Q: The sugar glucose (C6H12O6) is broken down in a decomposition reaction to ethanol (C2H6O) and…
A:
Q: Balance the reaction below: Use the lowest possible positive integers as coefficients H2C204-2H20 +…
A: The balanced equation is, 1 H2C2O4.2H2O + 2 NaOH ----> 4 H2O + 1 Na2C2O4
Q: balance the following unbalanced chemical equation showing the combustion of methane use 1 for…
A: Given that, Combustion of methane i.e., _CH4 + _ O2 ---------> _CO2 + _H2O Write balanced…
Q: Balance the following chemical reaction and determine the coefficients of H2O and AIK(OH)4,…
A: Balance chemical equation: Balanced chemical equation can be defined as a reaction in which number…
Q: 3. Give the balanced equation which shows the decomposition reaction of solid aluminum chlorate…
A:
Q: 100. Is it possible for a reaction to occur with a yield that is greater than 100%? Why or why not?
A:
Q: Balance the following chemical reaction and determine the coefficients of H20 and AIK(ÓH)4,…
A: Balance the following chemical equation Al + KOH + H2O --------> AlK(OH)4 + H2
Q: Balance the following equation: The combustion of Butane Gas: C4H10 + O2 --------->…
A: The unbalanced equation is C4H10 + O2 ----------> CO2 + H2O
Q: Balance the following chemical equation (if necessary) for the combustion reaction of propane:…
A: In multiple questions we solve only first question according to Bartleby guidelines. Balance the…
Q: Q) balance the following equation in details. 0 p.m. KCI + HNO3+O2>KNOg+Czt HeO
A: The balancing of chemical equation means that the number of atoms in the reactant are equal to…
Q: Many cars can run on a mixture of gasoline and ethanol, C2H5OH (density = 0.789 g/mL). What mass of…
A:
Q: Which of the following features is internal-intrinsic? A. Iron cube volume B. NaCl(s) Crystal…
A: Intrinsic features are those features which do not depend on the volume or mass or number of…
Q: Balance the following chemical reaction and determine the coefficients of H2O and AlK(OH)4,…
A: A balanced chemical equation states that the number of elements in the reactant side is equal to the…
Q: Balance the following equation. Include the states of matter for each reactant and product. Fe(s) +…
A: Answer:- This question is answered by using the simple concept of balancing the chemical equation in…
Q: Balance the following equation by algebraic method. CAH10 CO, H,0
A:
Q: Balance the following chemical equation using the smallest, whole-number coefficients: C6H12O6 + O2…
A: Coefficient of reactant : The number written before the formula of reactant is known as coefficient…
Q: Balance the equation below such that it has the smallest, whole-number coefficients.…
A: The given chemical equation is, CH3CH2CH2CH2OH+O2g→CO2g+H2Og The name of reactant CH3CH2CH2CH2OH is…
Q: Titanium is a strong, slightly corrosion resistant metal and is used in rockets, airplanes, jet…
A: Molar mass of Ti=47.86g/mol given mass of Ti=791×106g Molar mass of Mg=24.305g/mol Given mass of…
Q: Fe + Cuo-Fe2O3+ Cu
A: In this problem the provided equation has to be balanced: Fe + CuO → Fe2O3 + Cu Iron reacts with…
Q: Balance the following chemical equations by choosing the correct coefficients from the dropdown…
A:
Q: Balance the following chemical equation (if necessary): C.H«(g) + O:(g) – CO:(g) + H:O(g)
A:
Q: Balance the following chemical reaction and determine the coefficients of H2O and AIK(OH)4,…
A: A question based on stoichiometry that is to be accomplished.
Q: Balance the following chemical equations: Fe + O2 à Fe2O3
A: Process for balance the reaction 1. Determine n facor of reactant . 2. Cross multiply 3. Balance…
Q: Balance the reaction: ___ C6H14 (l) + ___ O2 (g) → ___ CO2 (g) + ___ H2O (g) Then give the…
A: A balanced chemical equation states that the number of elements in the reactant side is equal to the…
Q: Lime, CaO, can be produced in two steps as shown in the equations below. If the percentage yield of…
A:
Q: Balance the following chemical reaction and determine the coefficients of H2O and AIK(OH)4,…
A: The Chemical reaction follows the law of conservation of mass. According to the law of conservation…
Q: After balancing the chemical equation for the combustion in air of liquid C3H8O using the smallest…
A: Sum of the coefficients in the product side can be determined from the balanced chemical equation…
Q: Phosphoric acid, which is commonly used as rust inhibitor, food additive and etching agent for…
A: Chemical reactions can be added, subtracted, or multiplied like an algebraic equation.
Q: 2. Balance the following equation: BaSO4 + NiCl4 ------> BaCl2 + Ni(SO4)2…
A: Answer:- This question is answered by using the simple concept of balancing a chemical equation…
Q: Balance the equation by inserting coefficients as needed. ___C2H6O+____O2⟶___CO2+____H2O
A: Balancing the equation by inserting coefficients
Q: Phosphoric acid, which is commonly used as rust inhibitor, food additive and etching agent for…
A: Chemical reactions can be added, subtracted, or multiplied like an algebraic equation.
Q: Phosphoric acid, which is commonly used as rust inhibitor, food additive and etching agent for…
A: A chemical reaction can be added or subtracted like an algebraic equation.
Q: balance the following equation and then select the correct ordering of coefficients ___C8H18 + ____…
A: If the number of atoms on each element in each side are same then it is called balanced equation
Q: Balance the following equation and include the states of matter: C6H12O6 (s) → C2H6O (l) + CO2…
A:
Q: Consider the reaction of nitrogen monoxide with hydrogen to form nitrogen and water vapor, H2O(g):…
A: Given Reaction NO(g) + H2(g) → 12 N2(g) + H2O(g) ∆H = - 332.2 KJ Balance reaction with…
Q: balance the following unbalanced chemical equation showing the combustion of methane. use 1 for a…
A: The chemical name of CH4 is methane gas The chemical name of O2 is oxygen gas The chemical name of…
Q: A typical formulation for window glass is 75% SiO₂, 15% Na₂O, and 10.% CaO by mass. What masses of…
A: 75% SiO2 by mass means 75 g of SiO2 is present in 100 g of glass. Now Calculate the mass of SiO2 in…
Q: Balance the following equation. What is the sum of the coefficients in the balanced equation?…
A:
Q: Balance the following chemical equation: P4O10 + H2O --> H3PO4 What is the numerical coefficient in…
A: Balancing of the equation means that, Number of reactant side atom = Number of product side atom…
Q: Give the product of the given reactants, balance the equation, and identify the type of changes the…
A: Give the product of the given reactants, balance the equation, and identify the type of changes the…
Q: Write the net chemical equation for the production of phosphoric acid from phosphorus, oxygen and…
A: The phosphoric acid is called as orthophosphoric acid and phosphoric (V) acid. It is a weak acid.…
Q: The reaction between potassium chlorate and red phosphorus takes place when you strike a match on a…
A: Given mass of KClO3 = 52.9 gram mass of P4O10 = ? Molar mass of P4O10 = ( 4 × 31 ) + ( 10 × 16…
Q: Titanium is a strong, slightly corrosion resistant metal and is used in rockets, airplanes, jet…
A: We have to find the number of moles first. No of moles of TiCl4 = Mass / Molecular mass of TiCl4…
Q: When properly balanced, which of the following is the sum of the balancing coefficients for the…
A:
Q: Balance the following reaction: Al + Fe203 → Al203 + Fe *remember to enter a 1 for coefficient is…
A: In a balanced chemical reaction the total number of atom in left side is equal to the total number…
Q: NH3+02----->NO+H2O Balance the Equation
A:
Q: Phosphoric acid, which is commonly used as rust inhibitor, food additive and etching agent for…
A:
Q: Balance the following unbalanced chemical equation showing the combustion of methane. _______ CH4 +…
A: Balancing of a chemical reaction is to make total no. Of atoms same on both the side of arrow i.e on…
Q: Balance the following chemical reaction and determine the coefficients of Fez(SO4)3 and H20,…
A: In balanced reaction number of each atom on reactant side is equal to number of each atom on product…
Q: What is the balanced equation of 3m? What is the balance equation of Ksp= [Ag+]²[CrO4²-]
A: We have to just write the balanced equation.
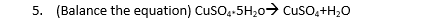
A balanced chemical reaction equation has equal number of atoms and charges on both reactant and product side.
Step by step
Solved in 2 steps with 1 images

- A rebreathing gas mask contains potassium superoxide, KO2, which reacts with moisture in the breath to give oxygen. 4KO2(s)+2H2O(l)4KOH(s)+3O2(g) Estimate the grams of potassium superoxide required to supply a persons oxygen needs for one hour. Assume a person requires 1.00 102 kcal of energy for this time period. Further assume that this energy can be equated to the heat of combustion of a quantity of glucose, C6H12O6, to CO2(g) and H2O(l). From the amount of glucose required to give 1.00 102 kcal of heat, calculate the amount of oxygen consumed and hence the amount of KO2 required. The ff0 for glucose(s) is 1273 kJ/mol.Use the web to determine the differences in the amounts of aluminum recycled in states where there are deposits on aluminum cans versus states where recycling is voluntary. What is the most reliable way to estimate this value? mat uncertainty is there in the estimate?A power plant is driven by the combustion of a complex fossil fuel having the formula C11H7S. Assume the air supply is composed of only N2 and O2 with a molar ratio of 3.76:1.00, and the N2 remains unreacted. In addition to the water produced, the fuels C is completely combusted to CO2 and its sulfur content is converted to SO2. In order to evaluate gases emitted at the exhaust stacks for environmental regulation purposes, the nitrogen supplied with the air must also be included in the balanced reactions. a Including the N2 supplied m the air, write a balanced combustion equation for the complex fuel assuming 100% stoichiometric combustion (i.e., when there is no excess oxygen in the products and the only C-containing product is CO2). Except in the case of N2, use only integer coefficients. b Including N2 supplied in the air, write a balanced combustion equation for the complex fuel assuming 120% stoichiometric combustion (i.e., when excess oxygen is present in the products and the only C-containing product is CO2). Except in the case of use only integer coefficients c Calculate the minimum mass (in kg) of air required to completely combust 1700 kg of C11H7S. d Calculate the air/fuel mass ratio, assuming 100% stoichiometric combustion. e Calculate the air/fuel mass ratio, assuming 120% stoichiometric combustion.
- When one mole of ethylene gas, C2H4, reacts with fluorine gas, hydrogen fluoride and carbon tetrafluoride gases are formed and 2496.7 kJ of heat are given off. What is Hf for CF4(g)?Natural gas companies in the United States use the therm as a unit of energy. One therm is 1105 BTU. (a) How many joules are in one therm? (1J=9.48104BTU) (b) When propane gas, C3H8, is burned in oxygen, CO2 and steam are produced. How many therms of energy are given off by 1.00 mol of propane gas?Carbon dioxide from the atmosphere weathers, or dissolves, limestone (CaCO3) by the reaction CaCO3(s)+CO2(g)+H2O(l)Ca2(aq)+2HCO3(aq) Obtain H for this reaction. See Table 6.2 for the data.
- The meat from one hazelnut has a mass of 0.985 g. (a) What is the mass of a millionth of a mole (106) of hazelnut meats? (A millionth of a mole is also called a micromole.) (b) How many moles are in a pound of hazelnut meats?The carbon dioxide exhaled in the breath of astronauts is often removed from the spacecraft by reaction with lithium hydroxide 2LiOH(s)+CO2(g)Li2CO3(s)+H2O(l) Estimate the grams of lithium hydroxide required per astronaut per day. Assume that each astronaut requires 2.50 103 kcal of energy per day. Further assume that this energy can be equated to the heat of combustion of a quantity of glucose, C6H12O6, to CO2(g) and H2O(l). From the amount of glucose required to give 2.50 103 kcal of heat, calculate the amount of CO2 produced and hence the amount of LiOH required. The H for glucose(s) is 1273 kJ/mol.Chlorine dioxide, ClO2, is a reddish yellow gas used in bleaching paper pulp. The average speed of a ClO2 molecule at 25C is 306 m/s. What is the kinetic energy (in joules) of a ClO2 molecule moving at this speed?
- 9.89 A sample of gas is 80.0% CH4 and 20.0% C2H6 by mass. What is the heat from the combustion of 1.00 g of this mixture? Assume the products are CO2 (g) and H2O (l).You have a chemical in a sealed glass container filled with air. The setup is sitting on a balance as shown below. The chemical is ignited by means of a magnifying glass focusing sunlight on the reactant. After the chemical has completely burned, which of the following is true? Explain your answer. a. The balance will read less than 250.0 g. b. The balance will read 250.0 g. c. The balance will read greater than 250.0 g. d. The scales reading cannot be determined without knowing the identity of the chemical.A _________ is a device used to determine the heat associated with a chemical reaction.











