A: Order the three compounds from least to most polar B: Which of these compounds should have the smallest Rf value? acetylferrocene diacetylferrocene ferrocene C: If you had to choose only one solvent system to use to separate these three compounds, which would give the most efficient separation? none of these are suitable for a column ethyl acetate 1:1 hexanes/ethyl acetate hexanes
A: Order the three compounds from least to most polar B: Which of these compounds should have the smallest Rf value? acetylferrocene diacetylferrocene ferrocene C: If you had to choose only one solvent system to use to separate these three compounds, which would give the most efficient separation? none of these are suitable for a column ethyl acetate 1:1 hexanes/ethyl acetate hexanes
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter20: Chemistry Of Selected Transition Elements And Coordination Compounds
Section: Chapter Questions
Problem 116QRT
Related questions
Question
Answer the following question regarding the pictures please:
The information for this set is based off of the column separation of ferrocene and acetylferrocene.
A: Order the three compounds from least to most polar
B: Which of these compounds should have the smallest Rf value?
acetylferrocene
diacetylferrocene
ferrocene
C: If you had to choose only one solvent system to use to separate these three compounds, which would give the most efficient separation?
none of these are suitable for a column
ethyl acetate
1:1 hexanes/ethyl acetate
hexanes
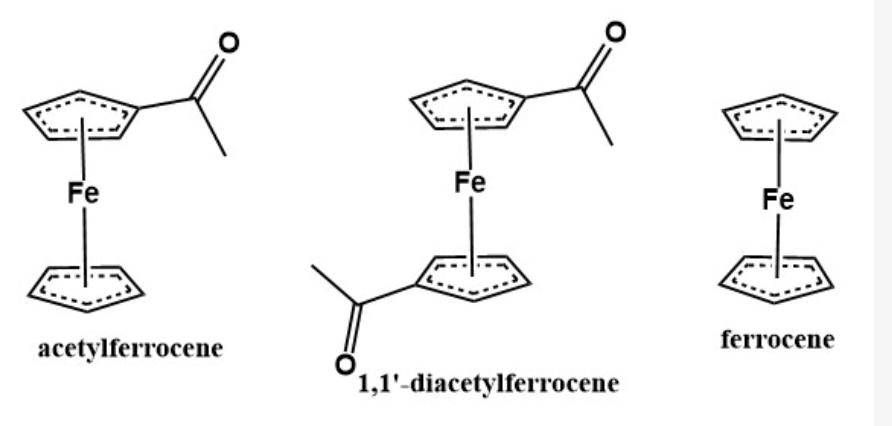
Transcribed Image Text:Fe
Fe
Fe
ferrocene
acetylferrocene
1,1'-diacetylferrocene
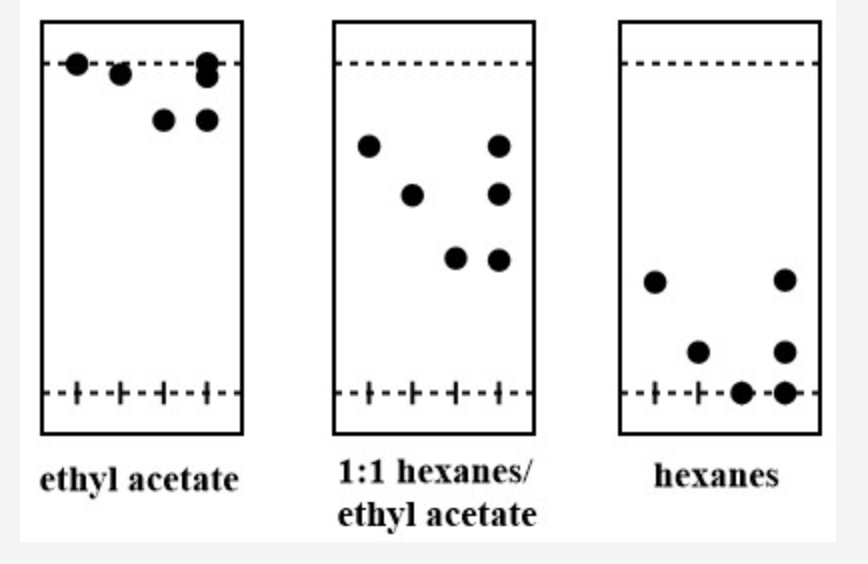
Transcribed Image Text:----
--+-----
ethyl acetate
1:1 hexanes/
hехanes
ethyl acetate
Expert Solution
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

