A 0.867 kg sample of ethanol is in its liquid state at its boiling temperature of 78 C. If 1.93e5 J of energy are transferred into the sample, what percentage of the sample is vaporized? ethanol has: a specific heat of 1000 J/kgK in solid form, a specific heat of 2010 J/kgK in liquid form, a freezing temperature of –114 C a boiling temperature of 78 C, a latent heat of fusion of 1.05e5 J/kg, and a latent heat of vaporization of 8.54e5 J/kg
A 0.867 kg sample of ethanol is in its liquid state at its boiling temperature of 78 C. If 1.93e5 J of energy are transferred into the sample, what percentage of the sample is vaporized? ethanol has: a specific heat of 1000 J/kgK in solid form, a specific heat of 2010 J/kgK in liquid form, a freezing temperature of –114 C a boiling temperature of 78 C, a latent heat of fusion of 1.05e5 J/kg, and a latent heat of vaporization of 8.54e5 J/kg
Physics for Scientists and Engineers, Technology Update (No access codes included)
9th Edition
ISBN:9781305116399
Author:Raymond A. Serway, John W. Jewett
Publisher:Raymond A. Serway, John W. Jewett
Chapter20: The First Law Of Thermodynamics
Section: Chapter Questions
Problem 20.8OQ: Beryllium has roughly one-half the specific heat of water (H2O). Rank the quantities of energy input...
Related questions
Question
A 0.867 kg sample of ethanol is in its liquid state at its boiling temperature of 78 C. If 1.93e5 J of energy are transferred into the sample, what percentage of the sample is vaporized?
ethanol has:
a specific heat of 1000 J/kgK in solid form,
a specific heat of 2010 J/kgK in liquid form,
a freezing temperature of –114 C
a boiling temperature of 78 C,
a latent heat of fusion of 1.05e5 J/kg, and
a latent heat of vaporization of 8.54e5 J/kg
Expert Solution
Step 1
Consider an ethanol of mass m having latent heat of vaporization Lv and Qgiven is the amount of heat is given to the sample.
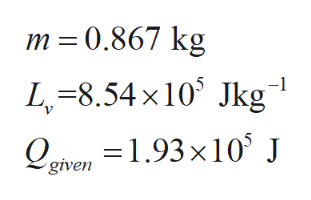
Step 2
Write the formula for heat(Qrequired) required to change its state to vapour from liquid of the entire ethanol and plug the required values to find it.
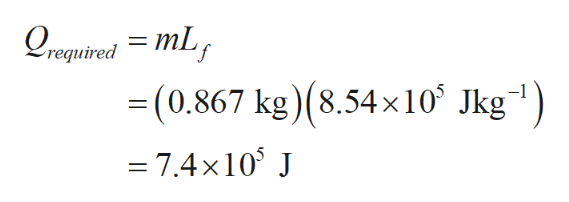
Step by step
Solved in 4 steps with 4 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you

Physics for Scientists and Engineers, Technology …
Physics
ISBN:
9781305116399
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:
9781133104261
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

Physics for Scientists and Engineers: Foundations…
Physics
ISBN:
9781133939146
Author:
Katz, Debora M.
Publisher:
Cengage Learning

Physics for Scientists and Engineers, Technology …
Physics
ISBN:
9781305116399
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:
9781133104261
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

Physics for Scientists and Engineers: Foundations…
Physics
ISBN:
9781133939146
Author:
Katz, Debora M.
Publisher:
Cengage Learning