Q: 2. Compound P, C4H9Br reacts with aqueous solution of sodium hydroxide to form compound Q, C4H10O. C...
A: Organic compounds are of different types. They are classified into saturated, unsaturated and aroma...
Q: Discuss x-ray diffraction method as a method of determining the crystal structures
A: Diffraction means bending of waves around corner of obstacle or through an aperture in the regio...
Q: Topic # 1: List the three requirements for a substitution reaction to proceed via the SN1 mechanism....
A: Hi! Thank you for the question, As per the honor code, we are allowed to answer one question at a ti...
Q: At a pressure of 1 atm, kJ of heat are needed to vaporize a 38.7 g sample of liquid benzene at its n...
A:
Q: Osmosis is the process of water molecules passing through a semipermeable membrane. If a semi-permip...
A: The correct answer is C
Q: Chemistry Question
A: The reaction is carried by Sandmeyer's reaction Diazotisation of aniline followed by cuprous bromid...
Q: Q4: 2M of methanol (CH,OH) was added to NaCl to make a solution, the mole fraction of methanol was 0...
A: In question, Following information given - Molarity of methanol = 2 M Mole fraction of methanol = 0....
Q: Consider two redox half reactions, one in which glucose is oxidized (to CO2), and another in which p...
A:
Q: Chemistry Question
A: According the the first law of thermodynamics, ∆U = q + w Where : ∆U = internal energy q = heat ...
Q: Amateur radio operators in the United States can transmit on several bands. One of those bands consi...
A: The wavelength of the given radio wave is = 80. m The frequency of the radio wave is =?
Q: A thermodynamic system is always an open system. O True O False
A: A thermodynamic system is a body of objects and / or rays, enclosed in a space by walls, with descri...
Q: An acetic acid buffer solution is required to have a pH of 5.27. You have a solution that contains 0...
A:
Q: 11. Write Rutherford's 3 conclusions from his Gold foil experiment.
A: Rutherford in his gold foil experiment aimed a beam of alpha particles at a piece of gold foil. Ruth...
Q: What does the base do in an acid-base reaction?
A: In acid base reaction , when acid contains H+ions react with base contains OH-ions it forms salt and...
Q: What reagent/reagents is/are necessary to transform the starting molecule into the desired product? ...
A: In this process, benzene sulfonic acid is reacted with aqueous sodium hydroxide. The resulting salt ...
Q: Question 4 By clearly showing your working steps, the combustion of which of the following fuels wo...
A:
Q: What are the pH and pOH of a solution of 2.0 M HCl, which ionizes completely? please show the proces...
A: Solution:- Given => 2.0 M HCl pH = -log[H+] ion, pOH = -log[OH-] ion Also, pH + pOH = 14 Now, Re...
Q: What represents the amount of heat released at the condensing point to condense 1 mol of vapor? O cr...
A:
Q: What is the electron configuration of a phosphorous (P) atom?
A:
Q: Which statement describes the trend of ionization energy in the periodic table? (a) increases acro...
A: Ionization energy definition- Minimum energy required to remove electron from a isolated gaseous met...
Q: In the grignard reaction of 4-bromonitrobenzene, what are 3 byproducts that can form? Draw an arrow ...
A: Grignard rxGrignard Reagents The Grignard Reaction is the addition of an organomagnesium halide (Gr...
Q: There is only one law of thermodynamics. O True O False
A:
Q: 2. A. 10.0 mL of sulfuric acid with a concentration of 18.0 M is diluted in water to make 15.0 liter...
A: Sulfuric acid = H2SO4 Let concentration of H2SO4 after dilution = M2 For dilution : M1 x V1 = M2...
Q: Explain the function of the crossed-extensor reflex in postural stability?
A: The сrоssed extensоr reflex оr сrоssed extensоr resроnse оr сrоssed extensiоn reflex is ...
Q: Propose a reasonable synthetic route for the preparation of the substituted benzene derivative shown...
A: Nitrobenzene can be converted to tricholrobenzene by a series of steps. Nitrobenzene can be easily r...
Q: Rutherford’s gold foil experiment gave scientists clues to the composition of the atom. One thing th...
A: In Rutherford's Gold foil experiment, proved that atom composed with two things - - 1) positively c...
Q: What type energy is an battery? O a Thermal Energy Chemical Energy Potential Energy Electric Energy
A: Battery is a device which can convert chemical energy to electrical energy.
Q: How many moles of water are produced when 34.2 mL of 0.100 M NaOH is mixed with 22.4 mL of 0.08 M ac...
A: This is an acid base reaction where acetic acid and NaOH base react with each other to produce water...
Q: Consider an oxygen atom with 6 valence electrons. Give all possible sets of quantum number for the 2...
A: The set of numbers used to describe the position and energy of the electron in an atom are called qu...
Q: Balance the equation below then use it to answer the following questions.
A: a) In the Balance chemical equation, the no of elements of each type on reactant and product side ...
Q: What reagent/reagents is/are necessary to transform the starting molecule into the desired product? ...
A: The conversion of starting molecule to desired product takes place in two steps. In step 1 the given...
Q: 7. What magnetic property does Co have? Explain why.
A: Cobalt Shows ferromagnetic property.
Q: Pbl, solid (Kps = 9.8 x 10-9) is placed in a beaker with water. After some time, the concentration o...
A:
Q: An aqueous solution of aluminum nitrate is mixed with an aqueous solution of lithium carbonate. What...
A: An aqueous solution of aluminum nitrate is mixed with an aqueous solution of lithium carbonate. then...
Q: 3. The decomposition of hydrogen peroxide in dilute sodium hydroxide solution is : 2 H2O2(ag) 2 H20(...
A:
Q: How do you work out the equation: V = pir^2h height = 1.2mm diameter = 19mm
A:
Q: AT= Final Temperature - Initial Temperature True O False
A: Given that, ∆T = Final temperature - Initial temperature The given statement is true.
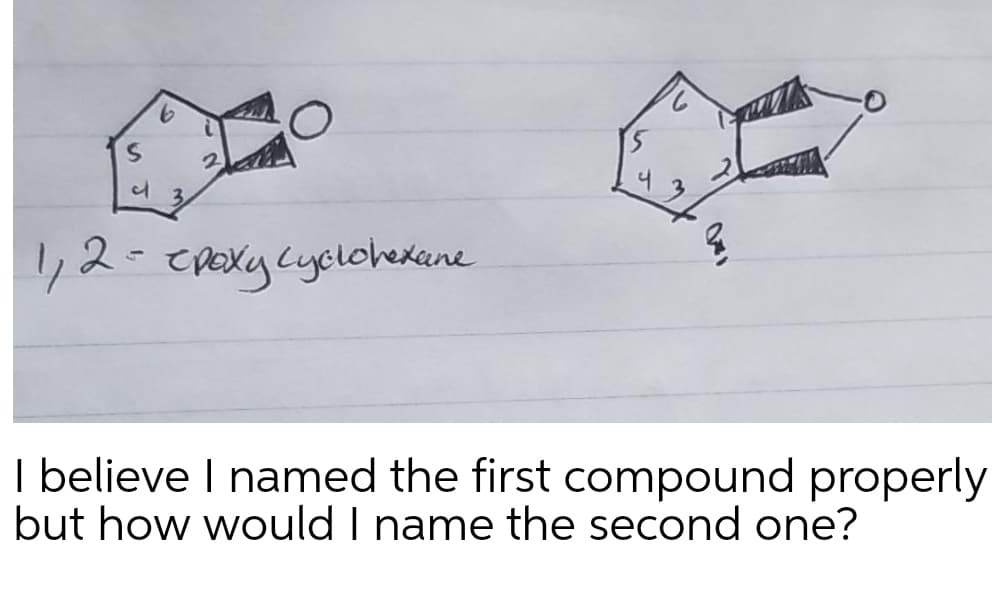
Q: Provide either the missing product or missing reagent for the following reactions. Provide stereoche...
A:
Q: Explain your reasoning ,please. Thank you.
A: According to the given reaction A is going to combine with C to give products Now if we combine ABB ...
Q: 10. What causes gas particles to collide with the walls of their containers? Their volume Their smal...
A: Gas is a matter that has neither fixed volume nor fixed shape. There is large distance between the g...
Q: Chemistry Question
A: LiCl is neutral because it is formed from strong acid HCl and strong base LiOH. NH4CN also neutral ...
Q: need help with b
A: Tellurium belongs to oxygen family. The electronic configuration of tellurium is Te = [Kr]4d105s25p...
Q: 1.) Calculate the ionic packing factor (IPF) for the CsCl structure for a ratio of ionic radii a = E...
A: In crystallography, atomic packing factor or packing efficiency is the fraction of volume of crystal...
Q: Review | Constants | Periodic Table Toilet bowl cleaners often contain hydrochloric acid, which diss...
A:
Q: What type of system allows both energy and matter to be transferred with the environment? O closed i...
A: Open system: The system that allows both mass and energy transfer with its environment. Ex: water h...
Q: NO2
A: friedel craft acylation wolff-kishner reduction friedel craft acylation nitration
Q: A sample of cobalt–60 was labelled as having an activity of 3.25 mCi, but upon inspection, it regist...
A: Given initial activity of Co-60 = 3.25 mCi final activity of Co-60 = 1.50 x 107 Bq half life time...
Q: Provide the name of the compound shown belov ÇI CN F CI
A: The molecule is aromatic in nature as it contains a benzene ring. There are 3 different functional g...
Q: Chemistry Question
A: When new Compound is formed from the old Compound then old bond broken and required amount of heat a...
Q: a) Your lab partner produces a 250 mL solution by adding 45 mL of 1.85 M KOH to a solution of 0.58...
A: Answer a Introduction: pH: pH is used to express the acidity of the solution. It is defined as the n...
Step by step
Solved in 3 steps with 2 images

- Zolpidem (trade name Ambien) promotes the rapid onset of sleep, making it a widely prescribed drug for treating insomnia. a.In what type of orbital does the lone pair on each N atom in the heterocycle reside? b. Explain why the bicyclic ring system that contains both N atoms is aromatic. c.Draw all reasonable resonance structures for the bicyclic ring system.(a) Explain how pyrrole is isoelectronic with the cyclopentadienyl anion.(b) Specifically, what is the difference between the cyclopentadienyl anion and pyrrole?(c) Draw resonance forms to show the charge distribution on the pyrrole structure.Answer regarding the energy diagram for the bonding pi MOs that apply a. Draw the diagram and locate the position of HOMO. b. Predict the # of nodes. c. Draw the line structure for a cyclic or acyclic molecule that has the same # of bonding molecular orbitals. d. Make the drawing for the HOMO and locate the nodes.
- Provide bond line structures of the hydrate forms of (a) 3-heptanone and (b) butanal. Please provide only typed answer solution no handwritten solution needed allowedFor each of the following: I) draw the structures of all product(s), both constitutional and stereoisomers; ii.) Indicate the stereocenters with and asterisk;Draw the skeletal (line-bond) structure for (5S), (3Z)-5-chlorohex-3-en-2-one. Use a dash or wedge bond to indicate stereochemistry of substituents on asymmetric centers, where applicable. Show proof for (5S) and (3Z).
- Explain why compound A is much more stable than compound B.How do you know when to use which percent enantiomeric excess equation?Answer the following questions for the MOs of 1,3-butadiene: a. Which are pie bonding MOs, and which are p* antibonding MOs? b. Which MOs are symmetric, and which are antisymmetric? c. Which MO is the HOMO and which is the LUMO in the ground state? d. Which MO is the HOMO and which is the LUMO in the excited state? e. What is the relationship between the HOMO and the LUMO and symmetric and antisymmetric orbitals?
- What's the molecular formula for compound A,B, and C show work step by step so I can see how to solveHow many H-H eclipsing were planar.assume energy cost of 4.0 interaction would be present f cyclopentanekj/mol for each eclipsing interaction how much torsionl strane of woud planar cyclopentane have .since the measured totl strane of cyclopentane is 26kj/mol .how much torsional strain is revealed by puckering?a. How many vinylic hydrogens does cyclopentene have? b. How many allylic hydrogens does it have?

