Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.10QAP
Related questions
Question
What will be in the empty box in the picture below?
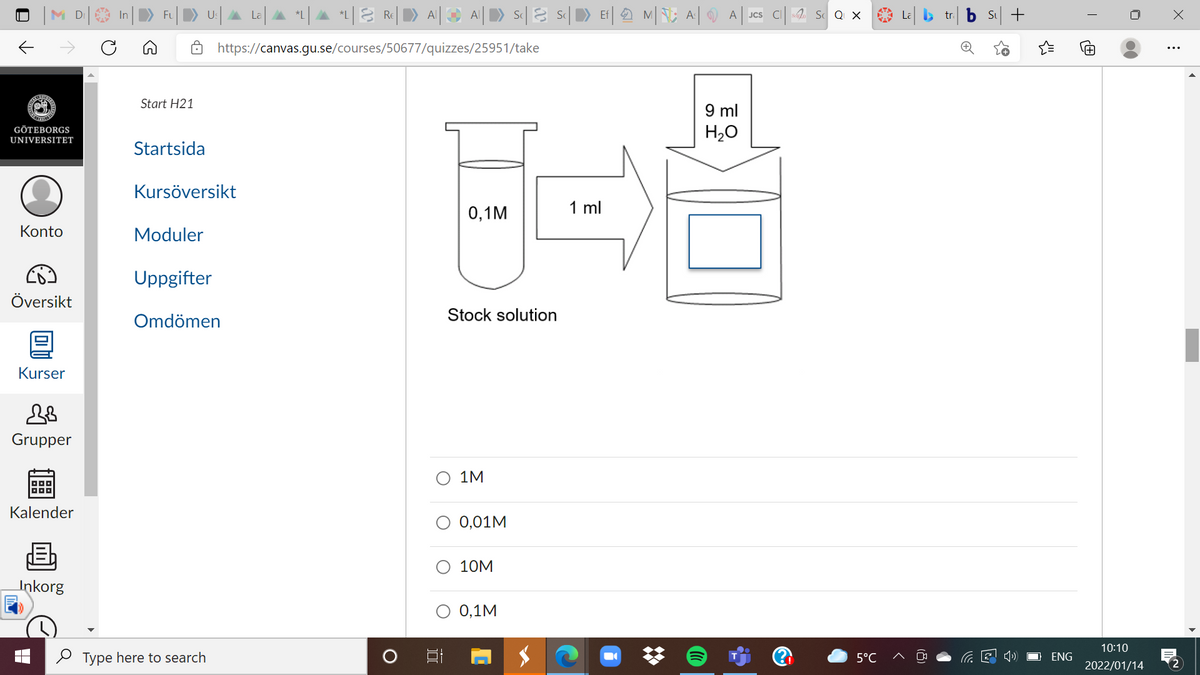
Transcribed Image Text:M D In
|) s«|8 s«) Ei| 2 M| A|0 A| scs C| -2 s. Q x La
Si +
Fu
tr
https://canvas.gu.se/courses/50677/quizzes/25951/take
Start H21
9 ml
H2O
GÖTEBORGS
UNIVERSITET
Startsida
Kursöversikt
0,1M
1 ml
Konto
Moduler
Uppgifter
Översikt
Omdömen
Stock solution
Kurser
Grupper
1M
Kalender
0,01M
画
10M
Inkorg
0,1M
10:10
2 Type here to search
5°C
O ENG
2022/01/14
2
(8)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning