Q: Avobenzone is a conjugated compound that absorbs ultraviolet light with wavelengths in the 320–400…
A: The structure of Avobenzone is - The retrosynthetic analysis of avobenzone are shown below -
Q: One step in the synthesis of the antihistamine fexofenadine (Section 23.5) involves acid-catalyzed…
A: Given Structure: Reaction of A in presence of acid-calatysed hydration of H+ Following products are…
Q: 1. Rank the following alkenes in order of increased stability. 2 5
A:
Q: Select all the possible enols for the following compound? (Colaet عديد LDL B C A
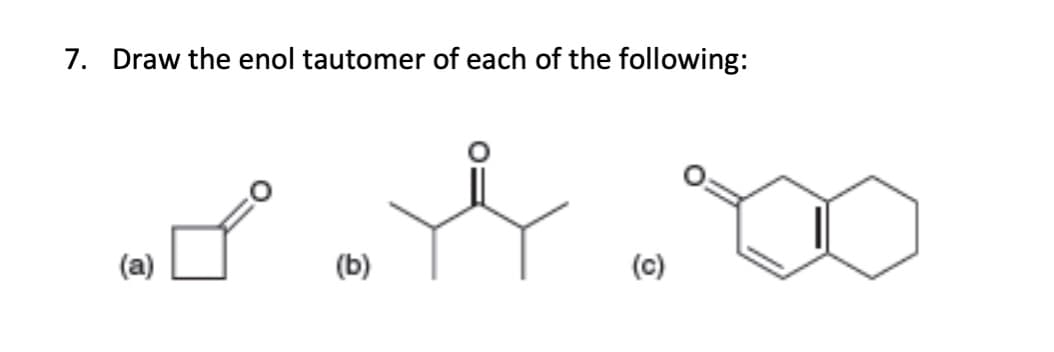
A:
Q: 8.46 Draw the structure (including stereochemistry) of an alkyl chloride forms each alkene as the…
A:
Q: Compound A is an alkene that was treated with ozone (followed by DMS) to yield only (CH3CH2CH2)¿C=O.…
A: Since only 1 compound is formed after ozonolysis, the alkene must be symmetric with two CH3CH2CH2-…
Q: c) Which of the following bicyclic ring system can be prepared by an intermolecular Robinson…
A: Organic reactions are chemical reactions which is involving in the organic compounds and the…
Q: What product is formed by the [3,3] sigmatropic rearrangement of each compound? a. b.
A: Given compounds,
Q: What product is formed by the [3,3] sigmatropic rearrangement of eachcompound?
A:
Q: Leptospermone is a herbicide produced by the bottlebrush plant. Draw all possible mono enol…
A:
Q: Draw a stepwise mechanism for the following reaction and explain the stereochemistry of the product.
A: The protonation step is shown below.
Q: What carbonyl compound and what phosphonium ylide are needed to synthesize the Attached compound?
A:
Q: 1. What will be the major product for the following reaction according to the Markovnikov's rule?…
A: Note : As per our guidelines ,we are supposed to answer first question , Please resubmit the other…
Q: 5.45 Use resonance structures of the possible carbocation intermediates to explain why bromination…
A:
Q: 7. Arrange each of the isomeric alkenes in order of stability: (a) (Ь)
A:
Q: Rank each of the enolates from least to most stabilized with 1 being the least and four being the…
A: The ions given are,
Q: 6. Provide the line structure of product F in the following reaction. Include stereochemistry for…
A: Due to presence of double bond alkene are reactive
Q: What alkyl halide reacts with lithium divinylcuprate 3(CH2“CH)2CuLi4 for the synthesis of each of…
A: The molecular formula of lithium divinylcuprate = C4H6CuLi (a) When bromocyclohexane is treated with…
Q: Draw the starting structure that would lead to this major product (and its enantiomer) under these…
A: We have to draw the starting structure for the following given product (and its enantiomer) as…
Q: (a) What product is formed by the Claisen rearrangement of compound Z? (b) Using what you have…
A:
Q: Treatment of ketone A with LDA followed by CH3CH2I did not form the desired alkylation product B.…
A: The product formed in the reaction is shown below.
Q: Which of the following pairs are keto–enol tautomers? a. CH3CH2CH=CHCH2OH and CH3CH2CH2CH2CH=O b.…
A: Since we know that for compound to be keto-enol pair, the OH should be connected to the Carbon which…
Q: 3. What is of the following compounds will produce a mixture of constitutional isomers when treated…
A: Unsymmetrically disubstituted alkene/alkyne upon reaction with HBr give a mixture of constitutional…
Q: c) d)
A:
Q: Identify the reagents necessary to achieve each of the following transformations: он он (a) (b) (c)
A:
Q: What compound undergoes metathesis to form each of the following compounds?
A: First break the double bond present in product made after metathesis reaction and add =CH2 to each…
Q: When a carbonyl compound is attacked by a nucleophile resulting in the formation of a new…
A: In the carbonyl compound nucleophilic addition reaction occurs.
Q: 7.1 Identify the allylic carbons in each of the following structures. (а) (b) H CH3 H.
A: An allyl group is a substituent with the structural formula H₂C=CH−CH₂R, where R is the rest of the…
Q: a) Consider the following transformations. CHCH3 CH3MGB (CH3)2NH Reagent I в A H3O* CH3 'N' CH3…
A: 1. In presence of ch3mgbr , nucleophilic attack occurs on carbonyl center which gives alcohol as a…
Q: What neutral nucleophile is needed to convert C to D? Imatinib, an effective treatment for certain…
A: Since in C and D, the difference is of and Cl part hence it means has attacked on the molecule and…
Q: Which carbonyl compound in each pair exhibits the higher percentage ofthe enol tautomer?
A: Enol is truly based on it name en-ol. C=C double bond and an OH group attached to it.
Q: 13. Draw the concerted mechanism for the following elimination. CH3O CI
A: In these reaction 2-chlorobutane react with strong base CH3O- to give but-2-ene. it follows…
Q: 7) Provide a synthesis of the following compounds using the given starting material and any other…
A:
Q: Which stereoisomer is obtained in the following intramolecular reaction? BrMg Br a. b. d. C. **** I,
A:
Q: Draw the product of the [3,3] sigmatropic rearrangement of each compound.
A: 3,3 sigmatropic rearrangement involves pericyclic reaction that proceeds via Huckel topology…
Q: Rank the following compounds in order of increasing reactivity in nucleophilic addition.
A: Given compounds,
Q: 3. Deduce the structures of A, B and C and reagent(s) D in reaction scheme shown below. What is the…
A: 1. Given substrate is an alkyl chloride (2-Chloro butane) with bulkier base like tertiary butoxide…
Q: Which carbonyl compound in each pair exhibits the higher percentage of the enol tautomer?
A:
Q: (a) What product is formed by the Claisen rearrangement of compound Z? (b) Using what you have…
A: Introduction: Claisen rearrangement is one of the most important reaction for the formation carbon…
Q: Heating A results in two successive [3,3] sigmatropic rearrangements–Claisen reaction followed by…
A: Claisen rearrangement is an organic chemical reaction that offers a powerful method in the formation…
Q: 1. Predicting Products/starting materials: Draw the structure of the major organie each reaction in…
A: The reaction 1) ketone react with Griganard reagent produced alchol. In reaction 2) primary alkayl…
Q: Which of the following undergoes a substitution reaction with sodium cyanide in DMSO at the fastest…
A:
Q: 15. Draw a detailed arrow-pushing mechanism for the following transformation, in MEOH ÖH ÖMe MeOH2…
A:
Q: Explain why two different products are formed from disrotatory ring closure of…
A: Trans isomer is formed on disrotatory ring closure of (2E,4Z,6Z)-octatriene, and this trans isomer…
Q: (R)-Carvone, the major component of the oil of spearmint, undergoes acid-catalyzed isomerization to…
A: The protonation of (R)- carvone is show below.
Q: Draw a stepwise mechanism for the following reaction, a key step in the synthesis of vernakalant, a…
A: The reactions involving organic compounds are called organic compounds. Organic reactions are…
Q: CI compound 1 CI ………I H compound 2 O a. Compound 1 is more reactive than compound 2 in both…
A: If a reaction centre is bridge head carbon then neither undergo substitution nor elimination
Q: Vitamin C is a stable enediol. Draw the structure of the two ketotautomers in equilibrium with the…
A: The equilibrium exist between carbonyl group and enol is known as Keto-enol tautomerism.
Q: 9. Explain in detail and with drawings why the following compound cannot make an E2 reaction when…
A: For E2 - elemination reaction compound must have anti-periplaner hydrogen.
Q: 4. Identify the position where electrophilic aromatic substitution is most favorable. * HN' CH3 CH3…
A: EWGs are meta directing whereas EDGs are ortho para directing.
please show all the steps.
thank you
Trending now
This is a popular solution!
Step by step
Solved in 5 steps with 6 images

- Identify the intermediates (A–C) in the following reaction sequence, which was used to prepare racemic ooxacin. One enantiomer of the product, levooxacin, is an antibiotic used to treat severe bacterial infections that have not responded to other drugs.(R)-Carvone, the major component of the oil of spearmint, undergoesacid-catalyzed isomerization to carvacrol, a major component of the oilof thyme. Draw a stepwise mechanism and explain why thisisomerization occurs.Draw all constitutional isomers formed in each E2 reaction, and predict the major product using the Zaitsev rule.
- What alkene is required to synthesize each of the following compounds? b. What other epoxide is formed in each synthesis? c. Assign an R or S configuration to each asymmetric center.Draw a stepwise mechanism for the following reaction, a key step in the synthesis of vernakalant, a drug approved in Europe in 2010 for the treatment of atrial fibrillation. Pure B was separated from a mixture of diastereomers. Your mechanism must explain the trans stereochemistry of the two substituents on the six-membered ring.One step in the synthesis of the antihistamine fexofenadine (Section 23.5) involves acid-catalyzed hydration of the triple bond in A. Draw a stepwise mechanism for this reaction and explain why only ketone B is formed.
- (a) What product is formed by the Claisen rearrangement of compound Z? (b) Using what you have learned about ring-closing metathesis in Chapter 24, draw the product formed when the product in part (a) is treated with Grubbs catalyst. These two reactions are key steps in the synthesis of garsubellin A, a biologically active natural product that stimulates the synthesis of the neurotransmitter acetylcholine. Compounds of this sort may prove to be useful drugs for the treatment of neurodegenerative diseases such as Alzheimer's disease.Identify the missing reagents a-f in the following scheme:Identify the reagents represented by the letters a-e in the following scheme:
- Provide the missing starting material, reactant or product. Show appropriate stereochemistry. a) b)Draw the major product (with stereochemistry) of the following reaction. Draw only one enantiomer for a racemic mixture.Addition of HCl to alkene X forms two alkyl halides Y and Z. (A) Label Y and Z as a kinetic or thermodynamic product and explain why. (B) Explain why the addition of HCl occurs at the exocyclic C=C, rather than the other C=C

