Aspirin is a weak acid with a pk, of 3.5. It is absorbed into the blood through the cells lining the stomach and the small intestine. Absorption requires passage through the plasma membrane, the rate of which is determined by the polarity of the molecule, charged and polar molecules tend to pass more slowly, whereas neutral hydrophobic molecules pass rapidly. The pH of the stomach contents is about 1.5, and the pH of the contents of the small intestine is about 6. Is the aspirin absorbed into the bloodstream from the stomach or the small intestine? Clearly justify your choice. ОН Acetylsalicylic Acid (Aspirin)
Aspirin is a weak acid with a pk, of 3.5. It is absorbed into the blood through the cells lining the stomach and the small intestine. Absorption requires passage through the plasma membrane, the rate of which is determined by the polarity of the molecule, charged and polar molecules tend to pass more slowly, whereas neutral hydrophobic molecules pass rapidly. The pH of the stomach contents is about 1.5, and the pH of the contents of the small intestine is about 6. Is the aspirin absorbed into the bloodstream from the stomach or the small intestine? Clearly justify your choice. ОН Acetylsalicylic Acid (Aspirin)
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter15: Acid–base Equilibria
Section: Chapter Questions
Problem 58P
Related questions
Question
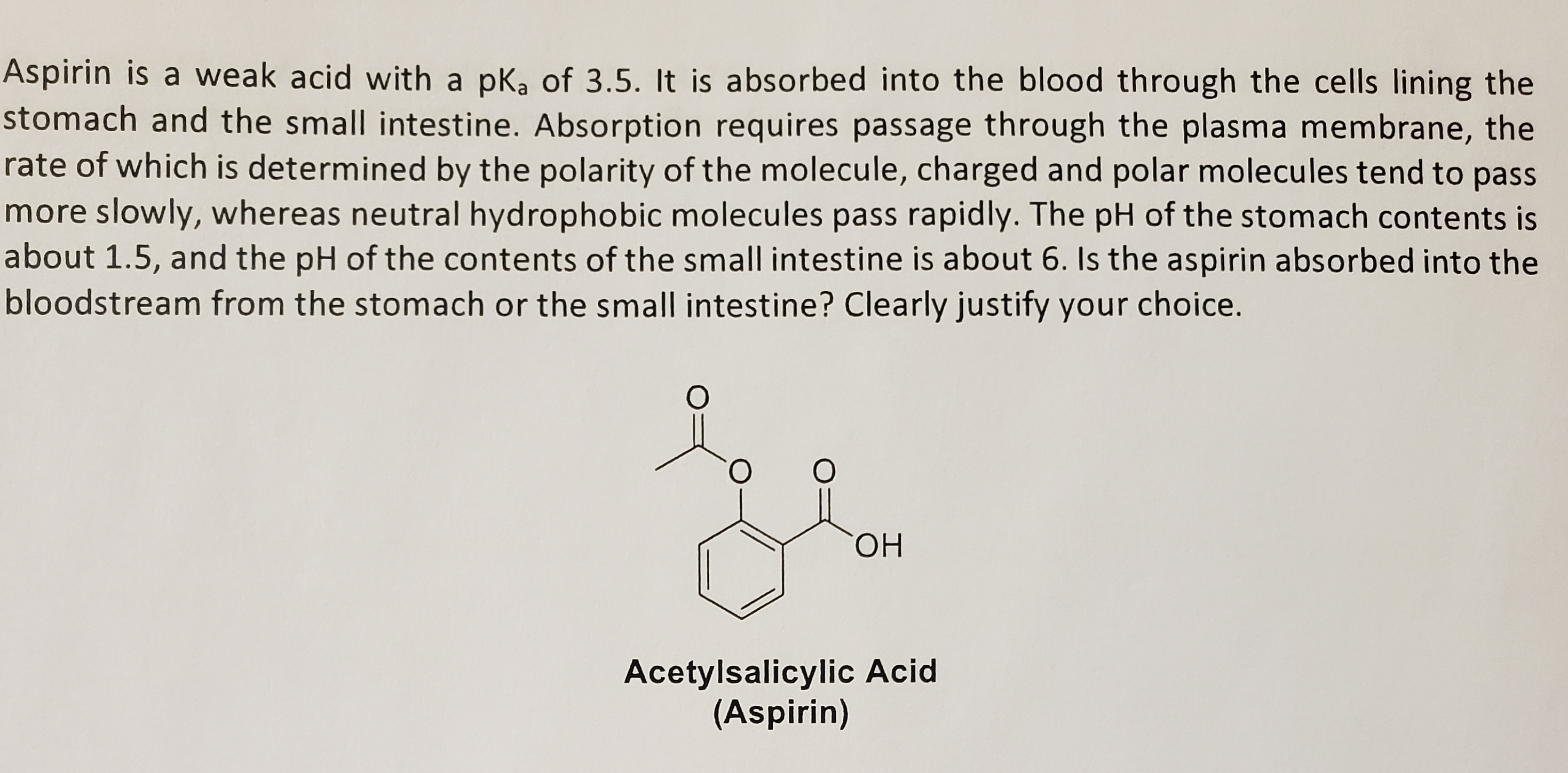
Transcribed Image Text:Aspirin is a weak acid with a pk, of 3.5. It is absorbed into the blood through the cells lining the
stomach and the small intestine. Absorption requires passage through the plasma membrane, the
rate of which is determined by the polarity of the molecule, charged and polar molecules tend to pass
more slowly, whereas neutral hydrophobic molecules pass rapidly. The pH of the stomach contents is
about 1.5, and the pH of the contents of the small intestine is about 6. Is the aspirin absorbed into the
bloodstream from the stomach or the small intestine? Clearly justify your choice.
ОН
Acetylsalicylic Acid
(Aspirin)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning