: 40. For Lactic acid, estimate the value of the bond angles identified by letters (A, B, C) in the Lewis diagram. Explain your reasoning in each case. H H:0: Н-С-С-С О-н 0: Н Н
: 40. For Lactic acid, estimate the value of the bond angles identified by letters (A, B, C) in the Lewis diagram. Explain your reasoning in each case. H H:0: Н-С-С-С О-н 0: Н Н
Chemistry by OpenStax (2015-05-04)
1st Edition
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Chapter10: Liquids And Solids
Section: Chapter Questions
Problem 18E: Under certain conditions, molecules of acetic acid, CH3COOH, form dimers, pairs of acetic acid...
Related questions
Question
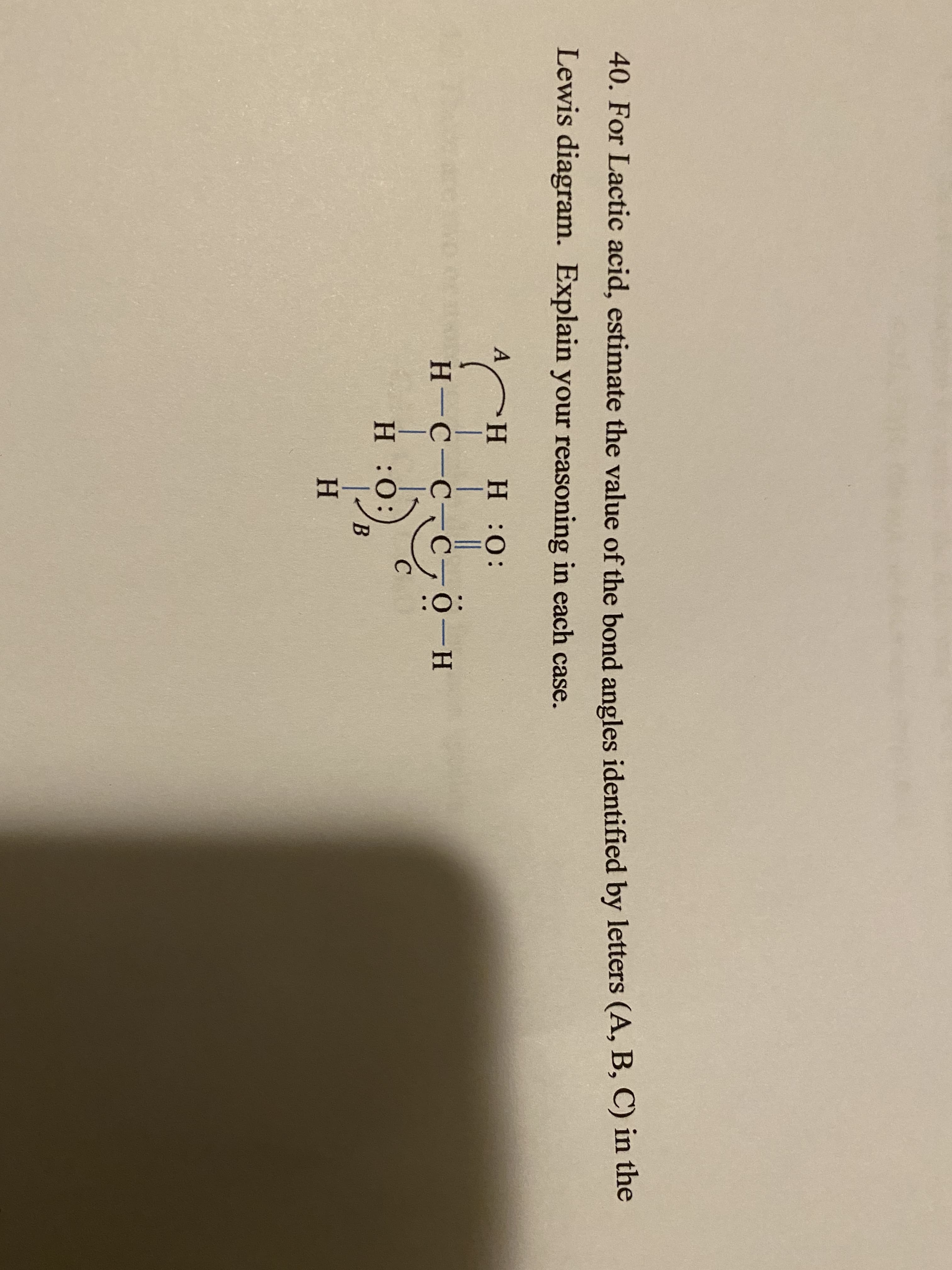
Transcribed Image Text::
40. For Lactic acid, estimate the value of the bond angles identified by letters (A, B, C) in the
Lewis diagram. Explain your reasoning in each case.
H H:0:
Н-С-С-С
О-н
0: Н
Н
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax