avallablé until you sign in again. Learning rning Ch 7 HW Najma Jama - > Normandale Community College - CHEM 1020 - Spring20 - NGWENDSON > Activities and Due Dates > Ch 7 HW ons く O Assignment Score: 42.6% L Give Up? O Hint Resources Check Answer く Question 8 of 23 100% Correct Suppose you have two samples that are equal in weight, 28.3 g Ni and 28.3 g Fe, O2. Calculate the number of moles of each substance. 85% Correct 100% Ni: mol Correct mol 0/100 Fe,O3: 100% Correct 100% Correct privacy policy terms of use contact us help about us careers arning, Inc. 13 59 6. Aa APR PDF 16 DD F12 DII F11 F10 F9 F8 F6 F5 F4 F3 F2 delete 24 4. * CO
avallablé until you sign in again. Learning rning Ch 7 HW Najma Jama - > Normandale Community College - CHEM 1020 - Spring20 - NGWENDSON > Activities and Due Dates > Ch 7 HW ons く O Assignment Score: 42.6% L Give Up? O Hint Resources Check Answer く Question 8 of 23 100% Correct Suppose you have two samples that are equal in weight, 28.3 g Ni and 28.3 g Fe, O2. Calculate the number of moles of each substance. 85% Correct 100% Ni: mol Correct mol 0/100 Fe,O3: 100% Correct 100% Correct privacy policy terms of use contact us help about us careers arning, Inc. 13 59 6. Aa APR PDF 16 DD F12 DII F11 F10 F9 F8 F6 F5 F4 F3 F2 delete 24 4. * CO
Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.27QAP
Related questions
Question
Transcribed Image Text:avallablé until you sign in again.
Learning
rning
Ch 7 HW
Najma Jama -
> Normandale Community College - CHEM 1020 - Spring20 - NGWENDSON > Activities and Due Dates > Ch 7 HW
ons
く
O Assignment Score:
42.6%
L Give Up?
O Hint
Resources
Check Answer
く
Question 8 of 23
100%
Correct
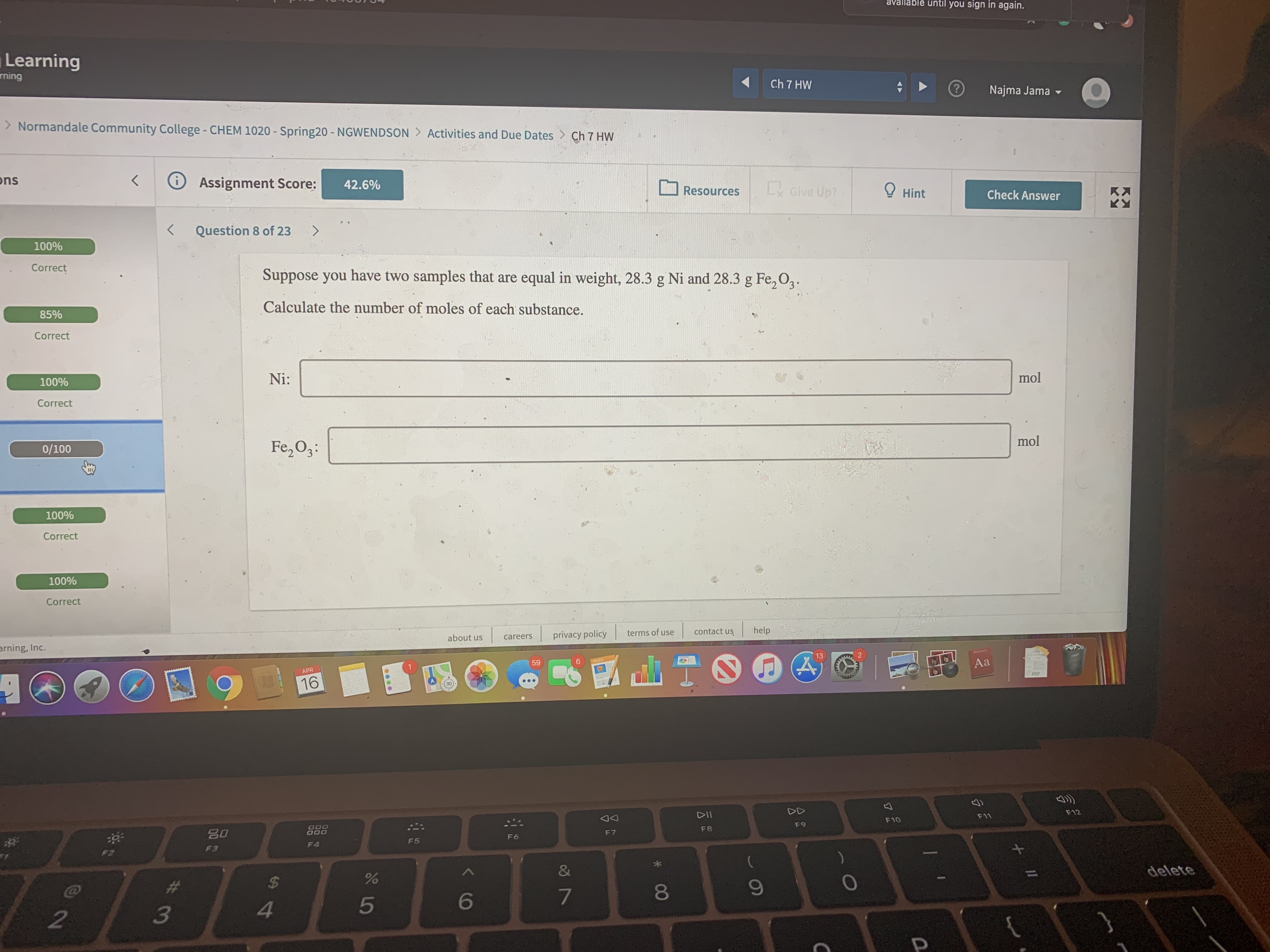
Suppose you have two samples that are equal in weight, 28.3 g Ni and 28.3 g Fe, O2.
Calculate the number of moles of each substance.
85%
Correct
100%
Ni:
mol
Correct
mol
0/100
Fe,O3:
100%
Correct
100%
Correct
privacy policy
terms of use
contact us
help
about us
careers
arning, Inc.
13
59
6.
Aa
APR
PDF
16
DD
F12
DII
F11
F10
F9
F8
F6
F5
F4
F3
F2
delete
24
4.
* CO
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

