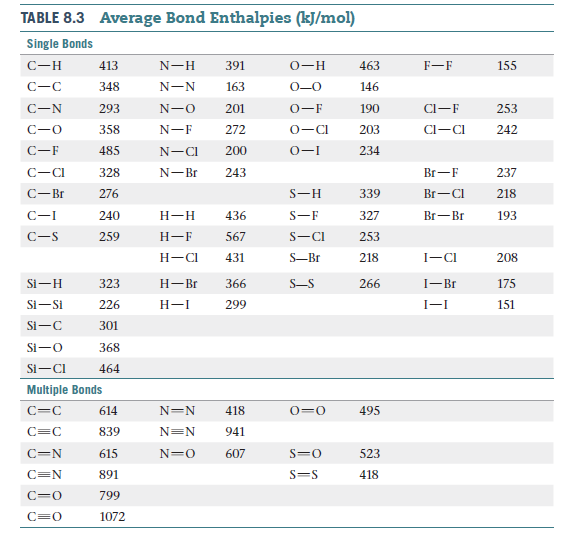
Average Bond Enthalpies (kJ/mol) TABLE 8.3 Single Bonds C-H 413 N-H 391 О—н 463 F-F 155 348 N-N 163 0-0 146 CI-F 293 201 0-F 190 253 C-0 358 N-F 272 0-CI 203 Cl-CI 242 C-F 485 200 0-I 234 N-CI 243 C-CI 328 N-Br Br-F 237 C-Br 276 S-H 339 Br-CI 218 C-I 240 Н-Н 436 S-F 327 Br-Br 193 C-S 259 Н—F 567 S-CI 253 Н—СІ 431 S-Br 218 I-CI 208 Si-H 323 Н—Br 366 S-S 266 I-Br 175 Si-Si 226 Н-I 299 I-I 151 Si-C 301 Si-O 368 Si-CI 464 Multiple Bonds 495 614 N=N 418 0=0 839 N=N 941 615 607 523 C=N 891 S=S 418 799 1072 нн Н Н (а) Н—С—С—о—Н Н—С—О- -Н Н Н Н Dimethyl ether Ethanol но (b) Н—ҫ—ҫ-н — нҫ—С—Н Н Н Н Ethylene oxide Acetaldehyde Н Н Н Н. нннн Н. —С—С—С—Н Н (с) Н Н Н Н Н Cyclopentene Pentadiene н Н (d) Н-ҫ—N с — Н- Н Н Acetonitrile Methyl isocyanide
Thermochemistry
Thermochemistry can be considered as a branch of thermodynamics that deals with the connections between warmth, work, and various types of energy, formed because of different synthetic and actual cycles. Thermochemistry describes the energy changes that occur as a result of reactions or chemical changes in a substance.
Exergonic Reaction
The term exergonic is derived from the Greek word in which ‘ergon’ means work and exergonic means ‘work outside’. Exergonic reactions releases work energy. Exergonic reactions are different from exothermic reactions, the one that releases only heat energy during the course of the reaction. So, exothermic reaction is one type of exergonic reaction. Exergonic reaction releases work energy in different forms like heat, light or sound. For example, a glow stick releases light making that an exergonic reaction and not an exothermic reaction since no heat is released. Even endothermic reactions at very high temperature are exergonic.
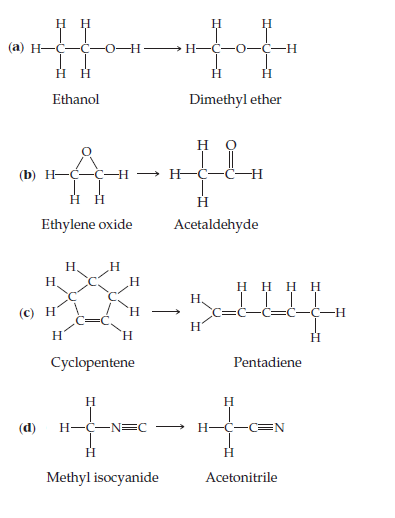
Two compounds are isomers if they have the same chemical
formula but different arrangements of atoms. Use Table 8.3
to estimate ΔH for each of the following gas-phase isomerization
reactions and indicate which isomer has the lower
enthalpy.
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 8 images









