73: Consider the equation for the combustion of acetone (C3H6O), the main ingredient in nail polish remover: C3H6O) + 402(g) -> 3CO2(g) + 3H2O(g) dHrxn = -1790 kJ. If a bottle of nail polish remover contains 155 g of acetone, how much heat is released by its complete %3D combustion?
73: Consider the equation for the combustion of acetone (C3H6O), the main ingredient in nail polish remover: C3H6O) + 402(g) -> 3CO2(g) + 3H2O(g) dHrxn = -1790 kJ. If a bottle of nail polish remover contains 155 g of acetone, how much heat is released by its complete %3D combustion?
Chapter10: Energy
Section: Chapter Questions
Problem 61A
Related questions
Question
These are my 10 chemistry questions I'd like help with and the answers to them!
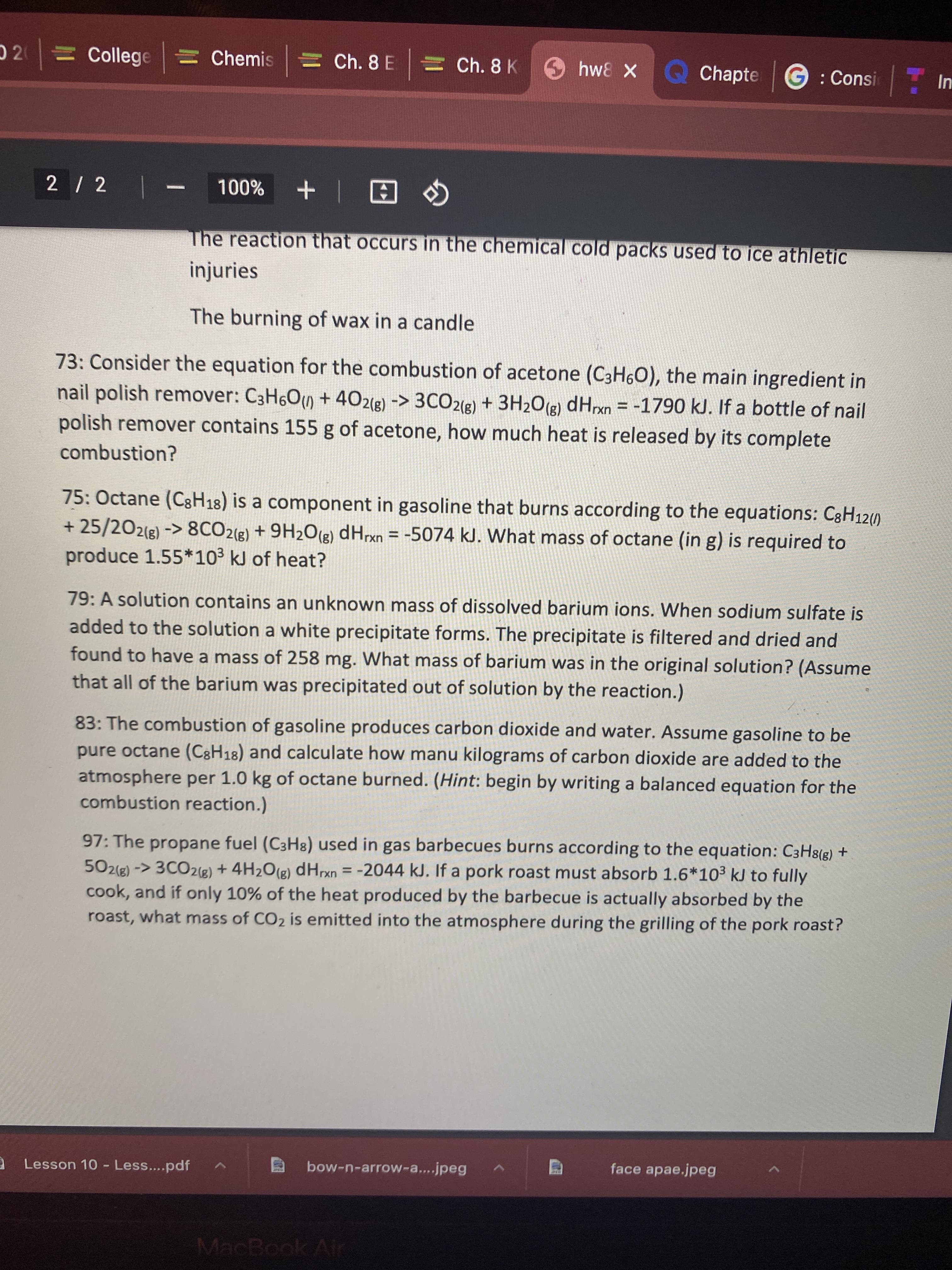
Transcribed Image Text:73: Consider the equation for the combustion of acetone (C3H6O), the main ingredient in
nail polish remover: C3H6O) + 402(g) -> 3CO2(g) + 3H2O(g) dHrxn = -1790 kJ. If a bottle of nail
polish remover contains 155 g of acetone, how much heat is released by its complete
%3D
combustion?

Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

World of Chemistry
Chemistry
ISBN:
9780618562763
Author:
Steven S. Zumdahl
Publisher:
Houghton Mifflin College Div

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

World of Chemistry
Chemistry
ISBN:
9780618562763
Author:
Steven S. Zumdahl
Publisher:
Houghton Mifflin College Div

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning
