Q: Rank the alkenes shown in the ball-and-stick models (A–C) in order of increasing stability.
A: Solubility is decided by the following factors a. At the double bond it should be more alkylated…
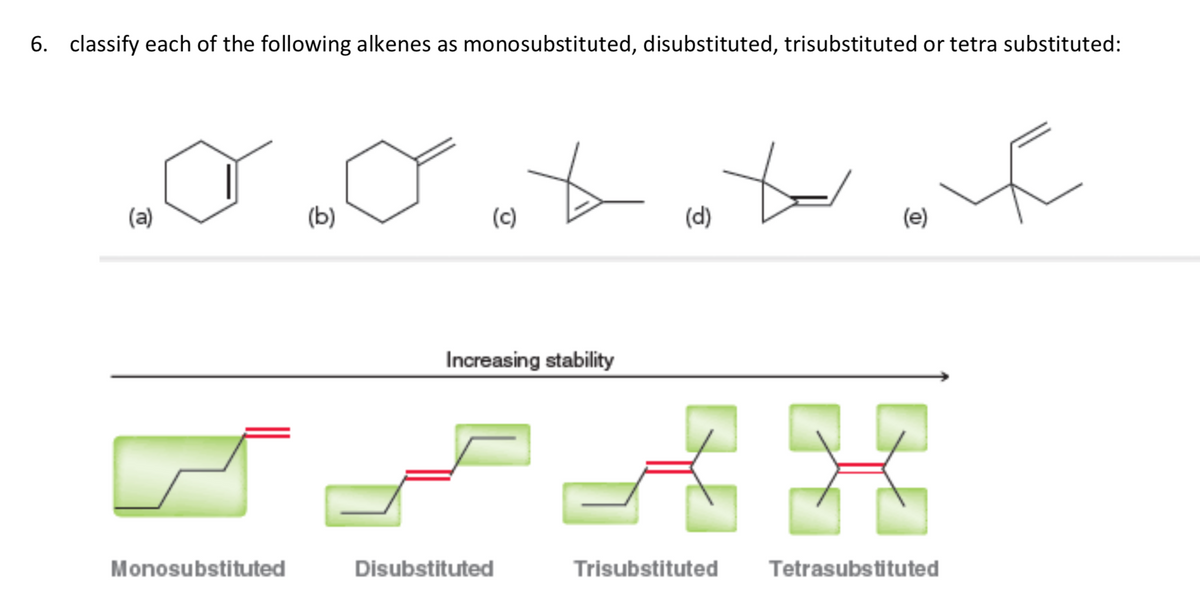
Q: Draw the six alkenes of molecular formula C5H10. Label one pair of diastereomers.
A: Alkenes are the hydrocarbon-containing carbon-carbon double bond. For example: CH3-CH=CH2propene ;…
Q: Which structure represents the most stable chair conformation of…
A: In 1, 3disubtuituted cyclohexane, (e,e) or (a,a) are cis-isomers and (e,a) or (e,a) are…
Q: Rank the alkenes shown in the ball-and-stick models (A–C) in order of increasing stability.
A:
Q: 6. Rank the alkenes in order of increasing stability. (1 = least stable, 4 = most stable) |
A: Stability of alkene ∝ Number of hyper conjugative structure ∝ Number…
Q: Draw a structural formula for the alkene with the molecular formula C5H10 that reacts with Br2 to…
A: The Bromination of alkene occurs through an intermediate bromonium ion. The compound 1,…
Q: Draw the structure of a 1°, 2°, and 3° carbocation, each having molecular formula C4H9+. Rank the…
A: The structure of a 1°, 2°, and 3° carbocation, each having molecular formula C4H9+ is drawn as,
Q: 6. What alkene would you start with if you wanted to prepare ethyl cyclohexane?
A: What alkene would you start with if you wanted to synthesize ethyl cyclohexane?
Q: Draw the structure of each alkene of molecular formula C7H14 that has a tetrasubstituted double bond
A: The structure of each alkene of molecular formula C7H14 that has a tetra substituted double bond has…
Q: Five isomeric alkanes (A–E) having the molecular formula C6H14 are each treated with Cl2 + hv to…
A: The isomers of C6H14 are as follows: A. Consider the treatment of 2-methylpentane with Cl2 + hv.…
Q: 1. Write the correct IUPAC names for the following organic compounds: а) Br CH3 H3C (Assign E or Z…
A: IUPAC nomenclature is used to name the organic compound.
Q: 3 Name each alkene and specify its configuration by the E,Z system.
A:
Q: Question 3 (a) Indicate the following alkenes in order of increasing stability. Justify your…
A: Alkenes are hydrocarbons having one or more double bonds.
Q: Explain why the addition of HBr to alkenes A and C is regioselective, forming addition products B…
A: Since in case of A, the product formed is because the stability of +ve charge will be more at the…
Q: 7. Which one of the following conformations of disubstituted cyclohexanes is expected to be the most…
A: Presence of one bulky methyl group at axial position will cause two gauche butane interaction. This…
Q: 8. For each of the following alkenes, assign the configuration of the double bond as either E or Z…
A: • When higher priority ower group present opposite sile in double then the…
Q: What bromo-substituted compound would be required to react with (CH2—CH)2CuLi in order to form each…
A:
Q: Draw a structural formula for the most stable carbocation with each molecular formula. Q.) C4H9+
A:
Q: Draw the constitutional isomer formed when the following alkenes are treated with each set of…
A: In this problem, we will discuss the formation of alcohol using terminal alkenes with two different…
Q: How many rings and π(pi) bonds are contained in compound A and draw one possible structure for this…
A:
Q: Five isomeric alkanes (A–E) having the molecular formula C6H14 are each treated with Cl2 + hν to…
A:
Q: Draw a structural formula for the alkene with the molecular formula C5H10 that reacts with Br2 to…
A: The halogenation reaction is the reaction which gives product with one or more halogens attached.…
Q: In assigning E or Z geometry, which two are the two high priority groups for the given alkene, and…
A: High priority groups are those, which have atoms of higher atomic number. We can determine the…
Q: Draw the products formed when both cis- and trans-but-2-ene are treated with a peroxyacid followed…
A:
Q: Draw a structural formula for the alkene with the molecular formula C5H10 that reacts with Br2 to…
A: In the addition reaction of bromine, the alkene react with Bromine solution in presence of…
Q: (b) (а)
A:
Q: 9. There are several possible forms of a trisubstituted cyclohexane with the formula C10H200. I have…
A: To identify the most and least commonly naturally occurring compound.
Q: 6. Answer the following questions regarding compound B. Compound B a. Draw two constitutional…
A: We have been provided with a ketone and we have to find its isomers. 1st isomer we need is an ether…
Q: Draw a structural formula for the most stable carbocation with each molecular formula. Q.) C3H7O+
A: Structural formula for the most stable carbocation with each molecular formula can be written as Q.)…
Q: CH3 CH3 (a) CH¿CH,C=CHCHCH3 + HBr → ?
A:
Q: Five isomeric alkanes (A–E) having the molecular formula C6H14 are each treated with Cl2 + hv to…
A: The isomers of C6H14 are as follows:
Q: Name each alkene and specify its configuration by the E,Z system
A: Name of the following compound : Identify the longest chain in the skeletal number it. Add the…
Q: Which pair of structures are constitutional isomers? (a) A and B (b) A and C (c) C and D (d) all are…
A: Note: According to our guidelines we are supposed to answer only one question. Kindly repost other…
Q: Part 2.2 Having identified the expected products for elimination of cis-1 we can now perform a…
A: Lower the energy of a conformer, higher is the stability. Hence we have to predict the energy of the…
Q: 7.1 Identify the allylic carbons in each of the following structures. (а) (b) H CH3 H.
A: An allyl group is a substituent with the structural formula H₂C=CH−CH₂R, where R is the rest of the…
Q: Choose the compound of molecular formula C7H13B1 that gives each alkene shown as the exclusive…
A: Alkyl halide on elimination gives alkene as final product.
Q: _CH=CH₂ _CH=CH₂ and CH3O 0₂N₁ A B Which will be true about the reaction of the two alkenes with HBr?
A: ->The reaction of alkene with HBr proceeds through formation of carbocation intermediate.Hence ,…
Q: 1) Which of the following statement(s) is(are) TRUE? a. Hydrogenation and combustion of alkenes are…
A: Exothermic reactions are those that give out heat during the reaction. For exothermic reactions,…
Q: 6. Provide a detailed, stepwise mechanism for the following transformation. Use the curved arrow…
A: Using deprotactation of carbonyl group by amine moiety by acidic hydrolysis.
Q: Complete the diagram by drawing a Newman projection of the product of this reaction in its most…
A:
Q: Draw the constitutional isomer formed when the following alkenes are treated with each set of…
A: a) Hydroboration followed by oxidation of methylenecyclopentane to give cyclopentylmethanol.…
Q: What bromo-substituted compound would be required to react with (CH2 = CH)2CuLi in order to form…
A: In order to get the product by using Bromo substituted compound, delete the (CH2 = CH2) group and…
Q: 1. Which has a lower boiling point cis-pent-2-ene or trans-pent-2-ene? Why?
A: Since, you have asked multiple questions, I solved first question for you with details explanation.…
Q: Rank by the stability of the alkene isomers. The most stable isomer is 1, while the least stable…
A: The stability of the alkene can be done on the basis of the number of groups attached to the carbon…
Q: What alkene with the molecular formula C6H12, when treated with ozone and then dimethyl sulfide,…
A: The alkene with molecular formula C6H12 when treated with ozone and dimethyl sulfide has to be…
Q: Classify each of the following alkenes as electron poor or electron rich, relative to ethene.…
A: electron poor alkene are those which have electron donor groups attached at alpha positions and…
Q: 9. For the following compound: CI a) Is the alkene an E or Z stereoisomer? b) Provide the IUPAC name…
A: The alkene given is as shown below.
Q: Part 2.2 Having identified the expected products for elimination of cis-1 we can now perform a…
A: Cyclohexane Close ring exist in 2 conformation. Axial And equtorial conformation. Equtorial…
Q: Wyerone, a compound found in broad beans, has antifungal properties. Wyerone Identify a trans and a…
A:
Can i get help with these problems
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

- Five isomeric alkanes (A–E) having the molecular formula C6H14 are each treated with Cl2 + hv to give alkyl halides having molecular formulaC6H13Cl. A yields five constitutional isomers. B yields four constitutionalisomers. C yields two constitutional isomers. D yields threeconstitutional isomers, two of which possess stereogenic centers. Eyields three constitutional isomers, only one of which possesses astereogenic center. Identify the structures of A–E.A chiral alkyne A with molecular formula C6H10 is reduced with H2 and Lindlar catalyst to B having the R conguration at its stereogenic center. What are the structures of A and B?Draw the structure(s) of all of the alkene isomers, C5H10, that contain a branched chain. Consider E/Z stereochemistry of alkenes.
- Compounds A and B are isomers having molecular formula C5H12. Heating A with Cl2 gives a single product of monohalogenation, whereas heating B under the same conditions forms three constitutional isomers. What are thestructures of A and B?Draw the most stable conformation of the disubstituted cyclohexane below.How many rings and π bonds are contained in a compound of molecularformula C8H12 that is hydrogenated to a compound of molecular formulaC8H14?
- The compounds drawn should each contain a cyclohexane ring. For all three compounds draw a wedge and dash structure, Chair I, and Chair II conformations. Formula: C9H18 with substitution 1,1- disubstituted with stereochemistry of (R,S) Formula: C7H13Cl with substitution 1,3- disubstituted with stereochemistry of (R,R) Formula: C7H14O with substitution 1,4- disubstituted with stereochemistry (S,S)Bogorol A is a natural product with the potential to fight antibiotic-resistant bacteria. Shown below is an intermediate that was used in a synthesis of bogorol A. Assign the configuration of the alkene unit as either Z or E. a. Z b. EIs the following alkene E,Z, or neither? a. E b. Z c. neither d. It is one or the other, but which is impossible to determine from the information given
- Five isomeric alkanes (A–E) having the molecular formula C6H14 are each treated with Cl2 + hv to give alkyl halides having molecular formula C6H13Cl. A yields five constitutional isomers. B yields four constitutional isomers. C yields two constitutional isomers. D yields three constitutional isomers, two of which possess stereogenic centers. E yields three constitutional isomers, only one of which possesses a stereogenic center. Identify the structures of A–E.1. Estimate the relative stabilities of the 4 conformationseclipsed and eclipsed n-pentane by drawing the corresponding Newman projections in descending order according to their stability. Place the most stable or lowest energy on the left. 2. Explain what the difference in energy between the most stable and the least stable is due to. 3. What is the difference in stability between the two staggered conformations?Which of the following alkene reactions occurs with a complete cleavage of the C=C ( breaking both pi and sigma bond components) ? Select one: A. Alkene + H2O2 with catalytic amount of OsO4 B. Reaction of an alkene with KMnO4/OH- at room temperature. C. Treating the alkene with O3 gas, followed by reduction with Zn / H2O D. Reaction of an alkene with water (H2O) in the presence of a H+ catalyst.

