A sample consisting of 1 mol of perfect gas atoms (for which Cv,m = 3/½R) is taken through the cycle shown below. Using the ideal gas law, the temperature at points 1, 2 and 3 were be found to be 273 K, 547 K and 273 K, respectively. In the questions below justify your reasoning and show all your calculations. 1.00 2 1 0.50 22.44 44.88 Volume /dm3 a) For step 1→ 2, calculate q, w, AU, and AH (give all answers in terms of kJ). b) For step 3 → 1, calculate q, w, AU, and AH (give all answers in terms of kJ). c) For step 2 → 3, determine w, AH and AS. Pressure /atm
A sample consisting of 1 mol of perfect gas atoms (for which Cv,m = 3/½R) is taken through the cycle shown below. Using the ideal gas law, the temperature at points 1, 2 and 3 were be found to be 273 K, 547 K and 273 K, respectively. In the questions below justify your reasoning and show all your calculations. 1.00 2 1 0.50 22.44 44.88 Volume /dm3 a) For step 1→ 2, calculate q, w, AU, and AH (give all answers in terms of kJ). b) For step 3 → 1, calculate q, w, AU, and AH (give all answers in terms of kJ). c) For step 2 → 3, determine w, AH and AS. Pressure /atm
Physical Chemistry
2nd Edition
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Ball, David W. (david Warren), BAER, Tomas
Chapter1: Gases And The Zeroth Law Of Thermodynamics
Section: Chapter Questions
Problem 1.32E
Related questions
Question
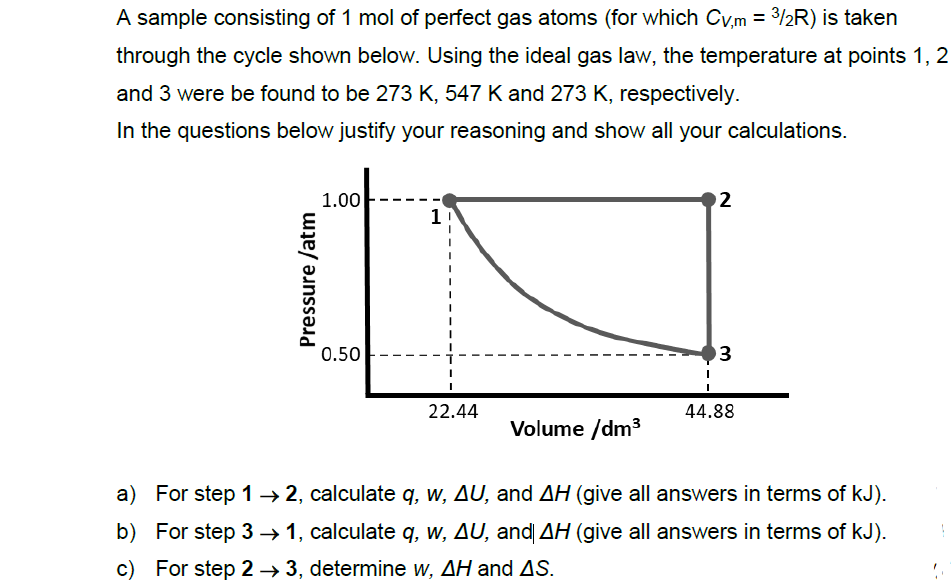
Transcribed Image Text:A sample consisting of 1 mol of perfect gas atoms (for which Cv,m = 3/2R) is taken
through the cycle shown below. Using the ideal gas law, the temperature at points 1, 2
and 3 were be found to be 273 K, 547 K and 273 K, respectively.
In the questions below justify your reasoning and show all your calculations.
1.00
2
1
0.50
3
22.44
44.88
Volume /dm3
a) For step 1→ 2, calculate q, w, AU, and AH (give all answers in terms of kJ).
b) For step 3 → 1, calculate q, w, AU, and AH (give all answers in terms of kJ).
c) For step 2 → 3, determine w, AH and AS.
Pressure /atm
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning