A precipitation reaction occurs when 655 mL of 0.639 M Pb(NO2), reacts with 313 mL of 0.855 M KI, as shown by the following equation. Pb(NO3)2(aq) 2 KI(aq)Pbl, (s) + 2 KNO3 (aq) Identify the limiting reactant. O Pb(NO3)2 O KI O Pbl2 Ο ΚNO, KNO3 Calculate the theoretical yield of PbI, from the reaction. mass of Pbl,: Calculate the percent yield of PbI, if 48.0 g of Pbl, are formed experimentally. percent yield of PbI,:
A precipitation reaction occurs when 655 mL of 0.639 M Pb(NO2), reacts with 313 mL of 0.855 M KI, as shown by the following equation. Pb(NO3)2(aq) 2 KI(aq)Pbl, (s) + 2 KNO3 (aq) Identify the limiting reactant. O Pb(NO3)2 O KI O Pbl2 Ο ΚNO, KNO3 Calculate the theoretical yield of PbI, from the reaction. mass of Pbl,: Calculate the percent yield of PbI, if 48.0 g of Pbl, are formed experimentally. percent yield of PbI,:
Chemistry: Principles and Practice
3rd Edition
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Chapter4: Chemical Reactions In Solution
Section: Chapter Questions
Problem 4.120QE
Related questions
Question
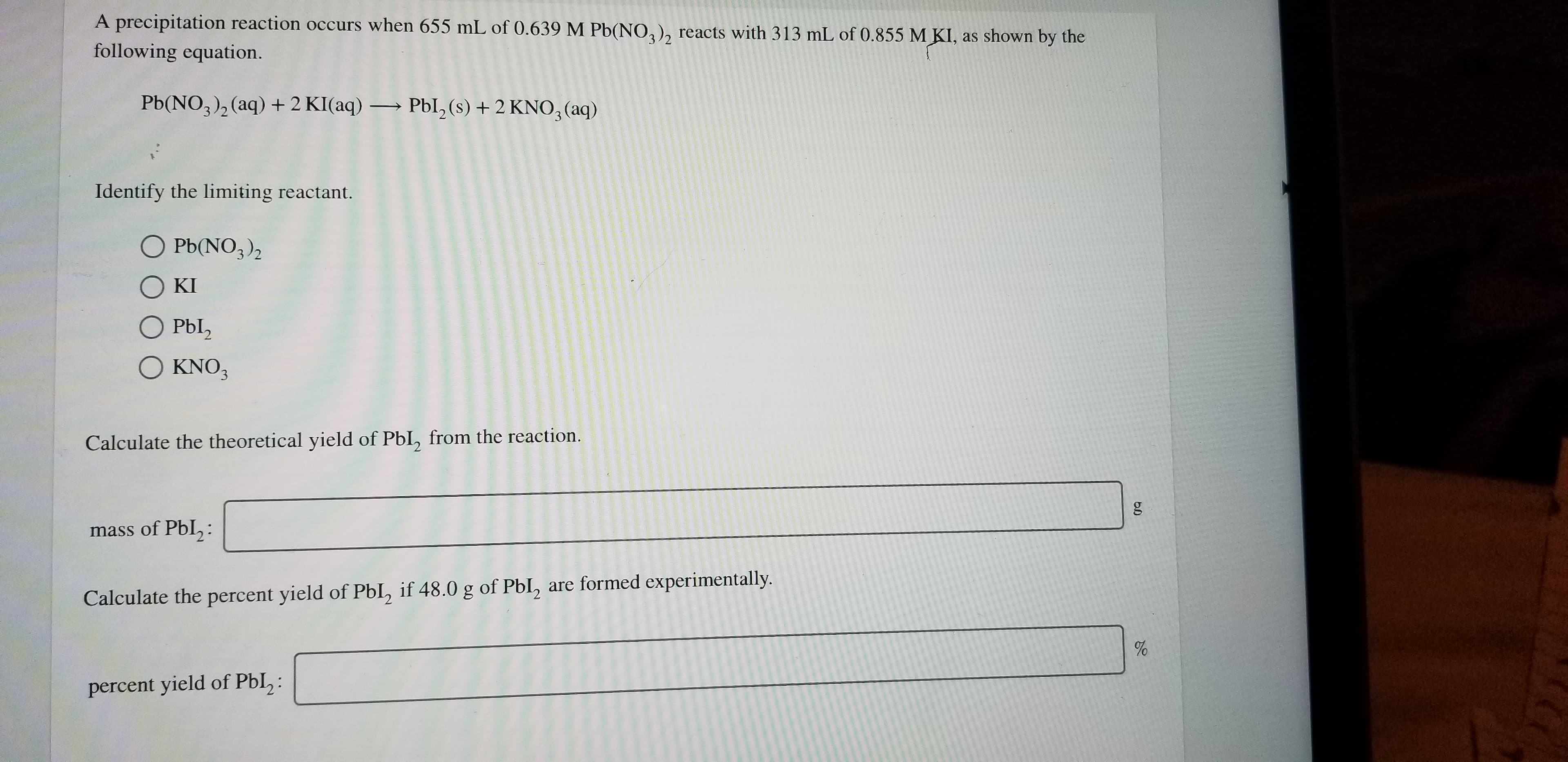
Transcribed Image Text:A precipitation reaction occurs when 655 mL of 0.639 M Pb(NO2), reacts with 313 mL of 0.855 M KI, as shown by the
following equation.
Pb(NO3)2(aq) 2 KI(aq)Pbl, (s) + 2 KNO3 (aq)
Identify the limiting reactant.
O Pb(NO3)2
O KI
O Pbl2
Ο ΚNO,
KNO3
Calculate the theoretical yield of PbI, from the reaction.
mass of Pbl,:
Calculate the percent yield of PbI, if 48.0 g of Pbl, are formed experimentally.
percent yield of PbI,:
Expert Solution
Trending now
This is a popular solution!
Step by step
Solved in 10 steps with 8 images

Recommended textbooks for you

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning