300.0 mg conc 0.100 M expID: HM1-001 scale 1) EDCI, HOBT hydrate DIPEA O`HN NH H3CO HN DCM:DMF (1:2) NH 2) workup with 1M HCI (aq.) (add first for 10 min (add second after pre-activation) pre-activation) Boc-Asp(OMe)-OH•DCHA H-Phe-OtBu•HCI Boc-Asp(OMe)-Phe-OtBu molar MW m d V
300.0 mg conc 0.100 M expID: HM1-001 scale 1) EDCI, HOBT hydrate DIPEA O`HN NH H3CO HN DCM:DMF (1:2) NH 2) workup with 1M HCI (aq.) (add first for 10 min (add second after pre-activation) pre-activation) Boc-Asp(OMe)-OH•DCHA H-Phe-OtBu•HCI Boc-Asp(OMe)-Phe-OtBu molar MW m d V
Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.9QAP
Related questions
Question
FILL IN THE BLANKS.
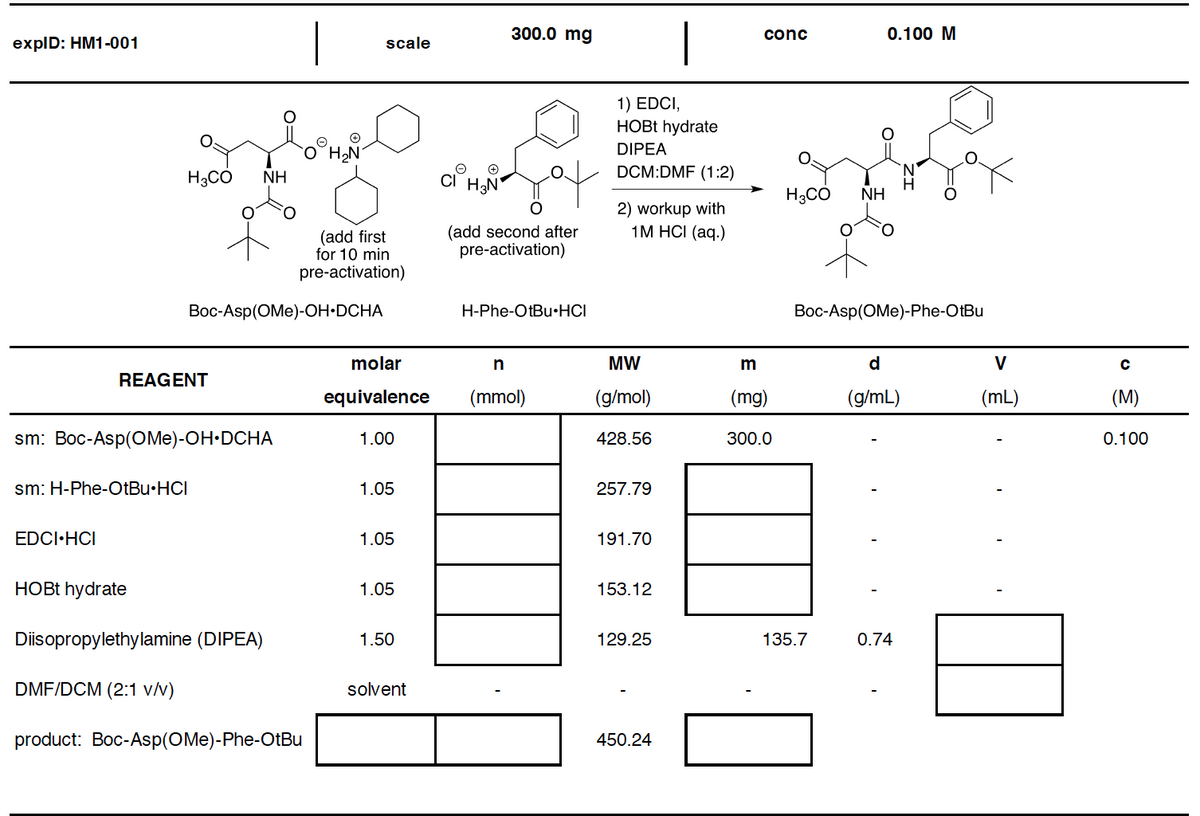
Transcribed Image Text:|
|
300.0 mg
conc
0.100 M
explD: HM1-001
scale
1) EDCI,
HOBT hydrate
DIPEA
H3CO
NH
DCM:DMF (1:2)
H3CÓ
NH
2) workup with
1М HC (аq.)
(add first
for 10 min
pre-activation)
(add second after
pre-activation)
Вос-Asp(OMe)-ОН-DCHA
H-Phe-OtBu•HCI
Вос-Asp(OMe)-Phe-OtBu
molar
MW
d
V
REAGENT
equivalence
(mmol)
(g/mol)
(mg)
(g/mL)
(mL)
(M)
sm: Boc-Asp(OMe)-OH•DCHA
1.00
428.56
300.0
0.100
sm: H-Phe-OTBU•HCI
1.05
257.79
EDCI·HCI
1.05
191.70
HOBT hydrate
1.05
153.12
Diisopropylethylamine (DIPEA)
1.50
129.25
135.7
0.74
DMF/DCM (2:1 v/v)
solvent
product: Boc-Asp(OMe)-Phe-OtBu
450.24
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 5 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

