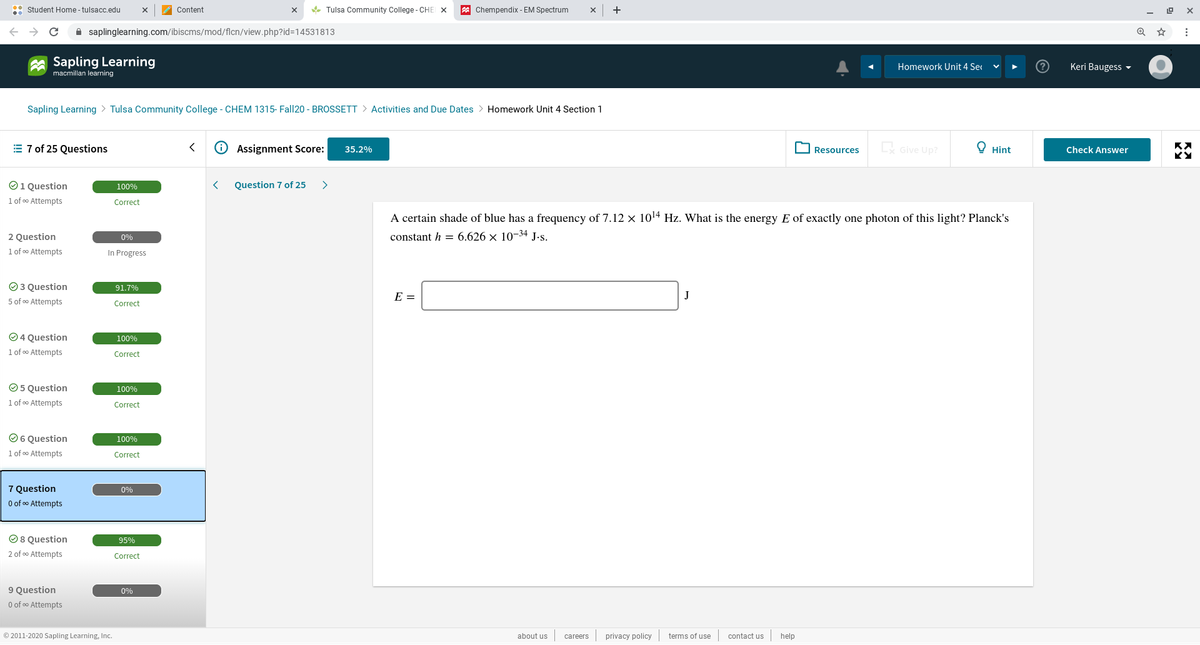
A certain shade of blue has a frequency of 7.12 x 1014 Hz. What is the energy E of exactly one photon of this light? Planck's constant h = 6.626 x 10-34 J-s. E =
A certain shade of blue has a frequency of 7.12 x 1014 Hz. What is the energy E of exactly one photon of this light? Planck's constant h = 6.626 x 10-34 J-s. E =
Chapter33: High-performance Liquid Chromatography
Section: Chapter Questions
Problem 33.17QAP
Related questions
Question
Transcribed Image Text:: Student Home - tulsacc.edu
Content
* Tulsa Community College - CHE x
A Chempendix - EM Spectrum
+
A saplinglearning.com/ibiscms/mod/flcn/view.php?id=14531813
Sapling Learning
Homework Unit 4 Ser
Keri Baugess ▼
macmillan learning
Sapling Learning > Tulsa Community College - CHEM 1315- Fall20 - BROSSSETT > Activities and Due Dates > Homework Unit 4 Section 1
= 7 of 25 Questions
Assignment Score:
Give Up?
У Hint
35.2%
Resources
Check Answer
O1 Question
Question 7 of 25
>
100%
1 of o Attempts
Correct
A certain shade of blue has a frequency of 7.12 × 1014 Hz. What is the energy E of exactly one photon of this light? Planck's
constant h = 6.626 x 10-34 J.s.
2 Question
1 of o Attempts
0%
In Progress
O 3 Question
91.7%
E =
J
5 of o Attempts
Correct
O 4 Question
100%
1 of o Attempts
Correct
O 5 Question
100%
1 of oo Attempts
Correct
O6 Question
100%
1 of oo Attempts
Correct
7 Question
0%
O of oo Attempts
O8 Question
2 of o Attempts
95%
Correct
9 Question
O of o Attempts
0%
© 2011-2020 Sapling Learning, Inc.
about us
privacy policy
terms of use
contact us
help
careers
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

