Q: Fill in all H's and lone pairs in each compound. b. c--c а. С—С—с—С С. С —С—С d. C-Ĉ-N-C е.
A:
Q: Draw in all H's and lone pairs in each compound. a. C–C-CI b. C-C=0 С. С -С—N
A: There are 5 H's and 3 lone pairs on chlorine.
Q: он NANON WIN NH2 HO но- -NH O Na* SINGTO NEWWIE
A: The molecule having highest conjugation without any break or cross conjugation are the most stable.
Q: Propyne contains bonds, _n bonds and has atoms connected linearly. The number of polar bonds in…
A: Given,
Q: w the simplest set of curved arrows that shows how the structure on the left could be turned into…
A:
Q: List these compounds in order of increasing carbon–carbon bondstrength and in order of decreasing…
A: The structures of HCCH, H2CCH2, H3CCH3 are:
Q: Name the molecule drawn below. Be as exact with your nomenclature as you can. ОН OH H C- ОН H НО С-…
A: The given compound consist of hydroxyl (OH-) group on each carbon atom of the cyclic ring.
Q: Determine the number of lone pairs (if any) on each oxygen, nitrogen, and charged carbon atoms in…
A:
Q: There are at least three different molecules with the formula C3H8O. Draw a Lewis structure for each…
A: The compounds having the same molecular formula but having different structural formulas are called…
Q: Which molecules show an appropriate number of bonds around each carbon atom?
A: A question about carbon, which is to be accomplished.
Q: Which condensed structure below contains an incorrect number of hydrogens? O CH3 CH2 CH3 O CH3 CH2…
A: The details solution for this is provided below in attach image.
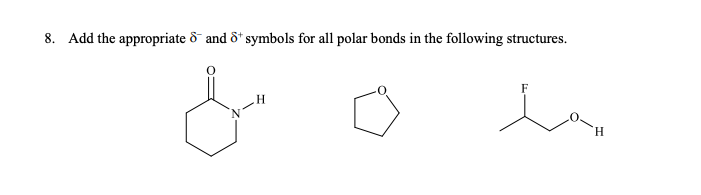
Q: Rank the following bonds from least polar to most polar: N-Cl C-N C-O
A: Polarity of any bond depends on the electronegativity difference between the atoms. More is the…
Q: How would I convert each condensed formula to a lewis structure 1a. (CH3)2CHOCH2CH2CH2OH 1b.…
A: Lewis structure : Lewis structure is a very simplified representation of the valance shell…
Q: Add curved arrows to both structures to show the delocalization of electron pairs needed to form the…
A: To make resonance structures, the lone pair electrons are converted into bond pair electrons and…
Q: Add curved arrows to both structures to show the delocalization of electron pairs needed to form the…
A: resonance is the delocalization of electron without changing the positions of atoms
Q: Ar- EN -F
A: Asuming Ar is for Argon. Diazepine is a seven-membered heterocyclic compound with two nitrogen atoms…
Q: 7. Indicate the number of bonds and lone pairs of electrons on each of the following atoms (a) a…
A: Different type of atoms contain different number of Electrons. When atoms are denoted by dots they…
Q: A triple bonded carbon can only bond with one other atom. True False
A: For any element Number of bond = Valence electron of element Valence electron : In the electronic…
Q: Convert each condensed formula to a Lewis structure.1.) (CH3)2CHOCH2CH2CH2OH2.)…
A: A Lewis Structure is a very simplified representation of the valence shell electrons in a molecule.…
Q: 7. Draw curved arrows indicating the movements of electrons between the following pair of resonance…
A: Since you have posted unrelated multiple questions, hence answering only first 2 related ones.
Q: Add the unshared (lone) electron pairs to the following structure
A: Lone pair indicates a pair of valence electrons that are not shared with another atom in a covalent…
Q: Tag all the carbons that are bonded to 2 H's in the molecule below.
A:
Q: How many hydrogen atoms are present around each highlighted carbon atom in the following molecules?…
A: Carbon atom can form maximum 4 bonds as the number of valence electrons of carbon is 4.
Q: a)
A:
Q: 3-methylpentane about the C2 -C3 bond (b) 3 3-dimethylhexane about the C3 –C4 bond
A: I THINK IT DOESNT MATTER
Q: H0 3
A: IUPAC Nomenclature for the given compound = 2,3-dimethyl-2-butene. It is a 4 carbon chain skeleton.…
Q: : Indicate the product resulting from the depicted curved arrows. Don't forget to indicate charges…
A: The reaction step given is,
Q: 5. Answer BOTH parts of this question. (a) Name molecules A and B shown in figure Q5.
A:
Q: 10. Name the following. Why is no number needed to indicate the position of the double bond?
A: The hydrocarbons which contain double covalent bond with the general formula Cn H2n are regarded as…
Q: On the worksheet, add any missing lone pairs.
A: Hi, since you have posted questions with multiple subparts, we will answer the first three subparts.…
Q: llowing condensed structures to Kekulé structures, showing ponds: ▾ Part A CH3NH(CH₂)2CH3 Draw the…
A: Kekule structure are similar to Lewis structure. It represents a structural formula for an organic…
Q: 1. Write the symbol for the following isotopes: Write the expanded, condensed and bond line formulas…
A: The expanded structural formula is the one where all the bonds by which atoms are connected are…
Q: Draw the Lewis structure of HCN. Include lone pairs.
A: Lewis structure is bonding of atoms in the molecule is represented by lines between atoms. the…
Q: Convert each condensed formula to a Lewis structure. a.CH3(CH2)4CH(CH3)2 b. (CH3)3CCH(OH)CH2CH3 c.…
A: a,b) Please find below the Lewis structure of a and b
Q: 9. Identify the relationship between the following pair of molecules?
A: We need to find the relationship between given pair of molecules
Q: Methyl isocyanate, shown as resonance structure 1, can also be represented by other resonance…
A: Resonance structure:If the Lewis structure of a molecule or ion cannot explain by a single structure…
Q: the compound that should have the lowest boiling point, based on each compound's small molecule…
A: Boiling point of a solvent is directly proportional to the force of attraction between the atoms or…
Q: a) Name each of the following organic molecules using IUPAC convention но Br Cl- Но но 1) 2) Cl, Но…
A: The given structures are,
Q: The structure below contains a charged carbon atom. Add all hydrogen atoms and lone pair(s) to the…
A: Carbon atom generally forms 4 bonds. So add an H atom until the central carbon atom has an octet i.e…
Q: How many (non H) axial positions are shown in the following molecule
A: In this question, you will find-out the total number of axial position without axial hydrogen (…
Q: Formula Lewis Electron Molecular Bond Polar or Attractive Structure Pair Geometry Angle Force Non…
A: Given: Since so many options are given here, i answered first three. please ask other options…
Q: Identify the structure that shows the correct placement of all lone pairs for the compound…
A: Lone pairs of electrons present on the atoms is calculated using the formula of formula charge.
Q: Convert each condensed formula to a Lewis structure. a. CH3(CH2)4CH(CH3)2 b. (CH3)3CCH(OH)CH2CH3 c.…
A: Since we answer upto 3 sub-parts, we'll answer first three. Please resubmit the question specifying…
Q: How many hydrogen atoms are connected to the indicated carbon atom? one two O three four none…
A: Answer
Q: Name the functional group. Some molecules will have more than one functional group; in those cases,…
A: "Since you have posted a question with multiple sub-parts. We will solve the first three subparts…
Q: Identify the highlighted functional groups in this molecule. HO- CH, CH H,N CH CH ČH, terms of use…
A: The functional group is the group of atoms within the molecule that show their specific character in…
Q: 5. Functional Groups can identify reactivity and predict chemical behavior Identify the functional…
A: Functional groups: These are the groups of atoms or an atom that present in the organic compounds…
Q: Circle the formula of all compounds below that is/are capable of engaging in hydrogen-bonding to…
A: Hydrogen bonding is a type of dipole-dipole interaction of molecules when the hydrogen is bonded to…
Step by step
Solved in 3 steps with 3 images

- Describe the orbitals used by each carbon atom in bonding and indicate the approximate bond angles. Drag the appropriate items to their respective bins.Using your model of butane (CH3CH2CH2CH3) , complete the following graph of the anglebetween the two Me groups vs. potential energy. a. Label each Newman projection of butane on the graph with the words staggered, eclipsed, gauche, and anti, as appropriate. (Note that some structures will have more than one label.) b. Draw a wedge and dash bond representation of butane in its lowest P.E. conformation.Is bond formation endothermic or exothermic? Write a + or sign above the arrow in the previousquestion to represent the sign of the energy change associated with the arrow.
- name the following compounds. only need part q, j and rProjection drawings of all isomers of C6H4Cl2. Can you convert one into another without breaking any bonds? What type of isomerism do these represent?Which of the given resonance structures (A, B,or C) contributes most to the resonance hybrid? Which contributes least?

