(a) Name each compound. (b). If a mixture of these compounds was fractionally distilled, give the order in which they would distil off, starting with the top of the fractionating column. Explain your answer.
(a) Name each compound. (b). If a mixture of these compounds was fractionally distilled, give the order in which they would distil off, starting with the top of the fractionating column. Explain your answer.
Chapter4: Organic Compounds: Cycloalkanes And Their Stereochemistry
Section4.SE: Something Extra
Problem 57AP: The German chemist J. Bredt proposed in 1935 that bicycloalkenes such as 1-norbornene, which have a...
Related questions
Question
Answer all questions please

Transcribed Image Text:(a)
Name each compound.
(b).
If a mixture of these compounds was fractionally distilled, give the order in which
they would distil off, starting with the top of the fractionating column. Explain
your answer,
6.
What are Conformational isomers?
7.
Which of the following hydrocarbons does not have isomers?
(a) CH16
(b) C&H14
(c) CSH10
(d) C4H8
(e) C3H8
8. How many isomeric alkanes of the molecular formula CSH12 are there?
(a) 1 (b) 2 (c) 3 (d) 4 (e) 5
9. Give a systematic name for each of the following alkanes
(a)
(b)
(c)
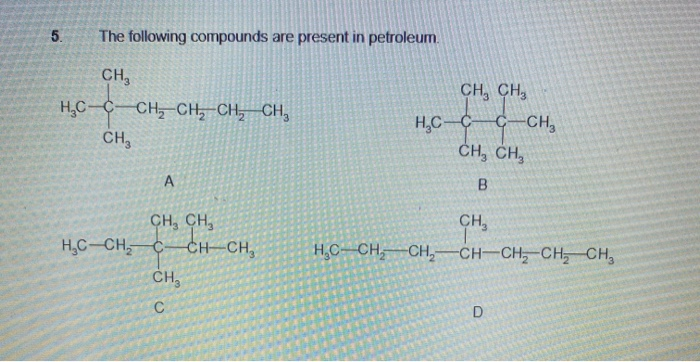
Transcribed Image Text:5.
The following compounds are present in petroleum.
CH,
CH, CH,
H,C-C-CH, CH, CH, CH,
H,C-C
CH, CH,
C-CH,
CH
CH, CH,
CH
H,C-CH, C-
CH CH,
H,C--CH,–CH,-CH-CH, CH, CH,
CH
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning


Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning