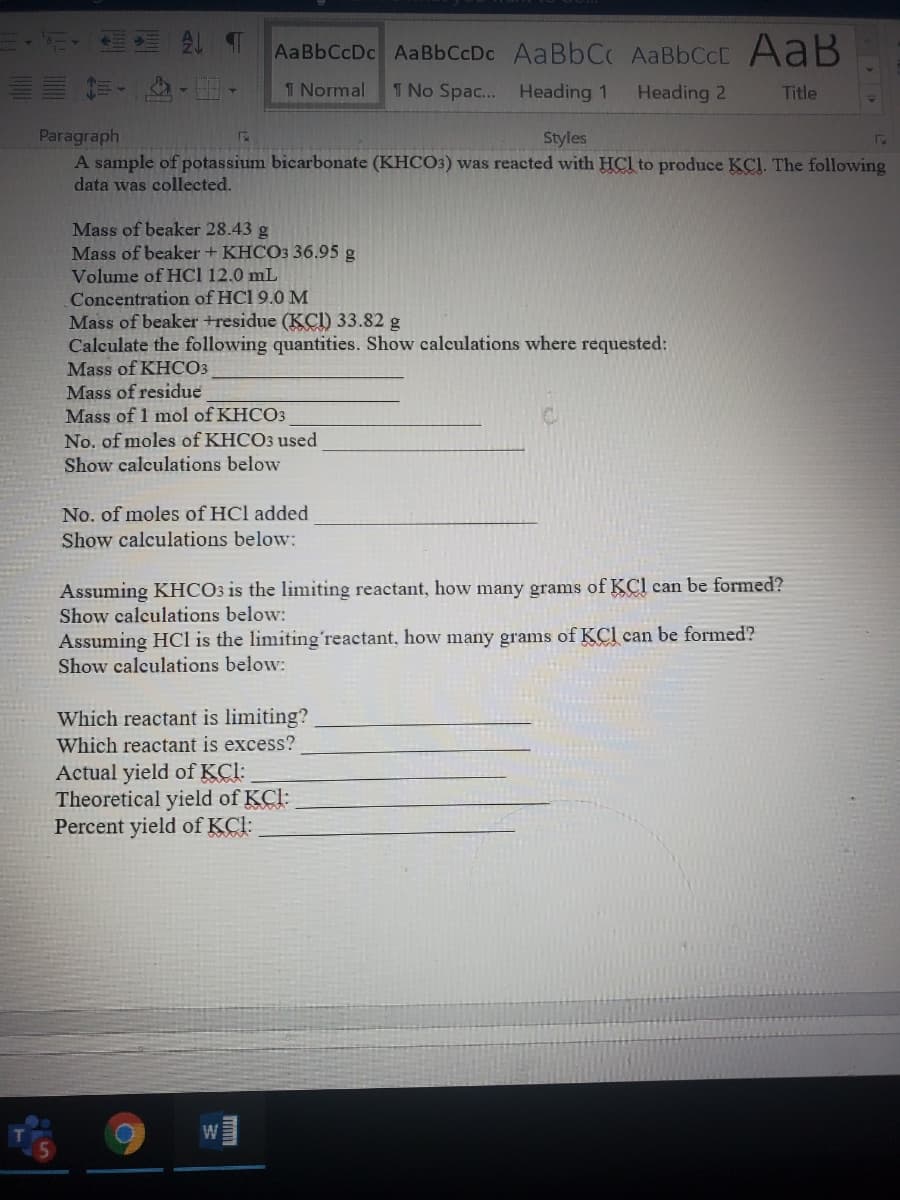
A sample of potassium bicarbonate (KHCO3) was reacted with HCl to produce KCl. The followin data was collected. Mass of beaker 28.43 g Mass of beaker + KHCO3 36.95 g Volume of HCl 12.0 mL Concentration of HCl 9.0 M Mass of beaker +residue (KCI) 33.82 g Calculate the following quantities. Show calculations where requested: Mass of KHCO3 Mass of residue Mass of 1 mol of KHCO3 No. of moles of KHCO3 used Show calculations below No. of moles of HCl added Show calculations below: Assuming KHCO3 is the limiting reactant, how many grams of KCl can be formed? Show calculations below: Assuming HCl is the limitingʻreactant, how many grams of KCl can be formed? Show calculations below: Which reactant is limiting? Which reactant is excess? Actual yield of KCl: Theoretical yield of KCl: Percent yield of KCl:
A sample of potassium bicarbonate (KHCO3) was reacted with HCl to produce KCl. The followin data was collected. Mass of beaker 28.43 g Mass of beaker + KHCO3 36.95 g Volume of HCl 12.0 mL Concentration of HCl 9.0 M Mass of beaker +residue (KCI) 33.82 g Calculate the following quantities. Show calculations where requested: Mass of KHCO3 Mass of residue Mass of 1 mol of KHCO3 No. of moles of KHCO3 used Show calculations below No. of moles of HCl added Show calculations below: Assuming KHCO3 is the limiting reactant, how many grams of KCl can be formed? Show calculations below: Assuming HCl is the limitingʻreactant, how many grams of KCl can be formed? Show calculations below: Which reactant is limiting? Which reactant is excess? Actual yield of KCl: Theoretical yield of KCl: Percent yield of KCl:
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter1: Matter And Measurements
Section: Chapter Questions
Problem 56QAP: Magnesium chloride is an important coagulant used in the preparation of tofu from soy milk. Its...
Related questions
Question
Transcribed Image Text:AaBbCcDc AaBbCcDc AaBbCC AABBCCC A aB
前。
1 Normal
1 No Spac... Heading 1
Heading 2
Title
Paragraph
A sample of potassium bicarbonate (KHCO3) was reacted with HCl to produce KCl. The following
Styles
7
data was collected.
Mass of beaker 28.43 g
Mass of beaker + KHCO3 36.95 g
Volume of HC1 12.0 mL
Concentration of HCl 9.0 M
Mass of beaker +residue (KCI) 33.82 g
Calculate the following quantities. Show calculations where requested:
Mass of KHCO3
Mass of residue
Mass of 1 mol of KHCO3
No. of moles of KHCO3 used
Show calculations below
No. of moles of HCl added
Show calculations below:
Assuming KHCO3 is the limiting reactant, how many grams of KCl can be formed?
Show calculations below:
Assuming HCl is the limitingʻreactant, how many grams of KCl can be formed?
Show calculations below:
Which reactant is limiting?
Which reactant is excess?
Actual yield of KCI:
Theoretical yield of KCl:
Percent yield of KCl:
W
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 6 steps with 7 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning