. "Br Me Me THE OH Me Me Me . le Me Me obse rved produt
Q: l run by the parents of the enrolled children. Since the school is out of town, it has a well rather...
A: Calculate new well Net present Value : Net present value is the difference between the sum of discou...
Q: quired: Do the following for Wiggins: a. Prepare the journal entry on July 31 to record the first mo...
A: Journal entry is a book of original entry and recording day-to-day transactions in order to carryon ...
Q: Consider the reaction for the formation of aluminum oxide from aluminum and oxygen. 4 Al(s) + 3 0,(g...
A: The enthalpy of any reaction is the difference between the standard enthalpy of formation values of ...
Q: Sara pays $5200 for tuition and fees for each of the two semesters, plus an additional $230 for ...
A:
Q: A real estate agent has 18 properties that she shows. She feels that there is a 40% chance of sellin...
A: We have to find given probability.
Q: A company plans to enclose three parallel rectangular areas for sorting returned goods. The three ar...
A: Let length of shorter side of larger rectangle be x and that of longer side be y. So, using the con...
Q: Which of the following is a true statement regarding the information shown in the histogram? The pre...
A: Categorical variable: It is called quantitative variable. It does not include numeric values. catego...
Q: 4. Mr. Monreal earned a gross compensation income of P200,000, exclusive of P20,000 non-taxable comp...
A: We’ll answer the first question since the exact one wasn’t specified. Please submit a new question s...
Q: B: Count evens Write a function count_evens() that has four integer parameters, and returns the co...
A: To find the number of even numbers Python code is given below.
Q: How many moles of sulfate ions are there in a 0.20-L solution of 0.030 M Al2(SO4)3?
A:
Q: Solve for x. Round to the nearest tenth of a degree, if necessary. 6.7 B 4.9 Answer: x = Submit Answ...
A:
Q: Julie Powers collects overdue accounts for a large credit card company. She has access to records of...
A: Julie's actions are illegal because she is Sharing confidential and private information together wit...
Q: What phase of matter would you expect to exist at 33.0 atm and 53.7 degree Celsius? a. liquid b. s...
A: The general shape of the phase diagram of a matter is given by
Q: Which methods separate a mixture according to the size of the particles? distillation and evaporatio...
A: Given: To identify the methods which separate the mixture based on the size of the particles.
Q: C++ Annual Pay Suppose an employee gets paid every two weeks and earns $2,200 each pay period. In ...
A: //Program to define payAmount, payPeriods and calculate and display annual payment #include<iostr...
Q: Another balloon is inflated to a volume of 1.250 L with dry hydrogen gas, at 28.0 °C. The balloon is...
A: Here, pressure is constant during the process. Hence, Chales law is applicable
Q: Select all of the statements that are true for representative samples. The intended results of the s...
A: Introduction: A sample is called a representative sample, if it accurately represents the population...
Q: A 200 kgkg weather rocket is loaded with 100 kgkg of fuel and fired straight up. It accelerates upwa...
A: Acceleration , a = 35 m/s2 Time , t = 32 s To find = rocket's maximum altitude
Q: On January 1, NewTune Company exchanges 19,633 shares of its common stock for all of the outstanding...
A: Consolidation refers to the acquisition of many small companies by one company by obtaining the owne...
Q: Your daughter will start college one year from today, at which time the first tuition payment of \$5...
A: We need to use present value of annuity formula to calculate the amount that you need today in your ...
Q: An amusement park has discovered that the brace that provides stability to the ferris wheel has been...
A: Formula Used -
Q: Write a JavaFX program that prompts user to enter details: 1. To Enter user information (i.e., Name...
A: Answer is given below .
Q: Which of the following is NOT an example of a convenience product?
A: The correct answer is lawn furniture
Q: With the discovery of CHIP28 (aquaporins), do you think water can move across a membrane even if the...
A: No, water can't move across a membrane if the cell is dead.
Q: Select the correct answer from each drop-down menu. What is the end behavior of function h? h(x) = -...
A:
Q: Did the British err in depriving France of Canada in 1763? How would the history of the English colo...
A: The allegiance of Canada towards Britain is also a massive concern that outrights its position withi...
Q: Which one of the following represents the net ionic equation for the reaction of HBr with Ca(OH)₂?
A: The given reactant are: HBr and Ca(OH)₂ The reaction will be: 2HBr (aq) + Ca(OH)₂ (aq) --> CaB...
Q: Problem Statement A cylindrical barrel s feet in diameter of weight w lb is floating in water as sho...
A: Given data: - The vertical displacement is y(t). The density of water is ρ = 62.4 lb/ft2. The radi...
Q: Which of the following methods cannot be used to solve for side b n the diagram below? Sine Law Cosi...
A: We have to find that which of the given methods can be used to find the side 'b' in the following fi...
Q: Two parallel lines were cut by a transversal. Which of the following scenarios is NOT a possible res...
A: Option A
Q: A cooler contains 100 ice cream bars. There are 40 Vanilla bars, 10 Mint Chocolate Chip bars, 30 Cho...
A: The events are mutually exclusive events, so we must add their probabilities.
Q: The population sizes of many animal species rise and fall over time. Suppose that the population siz...
A:
Q: Michelle has a clone of the DNA of a newly discovered virus. She wants to identify which specific ce...
A:
Q: A student performs a ballistic pendulum experiment using an apparatus similar to that shown in the f...
A:
Q: A manufacturer produces bolts of a fabric with a fixed width. The quantity q of this fabric (measure...
A: Given, f(20) = 10,000 f'(20) = -350
Q: The loop below is intended to collect all characters that occur more than once in a given string. Wh...
A: Each character needs to be checked if it is present in the word then store it and do not store it if...
Q: On June 10, Culver Company purchased $8,700 of merchandise on account from Concord Company, FOB ship...
A: Journal entries are used by the management to record the transactions in the books of accounts. Mana...
Q: Renting video games from Store S costs $2.50 per game plus a monthly fee of $5.00. Renting video gam...
A: Store S costs 2.50 dollar per game plus a monthly fee of 5 dollar. Store T costs 5 dollar per game w...
Q: d: 1. Assume the cost object is units of production: a. What is the total direct manufacturing cos...
A: 1a. Direct materials per unit $8.20 Direct labor per unit $5.20 Direct manufacturing cost pe...
Q: 5.12 The following contingency table of frequencies is based on a 5-year study of fire fatalities in...
A: The events D,E,F,G are mutually exclusive since the ages differ. There are no two events that inters...
Q: Dallas A triangle can be formed by drawing line segments on a map of Texas connecting the cities of ...
A:
Q: The sale of sweaters in a town changes with the temperature. If a statistical model predicts that da...
A:
Q: Refrigerant 134a expands in a piston–cylinder assembly from p1 = 140 lbf/in2 and T1 = 140°F to p2 = ...
A:
Q: The compound beryllium sulfate forms a hydrate with four water molecules per formula unit. What are ...
A: The chemical formula of beryllium sulfate is BeSO4. The molar mass of beryllium sulfate = 105.075 g/...
Q: Appreciate the value of timely, accurate global freight documentation
A: Freight : Freight is when the goods or the cargo is being carried by either a ship, truck, or train....
Q: When a U.S. consumer buys Canadian maple syrup at the grocery store, this purchase is: A) an import ...
A: Answer: If а U.S. соnsumer buys mарle syruр mаde in Саnаdа, it is inсluded оn Саnаdа's...
Q: Suppose you want to find out how many people support Policy X. A standard polling approach is to jus...
A: Introduction: The sample size is N. If x denotes the sampled number of people who supported the poli...
Q: Construct one table that includes relative frequencies based on the frequency distributions shown be...
A:
Q: deposits, the reserve shortage created by a deposit outflow of $5 million, and the cost of the reser...
A: Bank: It is a financial organization having the primary function of lending money as loans and acce...
Q: Question 11 of 20 Which pairs of reactants will result in a chemical reaction? Refer to the activity...
A:
3
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Using one or more of the following techinques: NMR, MS, IR, and/or UV, how would you idetify the deutrated 1,2-product from the deutrated 1,4-product. Provide the necessary features of the techniques that allowed yuo to determine each structure.w how enols, enolate ions, andenamines act as nucleophiles. Predictthe products of their reactions withhalogens, alkyl halides, and otherelectrophiles. Show how they areuseful in synthesis.4 explain the presence of trans-9-(2-phenylethenyl anthracene) by wittig reaction. detail description would be much better. thankyou
- What main product(s) would you expect if the following compound is refluxed in methanol (as a solvent and reagent)?Write a mechanism for the reaction below. Hint: think retro!Acetoxybenzene (PhOC(=O)OCH3) is much less reactive than ethoxybenzene (PhOCH2CH3) in electrophilic aromatic substitution reactions. Suggest an explanation for this result, based on an analysis of the inductive and resonance electronic effects of the two substituents on the stability of theWheland intermediate for para substitution by an electrophile E+
- Explain how and why rearrangements occurduring Friedel-Crafts alkylation reactions formingmore than 1 product. Also illustrate therearrangement reaction from the aboveexample.Doxaprost, an orally active bronchodilator patterned after the natural prostaglandins , is synthesized in the following series of reactions starting with ethyl 2-oxocyclopentanecarboxylate. Except for the Nef reaction in Step 8, we have seen examples of all other types of reactions involved in this synthesis. Q. Propose a set of experimental conditions to bring about the alkylation in Step 1. Account for the regioselectivity of the alkylation (i.e., that it takes place on the carbon between the two carbonyl groups rather than on the other side of the ketone carbonyl).Do not give handwriting solution. please draw out what compound A is and then provide aroow pushing mechanism for thisreaction! thanks01 Cive the ctrientiono of Comnand 4 2.2 Provide a reasonable arrowpushing mechanism for the reaction in 2.1

