Q: Examine Solved Problem 7.3. Your task is to prepare A in the highest possible yield by…
A: Interpretation: To prepare A i.e 2-methyl-1-butene by dehydrobromination in highest possible yield…
Q: Which arene is most likely to produce a major product with bromine meta to the substituent upon…
A: Multiple question.hence,answer of 1 question:- option c give major product with bromine which…
Q: C NasH Hgot
A:
Q: Provide the Cruved arrow mechanism of anisole treated with bromine with iron (III) bromide. Include…
A: Here we are required to predict the mechanism of anisole with bromine with FeBr3.
Q: Predict the major products of each of the following reactions, a, b.
A: From the conformational analysis, it is cleared that 2 is more stable but it is most deactivated…
Q: The compound shown here is highly unreactive under conditions that favor E2 reactions. Explain why.…
A: Reactions can have either substitution type of mechanism or an elimination type of mechanism. In a…
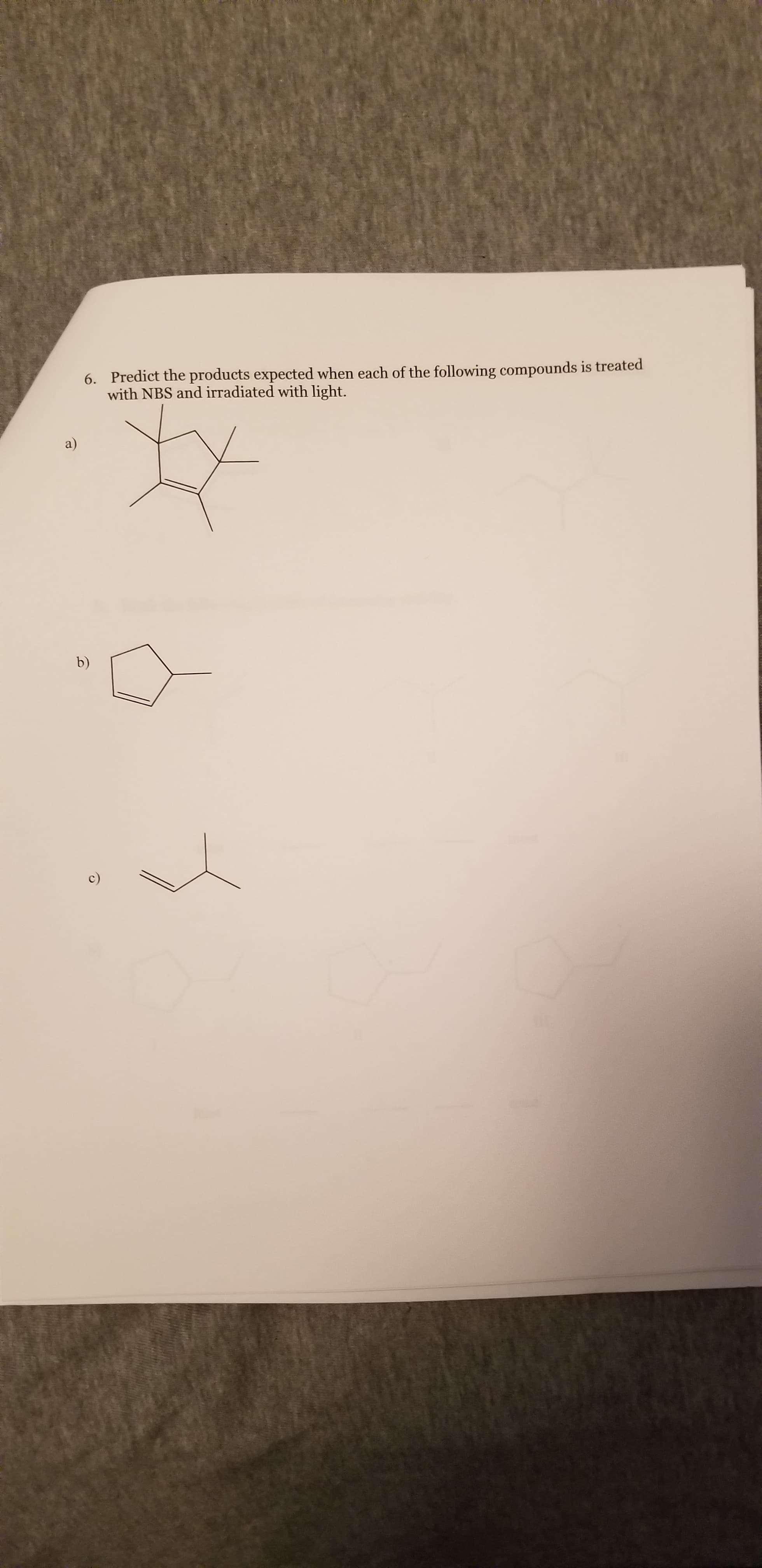
Q: Bromination of an alkene by N‑bromosuccinimide (NBS)(NBS) in the presence of light or peroxide is a…
A: In the given molecule, there are 2 allylic carbons, hence more than 1 product will be formed.
Q: Intramolecular Diels–Alder reactions are possible when a substrate contains both a 1,3-diene and a…
A: Intramolecular Diels–Alder reactions are possible when a substrate contains both a 1,3-diene and a…
Q: Draw the major organic product generated in the reaction below. Pay particular attention to regio-…
A: Regio-chemical means make or formed of chemical bond in one direction. Stereochemical means mixture…
Q: a) Aspirin Exp: H2SO4 HO.
A: A question based on IR spectroscopy that is to be accomplished.
Q: Write out a detailed mechanism (using curved arrow formalism) for the Friedel-Crafts alkylation of…
A:
Q: (a) Consider the reaction below, and answer the questions that follow: UV ight (i) Draw a curved…
A: The reaction sequence given in the question represents a uv mediated ring closing reaction of a…
Q: Part II Draw the structure of the synthesized product (do not include side or by products) from…
A: The given reaction is the acid catalysed esterification reaction between carboxylic acid and…
Q: Answer part a, b and c part a in image attached b) name the class of compound to which the…
A: The solution is as follows:
Q: Draw all constitutional isomers formed in attached E2 reaction, and predict the major product using…
A: The regioselectivity of E1 and E2 elimination reactions is predicted using Zaitsev’s rule. According…
Q: ) Propose a detailed curved-arrow. mehunim for the following transformetion.
A: Answer
Q: When naphthalene undergoes an irreversible electrophilic aromatic substitution, such as a…
A: Alpha position is more stable than beta position because
Q: Pentalene is most elusive molecule and has never been isolated. The pentalene dianion, however, is…
A: Aromatic Compounds are the compounds which are planar, cyclic, conjugation of π electrons and has to…
Q: Predict the favored product of each of the following reduction reactions. If the product is chiral,…
A: The reactions given are,
Q: II
A: The given reaction is an example of hyfrogenation reaction.
Q: 6. Draw the curved arrow mechanism(s) for the following transformation. Be sure to rationalize the…
A: Given is hydrochlorination reaction.
Q: Draw all constitutional isomers formed in attached elimination reaction.Label the mechanism as E2 or…
A: The base and the type of alkyl halide in a given reaction determine the mechanism. In the given…
Q: Select the member of each pair that undergoes SN1 solvolysis in aqueous ethanol more rapidly
A: In SN1, the rate determining step is formation of carbocation and order of the substrate that…
Q: What compound undergoes metathesis to form each of the following compounds?
A: First break the double bond present in product made after metathesis reaction and add =CH2 to each…
Q: a. Which of the following compounds cannot be prepared by a Heck reaction?b. For those compounds…
A: Introduction: Heck reaction: Heck reaction is used to synthesize the substituted alkene. It consist…
Q: How many singlets in will be observed in the ¹H NMR spectrum of this compound? od
A: NMR spectroscopy is nuclear magnetic resonance spectroscopy
Q: please explain clearly and in detail. thank you! 2-butene reacts with NBS yields and forms two…
A:
Q: Draw all constitutional isomers formed in attached E2 reaction, and predict the major product using…
A:
Q: One of the two diastereomers shown below reacts with potassium tertbutoxide significantly faster…
A:
Q: What compound undergoes metathesis to form each of the following compounds?
A:
Q: H2SO4 Он
A:
Q: C. Using any necessary reagents of 2 carbons or less, and any other necessary inorganic reagents,…
A: The synthesis of the given compound is given below.
Q: 7. Propose an efficient synthesis of for each of the following transformation: a) b)
A:
Q: 2.4. Draw the structures of the product(s) described for each reaction. Specify all aspects of the…
A:
Q: Draw all constitutional isomers formed in attached E2 reaction, and predict the major product using…
A: Given:
Q: The following Grignard reagents are desired: a) cyclohexylmagnesium bromide b) sec-butylmagnesium…
A: Grignard reagent falls under organometallic compounds and has magnesium bonded with halide or…
Q: In contrast, optically active butan-2-ol does not racemize on treatment with a solution of KOH.…
A: Optically active 2-butanol on reaction with KOH gives alkene. Hence; racemic product is not…
Q: Draw the organic product of the following nucleophilic substitution reaction. (Assume that only…
A: Given reaction,
Q: In each of the following compounds, which H would most likely be abstracted by a bromine radical,…
A: In the first step of a free radical reaction is the generation of free radical, then propagation and…
Q: Intramolecular Diels-Alder reactions are possible when a substrate contains both a 1,3-diene and a…
A:
Q: With this in mind, draw the product when each compound undergoes anintramolecular Diels–Alder…
A: Diels Alder reaction is [4+2] cycloaddition reaction. It is reaction between diene and dienophile…
Q: You have learned two ways to make an alkyl benzene: Friedel–Crafts alkylation, and Friedel–Crafts…
A: The general mechanism for Friedel-craft’s alkylation is given below\ where R is alkyl group
Q: Provide a reasonable stepwise mechanism for the following transformation. Show the structure of any…
A: Solution: We know all kinds are are electron reached due to buy bonds so they can act as nucleophile…
Q: Explain, with reference to the mechanism, why addition of one equivalent of HCl to diene A forms…
A: The given reaction is, Here, conjugated dienes undergo electrophilic addition to gives a mixture of…
Q: Draw the reaction of a conjugated diene with either HX (HCI, HBr, or HI), X2 (C12, Br2), or with a…
A:
Q: Provide the curved arrow mechanism of m-xylene treated with isopropylchloride with iron (III)…
A:
Q: 17. Provide a curved arrow mechanism for the following transformation. Br NaN, DMSO
A: As shown in the reaction, the rection follows SN2 pathway because the product is inversion in…
Q: Name the type of reaction and provide a possible reagent assign all the stereocentres found in the…
A: Compound C contains two functional groups ; Carbonyl /ketone and ether. Compounds D, E, F and G…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Name the type of reaction and provide a possible reagent assign all the stereocentres found in the compounds via cahn ingold prelog rules define the relation between compound. D E F G identify the 2 stereoisomers likely to be formed in compound cProvide a reasonable stepwise mechanism for the following transformation. Show the structure of any intermediates and use the arrow formalism to account for electron flow. Draw the structure of the regioisomer that might form and suggest a reason why it would not be produced.Consider compound I below, which is structurally related to a natural product that was isolated from an extract of a Caribbean marine sponge (See J. Chem. Soc. 1994, 116, 6015). Answer the following questions about this compound (please explain answers) How many double bonds with E stereochemistry are present? _________ How many double bonds with Z stereochemistry are present? _________
- Draw the product of this reaction and states its IUPAC name, and states what type of mechanism is occuring (eg. SN1, SN2, E1, E2, etc)?The reaction of cyclohexene with iodobenzene under Heck conditions forms E, a coupling product with the new phenyl group on the allylic carbon, but none of the “expected” coupling product F with the phenyl group bonded directly to the carbon–carbon double bond. Draw a stepwise mechanism that illustrates how E is formed.(A) Provide the major organic product for the reaction below (B) Would the product be optically active of optically in active?
- Provide the curved arrow mechanism of m-xylene treated with isopropylchloride with iron (III) chloride. Please include the resonance forms and major product. Thank you so much for the help!Draw all constitutional isomers formed in attached E2 reaction, and predict the major product using the Zaitsev rule.The following compound readily eliminates CO; to form a conjugated six membered ring. (a) Complete the reaction with drawing the possible structure and (b) explain why this reaction goes steadily.
- When naphthalene undergoes an irreversible electrophilic aromatic substitution, such as a Friedel–Crafts acylation, the major product is the kinetic product,which proceeds through the most stable arenium ion intermediate. InSection 23.7, we mentioned that substitution is generally favored at the αposition over the β position, which means that the arenium ion is more stable when the electrophile attaches to the α position. Explain this difference in arenium ion stabilities. Hint: Draw out allresonance structures for each arenium ion intermediate. Does each one have the same number of resonancestructures? How many resonance structures of each intermediate preserve the aromaticity?Draw a detailed mechanism for the FeBr3@catalyzed reaction of ethylbenzene with bromine, and show why the sigma complex (and the transition state leading to it) is lower in energy for substitution at the ortho and para positions than it is for substitution at the meta position.Identify compounds G and H in the following reaction scheme. H represents the structure of stemoamide, the chapter-opening molecule.



