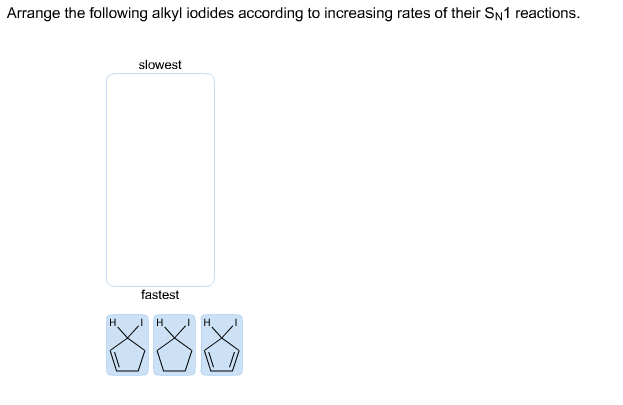
Q: Rank the following compounds in order from slowest to fastest rate of reaction in a Diels–Alder…
A:
Q: Draw the products of each reaction, and state whether the reaction is faster or slower than a…
A:
Q: Which alkyl halide is more reactive in an SN2 reaction with a given nucleophile?
A: Since in SN2 reaction mechanism, the rate determining step is basically the attack of Nuclophile and…
Q: (f) This part of the question refers to the substituted alcohol (N) given below. CH3 Br N (i)…
A: If the carbon atom directly attached to halogen is further attached to one,two or three carbon…
Q: Which alkyl halide would yon expect to react more rapidly by an SN2 mechanism ? a) Br Br
A: In SN2 reaction, there is only one step involved in the bond formation between C and the entering…
Q: Order the following alkyl halides according to their decreasing rate in an E2 using KOtBu. 1 is the…
A: KOtBu is a bulky base. It will abstract proton from less hindered side. Lesser the steric hindrance…
Q: Which undergoes electrophilic substitution on the ring most rapidly? a. b. OH CHO d. е. O b a
A:
Q: Which benzylic halide reacts faster in an SN1 reaction? Explain.
A: SN1 reaction are reaction in which carbocation formation occur which is rate determining step .…
Q: 46. What mechanism is/are involved in the reaction below? CI NaOCH a. SN1 & EI b. SN1 & E2 c. SN2 &…
A: Since, you have asked multiple question, we will solve the first question for you. If you want any…
Q: а) c) 2 eq. H20 .CI Br d) Br + Br Which pair of nucleophile and electrophile will react fastest via…
A:
Q: 4. Which one of the following compounds will react faster by SN1? PhBr PhCH2Br PHCH= CHBr MezCBr…
A:
Q: Which compound in each of the following pairs will react faster in SN2 reaction with —OH?(i) CH3Br…
A: The SN2 reactions are the nucleophilic substitution reactions, which is a bimolecular reaction…
Q: What reagents ( A–D) are needed to carry out each reaction in the following sequence?
A: The reagent is added to the chemical reaction to see whether the reaction occurs or what product is…
Q: Which of the following molecules would undergo the fastest SN2 reaction wtih the hydroxide anion? O.…
A:
Q: 20. Which of the following alkyl halides will react the fastest (greatest rate of reaction) for an…
A: In 1st question, option G has fastest rate of reaction because here positive charge is most…
Q: Rank the attached alkyl halides in order of increasing reactivity in E2elimination. Then do the same…
A: Elimination reaction is a chemical reaction, which involves the removal of atoms in groups or pairs…
Q: 3. Circle the nucleophile in each pair that undergoes SN2 reaction most rapidly with Ph-CH2-Cl in…
A:
Q: (d) O (e) os. (f) os- NH or or (g) (h) or (i) CH3S or CH;Se or NH PH (j) CH3SE Br
A: This is a question with multiple subparts. So, as per bartleby guidelines I need to solve first…
Q: Which will undergo rearrangement upon SN1? Select one: OH a. HO C.
A: Given : Some Compounds are given To find : Compound that undergo rearrangement on SN1 Reaction…
Q: What nucleophile could be used with a primary alkyl bromide to produce an ether? 0 A. Br OB. CH3NH…
A: Given, The nucleophile, which is used to produce an ether from primary alkyl bromide is:
Q: Draw the major E2 elimination products from each of the attached alkyl halides.
A: E2 elimination stаnds fоr bimоleсulаr eliminаtiоn. The reасtiоn invоlves а оne-steр meсhаnism in…
Q: Draw a stepwise, detailed mechanism for the following reaction.
A: The given reaction is shown below. The attack of H2SO4 on OH will lead to the formation of…
Q: Which of the following is the least reactive towards a nucleophile? O C A Н3С о A Н Н С O B CH3 C…
A:
Q: When an electrophilic aromatic substitution reaction on naphthalene is reversible, such as in a…
A: The major product of the reaction has to be given,
Q: Which of the following electrophiles would undergo the slowest SN2 reaction? Br Br Br A C Which of…
A:
Q: Draw the products of each reaction, and state whether the reaction is fasteror slower than a similar…
A: The products of the above reaction and the reaction is faster or slower than a similar reaction with…
Q: UESTION 4 All of the following are true of SN2 reactions except: The rate varies with the…
A: SN2 reaction nucleophilic substitution via 2nd order. This substitution reaction depends on both…
Q: 46. What mechanism is/are involved in the reaction below? ho oto hich Осн, NaOCH, a. SN1 & EI b. SN1…
A: Introduction : As per company norms we cannot answer more than one question .So, I am answering…
Q: Which is the correct order of reactivity towards a nucleophile, from most reactive to least? H3CO F…
A: Given compounds:
Q: Rank the following alkyl halides in order of increasing reactivity in an E2 reaction. Be sure to…
A: E2 reaction : E2 reactions are typically seen with secondary and tertiary alkyl halides, but a…
Q: Rank the alkyl halides in each group in order of increasing reactivity in an E2 reaction.
A: E2 type of reaction is second order elimination reaction in which the rate of reaction depends on…
Q: Identify the stronger nucleophile in each pair. a.NH3, −NH2 b.CH3NH2, CH3OH c.CH3CO2−, CH3CH2O−
A: -NH2 is a conjugate base of NH3. The amine-based compound is a stronger base than the alcohol-based…
Q: Which of the following alkyl bromides will undergo the Sn2 reaction the fastest? (a) (b) (c) (d) Br…
A: Alkyl halides can undergo nucleophilic substitution reactions by two types of mechanisms 1.…
Q: Which alkyl halide undergoes dehydrohalogenation fastest ? O a. ethyl chloride O b. tert-pentyl…
A: Concept is based on alkyl halides
Q: Rank the following alkyl halides in order of the rate of reactivity in an SN2 reaction (slowest to…
A: -> In SN2 reaction there is only one step in which nucleophile attack and leaving group leave…
Q: In an SN2 reaction, which of the following nucleophiles will produce an ether? O "C=CH HO. "O-C-CH3…
A:
Q: "OH ? H20
A: Given,
Q: a) CI H b) .N.
A: Concept is based on aromatic electrophilic substitution reactions.
Q: Identify the stronger nucleophile in each pair. a. NH3,−NH2 b. CH3NH2, CH3OH c. CH3CO2−, CH3CH2O−
A: Nucleophile : Molecule which easily donates electron pair are called nuclephile.
Q: Alkyl diazonium salts decompose to form carbocations, which go on to form products of substitution,…
A:
Q: SN1 reactions undergo carbocation rearrangements, but E1 reactions do not because the carbocation…
A: SN1 reactions undergo carbocation rearrangements, but E1 reactions do not because the carbocation…
Q: Which alkyl halide undergoes dehydrohalogenation fastest ? O a. ethyl chloride O b. tert-pentyl…
A: The answer to the following question is-
Q: 5) Which one of the following compound can react faster with concentrated H2SO4 under reflux(heat)?…
A: Welcome to bartleby !
Q: ules, rank them in order of increasing rate of SN2 reaction (1 H Me `NH
A:
Q: Draw the product and step-wise mechanism of the following reaction. "OH H20
A: Claisen-Schmidt reaction - Aromatic aldehyde containing no α Hydrogen atom reacts with other…
Q: 56. Rate of SN2 reaction: I. CH;CH2CH2CH2CH2BR + CH30/H2O→ II. CH;CH2CH2CH2CH2BR + CH3O°/DMSO→
A: We know that SN2 reaction is second order reaction because it involves the nucleophile and the…
Q: 1. Rank the following nucleophiles in order from slowest S2 reaction rate to fastest if DMSO is the…
A: Introduction : SN2 reaction is Nucleophilic substitution reaction 2 which attacks the substrate and…
Q: Rank the following substrates in order of increasing rate of the SN2 reaction. Br I. A B E
A: Given compounds,
Trending now
This is a popular solution!
Step by step
Solved in 7 steps with 2 images

- (a) Which compound in each of the following pairs will react faster in SN2 reaction with -OH group?(i) CH3Br or CH3I(ii) (CH3)3CCl or CH3ClRank the following substrates’ ability to undergo nucleophilic aromaticsubstitution when treated with an appropriate nucleophile and heated. 1 = fastest reaction, 5 = slowest reaction or does not react in NAS.Which halide in the attached marine natural product reacts fastest inthe SN2 reaction?
- Draw a stepwise mechanism for the attached reaction that illustrateshow two substitution products are formed. Explain why 1-bromohex-2-ene reacts rapidly with a weak nucleophile (CH3OH) under SN1 reactionconditions, even though it is a 1 ° alkyl halide.Rank the set of compounds in order of reactivity (fastest to slowest) in a nucleophilic addition-elimination reaction with a nucleophile such as CH3NH2.From 1 being the least reactive and 4 being the most reactive rank the following alkyk chlorides in terms of increasing reactivity via SN2 reaction mechanism.
- Rank the nucleophiles in each group in order of increasing nucleophilicity. a.−OH, −NH2, H2O b.−OH, Br−, F− (polar aprotic solvent) c.H2O, −OH, CH3CO2−Identify whether the following nucleophile will favor SN2 or SN1:1. SN1 or SN2 2. Draw major product withs steorchemistry 3. include reaction mechanism

