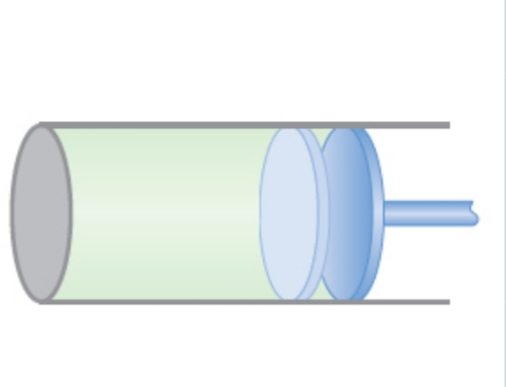
A cylinder with a frictionless, movable piston like that shown in the figure, contains a quantity of helium gas. Initially the gas is at a pressure of 1.00×105Pa, has a temperature of 300K, and occupies a volume of 1.50L. The gas then undergoes two processes. In the first, the gas is heated and the piston is allowed to move to keep the temperature equal to 300K. This continues until the pressure reaches 2.50×104Pa. In the second process, the gas is compressed at constant pressure until it returns to its original volume of 1.50L. Assume that the gas may be treated as ideal. a. Find the temperature of the gas at the end of the second process. b. Find the total work done by the gas in the first process. c. Find the total work done by the gas in the second process.
A cylinder with a frictionless, movable piston like that shown in the figure, contains a quantity of helium gas. Initially the gas is at a pressure of 1.00×105Pa, has a temperature of 300K, and occupies a volume of 1.50L. The gas then undergoes two processes. In the first, the gas is heated and the piston is allowed to move to keep the temperature equal to 300K. This continues until the pressure reaches 2.50×104Pa. In the second process, the gas is compressed at constant pressure until it returns to its original volume of 1.50L. Assume that the gas may be treated as ideal.
a. Find the temperature of the gas at the end of the second process.
b. Find the total work done by the gas in the first process.
c. Find the total work done by the gas in the second process.
Trending now
This is a popular solution!
Step by step
Solved in 7 steps with 6 images









