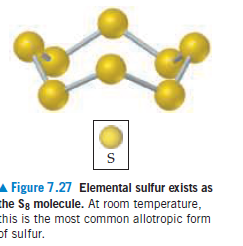
Average Bond Enthalpies (kJ/mol) TABLE 8.3 Single Bonds C-H 413 N-H 391 О—н 463 F-F 155 348 N-N 163 0-0 146 CI-F 293 201 0-F 190 253 C-0 358 N-F 272 0-CI 203 Cl-CI 242 C-F 485 200 0-I 234 N-CI 243 C-CI 328 N-Br Br-F 237 C-Br 276 S-H 339 Br-CI 218 C-I 240 Н-Н 436 S-F 327 Br-Br 193 C-S 259 Н—F 567 S-CI 253 Н—СІ 431 S-Br 218 I-CI 208 Si-H 323 Н—Br 366 S-S 266 I-Br 175 Si-Si 226 Н-I 299 I-I 151 Si-C 301 Si-O 368 Si-CI 464 Multiple Bonds 495 614 N=N 418 0=0 839 N=N 941 615 607 523 C=N 891 S=S 418 799 1072 Figure 7.27 Elemental sulfur exists as the Sg molecule. At room temperature, chis is the most common allotropic form of sulfur.
Elemental sulfur is a yellow solid that consists of S8 molecules. The structure of the S8 molecule is a puckered, eightmembered
ring (see Figure 7.27). Heating elemental sulfur to high temperatures produces gaseous S2 molecules:
S8(s)---->4 S2(g)
(a) The electron configuration of which period 2 element is most similar to that of sulfur? (b) Use the VSEPR model to predict
the S¬S¬S bond angles in S8 and the hybridization at S in S8. (c) Use MO theory to predict the sulfur–sulfur bond
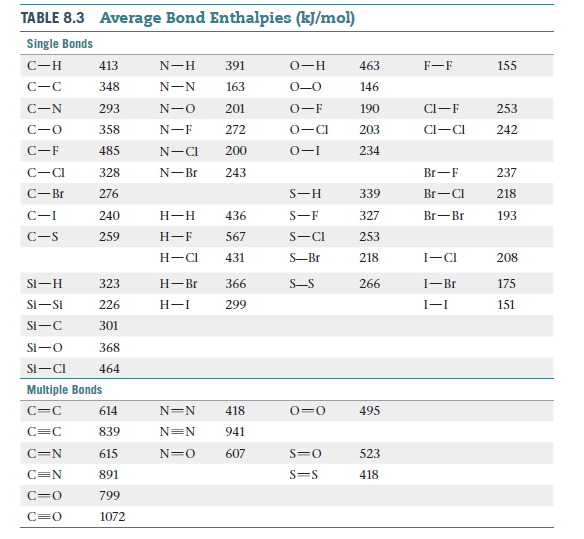
order in S2. Do you expect this molecule to be diamagnetic or paramagnetic? (d) Use average bond enthalpies (Table 8.3) to
estimate the enthalpy change for this reaction. Is the reaction exothermic or endothermic?
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images









