Q: Following are 'H-NMR spectra for compounds G, H, and I, each with the molecular formula C,H,,0. Each…
A: a) The structure for given H-NMR spectrum are:
Q: Treatment of compound C (molecular formula C4H8O) with C6H5MgBr, followed by H2O, affords compound D…
A:
Q: Explain why the β carbon of an α,β-unsaturated carbonyl compound absorbs farther downeld in the 13C…
A: Generally, the more shielded C will have less chemical shift value and its corresponding signal will…
Q: The 'H and "C NMR spectra of compound A are shown below. In the mass spectrum the parent ion appears…
A:
Q: compound gives only one signal in its 1H-NMR spectrum. Propose a structural formula for compound.…
A: The given compound is C4H6. The first step is to calculate the number of double bond equivalence.
Q: How could 1H NMR spectroscopy be used to distinguish between each pair of compounds?
A: 1H NMR spectroscopy : It is a powerful technique to determine the exact structure of a molecule. In…
Q: 6. A compound with a molecular formula C,H12 has the following 'H NMR spectrum. Which of the…
A: 1H NMR spectroscopy is used to determine the structure of compound with respect to hydrogen nuclei…
Q: aqueous workup. Compound B gave a positive lodoform test. "H NMR spectrum of B is shown below. What…
A:
Q: Which of the diethylbenzene isomers (ortho, meta, or para) correspondsto each set of 13C NMR…
A: The given 13C NMR spectral data, 13C NMR signals: 16, 29, 128, and 141 ppm
Q: *18. Draw the structure of an ether with formula C5H12O2 that fits the following NMR spectrum:
A:
Q: Compound X (molecular formula C10H12O) was treated with NH2NH2, -OH to yield compound Y (molecular…
A: The molecular formula of X is C10H12O and the molecular formula of Y is C10H14. The 1H NMR peak of…
Q: An alkyl halide reacts with an alkoxide ion to form a compound whose 1H NMR spectrum is shown here.…
A:
Q: A compound reacts with methylmagnesium bromide followed by acidification to form the product with…
A:
Q: Reaction of unknown A with HCl forms chlorohydrin B as the major product. A shows no absorptions in…
A: a). Structure of A: The lack of IR absorption suggests the absence of CO bond and NH bond or OH…
Q: Give the structure that corresponds to the following molecular formula and 'H NMR spectrum:
A:
Q: Qs8. Tell precisely how you would use the H-NMR and IR spectroscopy to distinguish between the…
A: Propanal and propanone have the carbonyl group as the functional group. In the infrared spectrum,…
Q: Answer the following questions about each of the hydroxy ketones: 1-hydroxybutan-2-one (A) and…
A: Complete solution is given below in next step in jpeg format
Q: Isomer set #1: Love CH3OCCH2CH3 CH3ČOCH2CH3 a b Compound D, C,H§O2, shows a downfield singlet in its…
A: 1 H NMR spectroscopy.
Q: What compound with molecular formula C4HgO is consistent with the following 1H NMR spectrum? PPM
A:
Q: bllowing compound from the IR, 'H NMR and b) What information can you obtain from the IR spectrum?…
A: Given IR spectrum:
Q: Identify the structure of a minor product formed from the radical chlorination of propane, which has…
A: Figure 1
Q: Compound A has molecular formula C8H18. It shows one singlet in the ¹H-NMR spectrum. Identify A and…
A:
Q: (c) Formula: C3H6O2 Spectroscopy Reference 3H Í FPM 100
A:
Q: 11. (4) compound X has a molecular formula of C6H8O2, and gave two peaks in its C NMR spectrum but…
A: Identification of unknown organic compound
Q: A compound with a formula of C8H100 has the IR and NMR below. Propose a structure.
A: C8H10O DBE = 8 + 1 - 10/2 = 4
Q: Compound X (molecular formula C10H12O) was treated with NH2NH2, −OH to yield compound Y (molecular…
A: Given: Molecular formula of compound X= C10H12O Molecular formula of compound Y= C10H14
Q: Compound Q has the formula CaHOz. The 'H and "C NMR and IR spectra are shown 8. below. Propose a…
A: CALCULATION OF INDEX OF HYDROGEN DEFICIENCY Molecular formula = C4H8O2 Number of unsaturated bonds…
Q: Briefly explain how you might distinguish between the following substances by comparing their 'H NMR…
A: A question based on 1HNMR spectroscopy, which is to be accomplished.
Q: Methyl 2,2-dimethylpropanoate (CH3)3CCO2CH3 has two peaks in its 1H NMR spectrum. What are their…
A: The NMR spectrum provides the following details about the structure of the molecules: The number of…
Q: Briefly explain how you might distinguish between the following substances by comparing their 1H NMR…
A: 1. A nearby electronegative atom withdraws electron density from the neighbourhood of the proton ,so…
Q: A and B, isomers of molecular formula C3H5Cl3, are formed by the radical chlorination of a dihalide…
A: a. Compound A give two peaks, singlet at 2.23 ppm and singlet at 4.04 ppm. The molecular formula of…
Q: How can you use UV spectroscopy to distinguish between the compounds in each of the following pairs?
A: Concept introduction: Conjugation on λmax: The wavelength at which the pi to pi star transition…
Q: A compound of molecular formula C4H8O2 shows no IR peaks at 3600–3200 or 1700 cm-1. It exhibits one…
A: The given molecular formula of the compound is C4H8O2.The double bond equivalence is calculated by –
Q: 4. Draw the expected NMR spectrum for the following compound. a.
A: A. No. of signals obtained in 1H-NMR spectrum equals to the no. of different types of hydrogens…
Q: How can you use UV spectroscopy to distinguish between the compounds in each of the following pairs?
A: Conjugation on λmax: The wavelength at which the pi to pi star transition occurs increase as the…
Q: Cyclopropenyl chloride reacts with the Lewis acid aluminum chloride in nitromethane (solvent) at…
A: Highly stable conjugated cyclic compounds that do not give addition reactions and follow the Huckel…
Q: 5. A compound having molecular formula C,H,OS reacts with hydrazine hydrate forming a product C with…
A:
Q: Compound X (molecular formula C10H12O) was treated with NH2NH2,−OH to yield compound Y (molecular…
A: The molecular formula of X is C10H12O and the molecular formula of Y is C10H14.
Q: 3. How would you distinguish between each of the following pair of compounds by proton nmr? b)
A: NMR spectroscopy is a very important tool for determining the structure of the organic compound. The…
Q: The 1H NMR chemical shifts of nitromethane, dinitromethane, and trinitromethane are at δ 6.10, δ…
A:
Q: Use 'H NMR spectroscopy to distinguish between the following pairs of compounds. (8 points) а. and…
A:
Q: When a sample of the compound below is analyzed by mass spectrometry, the molecular ion will appear…
A: Mass spectrometry is used to find out the molecular weight of the compound. In this technique sample…
Q: Identify products A and B from the given 1H NMR data. Treatment of acetone [(CH3)2C=O] with dilute…
A: 1H NMR stands for proton nuclear magnetic resonance.
Q: Identify the structure of a minor product formed from the radical chlorination of propane, which has…
A: Figure 1
Q: A and B, isomers of molecular formula C3H5Cl3, are formed by the radical chlorination of a dihalide…
A: (a) Compound A give two peaks, singlet at 2.23 ppm and singlet at 4.04 ppm. The molecular formula of…
Q: Give the structure that corresponds to the following molecular formula and 'H NMR spectrum: C,H10: 8…
A:
Q: Reaction of unknown A with HCl forms chlorohydrin B as the major product. A shows no absorptions in…
A: (a) Structure of A: The lack of IR absorption suggests the absence of CO bond and NH bond or OH…
Q: An unknown compound X has the empirical formula C,HO and a molecular ion in its mass spectrum at…
A:
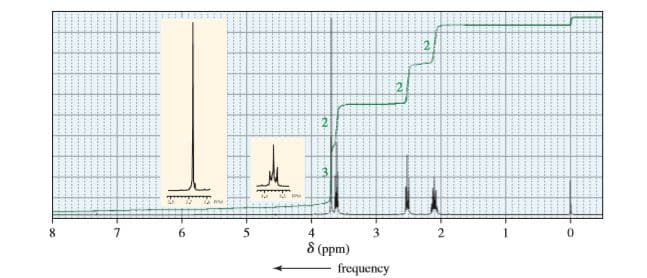
When treated with an equivalent of methanol, compound A, with molecular formula C4H6Cl2O, forms the compound whose 1H NMR spectrum is shown here. Identify compound A.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- What is the simplest CGS unit for "poise"?After Maria’s 8th cup of coffee one night during finals week, she remembered that high doses of caffeine can kill you. Maria was worried that she was more susceptible because she weighed only 47.7 kg. Maria did some research and found that 150 mg/kg of caffeine was lethal and the coffee she was drinking is 0.04% m/v caffeine. How many cups of coffee could Maria drink before she would likely die?Calculate the values of vrms, vmp, and vmean for Cl2 (70.90 g mol-1) at 25 °C
- What is the valency factor (n-factor) of sodium oxalate?pls help with the following question aspa and explain if possibleEugenol, the main component of clove oil, can be prepared by heating 2-allyl guaiacol (in the absence of oxygen) to temperatures >205 °C. In practice, however, it is most cost-effective to extract eugenol from cloves, its natural source. Given a weight percentage of eugenol in cloves at 15.0%, calculate the minimum mass of cloves one needs to obtain 8.00 g of eugenol as a product. Report your answer (as a mass in grams, without units) with three significant digits.
- In a metered-dose inhaler (MDI), such as those used for asthma medication, medicine isdelivered by a compressed-gas propellant. (The device is similar in concept to a can of spraypaint.) When the inhaler is activated, a fixed amount of the medicine suspended in thepropellant is expelled from the mouthpiece and inhaled. In the past, chlorofluorocarbons(CFCs) were used as propellants; however, because of their reactivity with the Earth's ozonelayer, they have been replaced by hydrofluorocarbons (HFCs), which do not react withozone. Now HFC use is also being reduced due to their high global warming potential. In one brand of inhalers, the original CFC propellant was replaced by HFC 227ea (C3HF7,heptafluoropropane). The volume of the inhaler propellant reservoir is 1.00×102 mL, and thepropellant is charged into the reservoir to a gauge pressure of 4.443 atm at 23°C. An onlinesearch for properties of HFC 227ea yields the information that the critical temperature andpressure of the substance…In a survey of 1000 large corporations, 250 said that, given a choice between a job candidate who smokes and an equally qualified nonsmoker, the nonsmoker would get the job (USA Today).(a) Let p represent the proportion of all corporations preferring a nonsmoking candidate. Find a point estimate for p.(b) Find a 0.95 confidence interval for p.(c) As a news writer, how would you report the survey results regarding the proportion of corporations that hire the equally qualified nonsmoker? What is the margin of error based on a 95% confidence interval?5. Permanganatometry is used to quantify?A) Iron (II) sulfateB) Zinc sulfateC) Silver nitrateD) Basic bismuth nitrateE) Copper sulfate
- Activity 1-1 part 4A dilute aqueous solution containing 1 ppm of solute has a density of 1.00 g/ mL. Express the concentration of solute in g/L, g/mL, and mg/ L. (Show work/calculations)A clinic had 25 patients on Friday morning. If 21 patientswere given flu shots, what percentage of the patientsreceived flu shots? Express your answer to the ones place.

