Q: Select all that apply. Sight down the C-2-C-3 bond, and choose the Newman projection formulas for…
A: We need to consider the dihedral angle between the methyl substituents and the H atoms to decide on…
Q: Can you help me with the explanation of the binary covalent (molecular) compound, ionic compound and…
A: Introduction Chemical bonding is the process of forming bonds between two atoms or molecules in…
Q: Explain how soaps or surfactants decrease the surface tension of a liquid. How does the meniscus of…
A: Soaps & surfactants are compounds that lower the surface tension of liquids. They work by…
Q: An aromatic ring has how many units of unsaturation? 1 2 3 4 5
A: Introduction
Q: The following thermochemical equation is for the reaction of HCl(g) with NH3 (9) to form NH4Cl(s)…
A: This is exothermic reaction as ∆H of the reaction is negative. 1 mole ammonia react with 1 mole…
Q: A 73.0 kg patient being treated for leukemia is to receive an IV infusion of 5 cyclophosphamide over…
A: Given that, a 73 kg patient being treated for leukemia is to receive an IV infusion of 5 mg/kg…
Q: You make 20.0 g of a sucrose (C₁₂H₂2O11) and NaCl mixture and dissolve it in 1.00 kg water. The…
A: According to the question, Mass of sucrose and NaCl = 20.0 g Mass of water = 1.00 kg Freezing…
Q: Provide solutions for the following problem. Express- your final answer as indicated otherwise show…
A: (4) Given that - Volume of solution = 5.38 L = 5380 mL Mass of solute substance = 38.97 g…
Q: What is true of synthesis reactions? They must have more than one product. They must have two or…
A: we have to select the true statement for a synthesis reaction
Q: 5. How does temperature affect entropy?
A: To determine the effect of temperature on entropy
Q: Choose the resonance structure that results from the arrow pushing scheme below: H سفی مشهد مع 00 O-…
A: We have to determine the resonance structure of the given molecule
Q: What kind of intermolecular forces act between a hydrogen iodide molecule and a chloroacetylene…
A: Introduction Intermolecular forces are the forces of attraction or repulsion that act between…
Q: A pure sample of Substance S is put into an evacuated flask. The flask is then heated steadily and…
A:
Q: A certain liquid X has a normal freezing point of 4.10 °C and a freezing point depression constant…
A:
Q: hybridization states for each C and O atom in ketene -o atom -c atom 1 -c atom 2
A:
Q: Which of the following represent isotopes? 56 A: X 26 56 B: X 27 O A and D A and C O C and D B and C…
A: The given information is as follows: A: X2656 B: X2756 C: X2655…
Q: Thalidomide exists in two enantiomeric isomers as shown below. ((R) N۰۰۰ ((S) If a solution is…
A: Given that thalidomide exists in two enantiomeric isomers as shown below A solution is prepared by…
Q: How many peaks are present in the NMR signal of each labeled proton? a. b. H. C. d. H H Br
A: Given Different organic compound
Q: At a particular temperature, N₂O5 decomposes according to a first-order rate law with a half-life of…
A:
Q: Assuming equal concentrations and complete dissociation, arrange these aqueous solutions by their…
A: The depression in the freezing point of a solution can be calculated using the equation: ΔT = i…
Q: You need to use a dilute hydrochloric acid solution in an experiment. However, the only bottle of…
A: We have to calculate the pH of the solution
Q: Perform a confirmational analysis on 2-bromo-3-methylbutane.
A: Given 2-bromo-3-methylbutane
Q: 12. Consider the reaction 4PH3(g) →→→ P4(g) + 6H₂(g) - If, in a certain experiment, over a specific…
A: Given -> 4PH3(g) ---> P4(g) + 6H2(g) Moles of PH3 consumed = 0.0048 mole Volume= 2.0 L
Q: Convert the following from mg/L as CaCO3 to mg/L as the ion or compound a. 29.50 mg/L CO2…
A: Since you have posted a question with multiple sub-parts, we will solve the first three subparts for…
Q: What is the electron arrangement for the element that has two more protons than carbon?
A: Introduction Electron arrangement refers to the way in which electrons are arranged in an atom or…
Q: Standardization of Sodium Hydroxide (NaOH) Solution Mass of KHP used (g) Initial buret reading (mL)…
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: When assessing the relationship between the Quantity and Volume of a gas, what two factors must be…
A: Given Quantity and volume of gas
Q: Calculate the boiling point (T), in degrees Celsius, for a solution that contains 110.0 g of AICI,…
A:
Q: Draw mirror images for these molecules. Are they different from the original molecule? (a) • Use the…
A: Given molecule contains chiral carbon. chiral carbon = carbon atom with 4 different atoms or group…
Q: A. PHYSICAL PROPERTIES OF ELEMENTS AND COMPOUNDS Using the information obtained during the…
A: Element is pure form of substance consist only one type of atoms
Q: Calculate the molality of a 0.845 M aqueous solution of cadmium bromide with a density, p= 1.029…
A:
Q: Arrange the following solutes in the order of decreasing solubility in water: He, N₂, KI. N₂ N₂> >…
A: Solubility is a property of a substance that describes its ability to dissolve in a solvent to form…
Q: 3. Identify the hybridization and the approximate bond angles around each atom marked with an arrow…
A: Geometry shape is approximate arrangements of all bond pairs and loan pairs around the atom which…
Q: 애 CH₂OH H 애 -0 애 OH H In the box below, draw the open-chain structure (as a Fischer projection)…
A: To convert a Fischer projection to a Haworth projection, follow these steps: Locate the horizontal…
Q: The pressure in a vessel that contained pure oxygen gas dropped at a rate of 0.440 atm/min as the…
A:
Q: For each solute, click the button under the better solvent. solute CaCl₂ CH3(CH₂) CH3 4 1₂ Which is…
A:
Q: Consider the following formula, which of the following BEST identifies them: (a) CH₂O (b) CH3NO (c)…
A: Molecular formula represent the number of atoms present in a molecule while the structural formula…
Q: Select all of the true statements about melting points. a. The melting point range of a pure…
A: The melting point of a substance is the temperature at which it changes from a solid to a liquid…
Q: What is the correct IUPAC name of the following compound? H H CH₂CH3 CH₂ Multiple Choice…
A: Introduction IUPAC nomenclature is a system of naming chemical compounds and identifying the…
Q: Consider the following reaction at 298 K. C(graphite) + 2 H₂(g) → CH₂(g) AH' = -74.6 kJ Calculate…
A:
Q: Based on the concentration of Citric acid you calculate for 10 mL, is it safe to drink a 7-Up can…
A: Solution- Based on the concentration of Citric acid you calculate for 10 mL, Is it safe to drink a…
Q: A flask contains a mixture of two gases: NO2 and Ne. If the flask contains 0.518 mol of NO2and 16.7…
A:
Q: Calculate all the concentrations in equilibrium in a 0.4M solution of magnesium hydroxide magnesium…
A: We have find out the concentrations in equilibrium in a 0.4M solution of magnesium hydroxide.
Q: Una estructura resonante posible para la siguiente molécula es: a. b. C. NH₂ NH₂ +NH₂ + 1 :NH₂ :NH₂
A: In this question, we will identify the possibile resonance structure of the given compound. You can…
Q: A C 1.2 % NaCl 280 mOsm/L B D 5% Glucose 2% Glucose Assuming equal volume in all the sections, what…
A: Here it is given that the volume of all the sections are same. So A, B, C and D have se volume. We…
Q: A patient is suspected of having low stomach acid, a condition known as hypochloridia. To determine…
A: In the given reaction, the gastric juices contain hydrochloric acid(HCl). First, write down the…
Q: If I wanted to determine the enthalpy of a phase change for a substance, which seg- ments of the…
A: Enthalpy (H) is a measure of the total energy contained in a system, including both the internal…
Q: A student prepares a reaction in which hexane is converted to 1,3-hexadiene. In this reaction, what…
A: Please find your solution below : Oxidation is a type of chemical reaction in which the loss of…
Q: How many L NaOH of a 0.075 M NaOH solution is needed to completely neutralize 100.0 mL of a 0.250 M…
A: Given, volume of H2SO4 solution = 100.0 mL = 100.0 mL x ( 1 L / 1000 mL) = 0.100 L molarity of H2SO4…
Q: a) SO₂ gas at a pressure of 7.50 atm is heated from 0.0°C to 481 °C and is simultaneously compressed…
A:
Give typed answer only not written
Step by step
Solved in 2 steps with 2 images

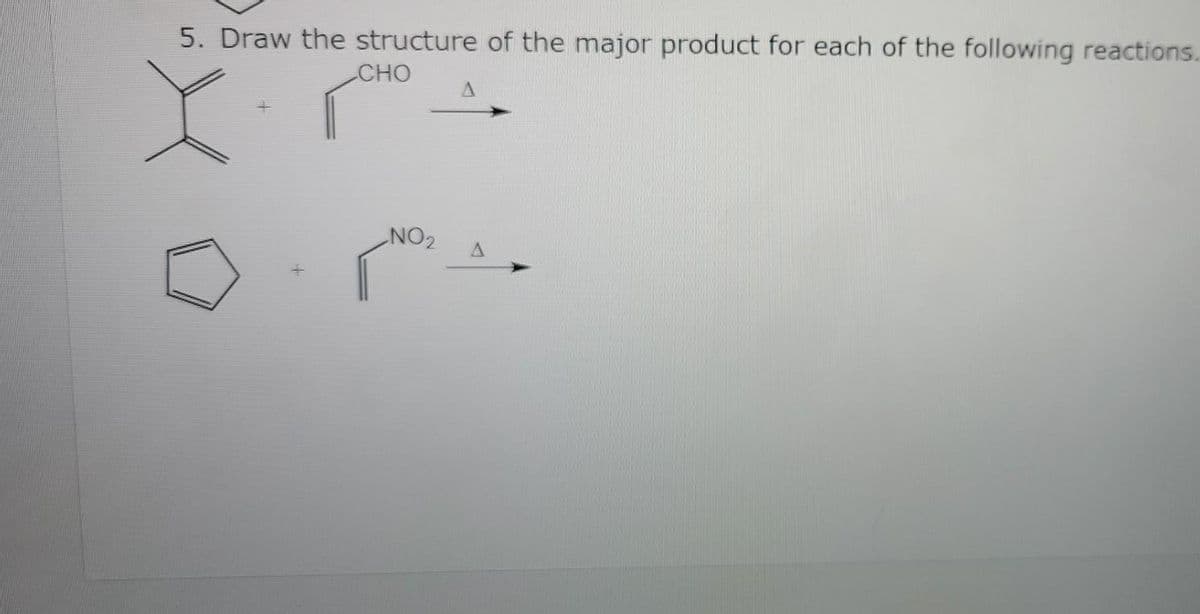
- Draw the products of the following reactions. Use curved arrows to show where the pair of electrons starts and where it ends up. a. ZnCl2 + CH3OH b. FeBr3 + Br c. AlCl3 + Cl−Fill in the missing reagents, conditions, intermediates or products in each box below.The major product formed by addition of HBr to (CH3)2C=CH– CH=C(CH3)2 is the same at low and high temperature. Draw the structure of the major product, and explain why the kinetic and thermodynamic products are the same in this reaction.
- Draw the products formed when each compound is treated with HNO3 and H2SO4.State whether the reaction occurs faster or slower than a similar reaction with benzene.Draw the major product formed when HBrHBr reacts with the epoxide. Use wedge–dash bonds, including hydrogen atoms at each stereogenic center, to show the stereochemistry of the product.Fill in any starting materials, reagents, or products to complete the reaction.



