Q: What volume of gas (in liters) is occupied by 2.7 grams of F2 gas at 25 degrees Celsius and 1.01…
A: We can solve this problem by Ideal gas equation -PV = nRTWhere -P = pressureV = volumen = moles of…
Q: Predict the product H3C H2 IO CH3 ОН CH2 [0]
A:
Q: In the Lewis structure for the NO2 molecule, the number of lone pairs of electrons around the…
A: The correct answer is OPTION B (1)Let us first know the number of valence electrons,Valence…
Q: Calculate the concentration of the products of the following equilibrium when the equilibrium…
A: Equilibrium constant= 12.3 Equilibrium concentration of NOBr2 = 2.17
Q: explain the reason why the boiling point of alkane is decrease as branching increse
A: Boiling point of a compound depends on the intermolecular forces present in it. Only type of…
Q: Select the single best answer. Considering only electron density, will the following reaction occur?…
A: The reactants given are,
Q: 3. Calculate the molar amount AND the mass (in grams) of sodium acetate (CH-COONa) that must be…
A:
Q: A brain scan uses the radioisotope oxygen-15. The recommended dosage is 50 mCi . A supply of 230 mCi…
A: Recommended dosage = 50 mCisupply of 250mCi correspond to 40ml
Q: Considering only ions with charges of +1, +2, -1 and -2, or neutral atoms, give the symbols for 4…
A: Isoelectronic compound will have same number of electrons. The ion which can show the same type of…
Q: c) 43.0 x 107 d) 1.3 x 10³ 3. In an infrared (IR) spectrum, which of the following functional groups…
A: IR is powerful tool to determine or identification of functional group in compound. In IR the…
Q: Calculate the mass percent of water of crystallization in the hydrate FeCl2.8H₂0. Given the molar…
A:
Q: Vapor pressure (mm Hg) 900 800 700 600 500 400 300 200 100 Carbon disulfide 0 10 20 30 40 °C Methano…
A:
Q: Chem
A: The pressure equilibrium constant, Kp, is a thermodynamic equilibrium constant that expresses the…
Q: Hemic Upgrading Chemistry 2. Magnesium was added to hydrochloric acid, HCI (aq), and produced 5.25 L…
A: The amount of reactant is calculated by the balanced chemical reaction. The mass will be determined…
Q: In the number 723.4, which digit is estimated? 02 0 3 01 4
A: The given number is = 723.4This number contains a decimal place.
Q: How many atoms are in each elemental sample? A. 16.8 g Sr B. 7.77 g Bi C. 32.9 g P
A:
Q: What is the product of the reaction below? CH₂ OH Select one: O a. O b. CH₂ CH₂ CH; CH₂ K₂Cr₂O
A: We know that potassium dichromate, K2Cr2O7 is a strong oxidizing agent. It is oxidised primary…
Q: Molecular orbitals in metals... A. Overlap significantly to allow free electron movement through the…
A: Molecular orbital or band theory describes the electronic structure and electronic energy level in…
Q: 16. Bismuth -210 is an alpha emitter. Write an equation for this nuclear reaction and identify the…
A: Note: Since you have asked multiple questions, we will solve the first question for you. If you want…
Q: Structure A Br H3C...C H CH₂CH3 Structure B CH 3 Hmo Br CH₂CH3 Determine the relationship between…
A:
Q: can be nical chan H of cor negative, egative/ le neleme 1. Write equations for the reaction between…
A: a. write the balanced half-cell reactionsb. write the overall balanced equation for the reactionc.…
Q: Write the net ionic equation for the reaction for the following acid-base reaction: Ca(OH)2(aq) + 2…
A: The question is based on the concept of organic reactions.We need to write net ionic equation for…
Q: The voltage generated by the zinc concentration cell described by the line notation Zn(s) Zn² (aq,…
A: Cell voltage (Ecell)=14 mV Concentration of Zn^2+(aq) in anode=0.1 M
Q: If myristic acid is molecular, what is I in the ∆Tf=ikfm equation?
A: In the given equation i represent van't Hoff factor. For ionic compounds, it will be equal to the…
Q: 2. For each pair of the following compounds, identify which compound and conditions would react more…
A: Biomolecular nucleophilic substitution reaction (SN2 reaction) involves the attack of nucleophile…
Q: The reaction conditions to carry out the production of cyclopentene using bromocyclopentane as the…
A:
Q: 4. A student tested the pH of 0.1 mol/L solutions of hydrochloric acid and acetic acid. The pH the…
A: The question is based on the concept of the pH of the solution. It is defined as a negative…
Q: ify each of the molecules as one of the central electron geometries and molecular shapes. Molecule:…
A: -> electron geometry refers to arrangement of electron density around the central atoms. Geometry…
Q: 6. For each of the following acid base reactions shown below, draw curved arrows to show proton…
A: Sodium hydroxide (NaOH) is a very strong base, thus it can easily abstract the acidic proton on…
Q: Requirements must HANDWRITING ok dont copy paste. Must solve correct thanks. asap 1. Give an example…
A: A question based coordination complexes. Description about a ligand, its structure, its binding…
Q: 17. The energy was associated with the gamma ray at frequency equal 1 x 1020 Hz is: (h-6.63 x…
A: The frequency of gamma rays =The energy associated with the gamma ray = ? Planck's constant (h) =
Q: Question 5 Isomer A CI Which isomer is chiral? Isomer B CH₂CI
A: Given two of the compounds are bridge compounds. One of them isomer is called as a norbornene…
Q: What is released when HCI dissolves in water? O a hydroxide ion O a chlorine ion a hydrogen ion O an…
A: HCl(g) + H2O(l) ---------> H3O+(aq) + Cl-(aq)
Q: Reaction Kinetics and Dimensional Analysis (a) Consider an example first-order reaction of the…
A: A) A first order reaction is given above where A is the reactant which gives the two products B and…
Q: If the initial amount of Fe(NO3)3 transferred to the cuvette is 0.029 mol, and the absorbance…
A: Given Data :Initial Amount of Fe(NO3)3 = 0.029 molEquilibrium amount of FeSCN2+ = 0.019 molTotal…
Q: If the amount of product made (Fe2O3) was determined to contain 4.82 moles of Fe2O3, how many MOLES…
A: The number of moles of atom in a compound is determined based on the number of moles of compound.…
Q: Given the following: P(s) +50₂(g) →PO(s) A Gº=-2697.0 kJ/mol 4 10 2H₂(g) + O₂(g) → 2H₂O(g) A G° = −…
A: The Gibbs free energy of a reaction is calculated by the addition or subtraction of the Gibbs free…
Q: 8. What is the pH of a buffer solution made with 1.8M benzoic acid with Ka = 6.5 x 105 and 0.478M…
A:
Q: 1) Which of the following elements is a metal? A) nitrogen 2) Which of the following elements is a…
A: 1.nitrogen is a non metalic element Which belongs to group 15.2.fluorine is also a non metal which…
Q: Which substance is not readily oxidized by acidified potassium A. propan-1-ol propan-2-ol propanal…
A: -> potassium dicromate can oxidize 1° alcohol to Carboxylic acid.-> potassium dicromate can…
Q: 49. Identify the unknown particles (AZX) in the equations below. Show your work and mention the…
A:
Q: In 1c, the head groups are in the aqueous phase. Why would this affect the membrane fluidity?
A: Choline head groups are the substituents attached to phosphate group in phospholipids. The graph…
Q: d) In the following oxidation-reduction reaction, Ecell is measured as 0.12 V in the electrochemical…
A:
Q: Which statements are true about the isolated, single p orbital shown below? 8 I. It can hold up to…
A: 1. Orbital is the region where probability of finding electron is maximum. 2. Various orbital are s…
Q: Select one: A. +278.3 J/K B. +89.3 J/K C. -89.3 J/K D. +147.3 J/K
A: The change in enthalpy of a reaction, often denoted as ΔH, can be calculated using the formula:ΔH =…
Q: The new resonance structure (based on the pattern described) is provided in the sketch box. Add the…
A: We have to show , the movement of electrons (by arrow) in the given resonance.
Q: Which of the following salts is the least soluble in a given volume of water? PbCl₂ Al(OH)3 Agl…
A: The question is based on the concept of solubility product principle. It that states that when a…
Q: Suppose a thermometer has marks at every one degree increment and the meniscus level on the…
A: Correct answer is to be identified.
Q: 1) An aqueous solution of nitric acid is standardized by titration with a 0.182 M solution of barium…
A:
Q: For the cell shown, the measured cell potential, Ecell, is -0.3537 V at 25 °C. Pt(s) | H₂(g, 0.837…
A: Cell potential, Ecell= -0.3537 VHalf-cell reactions are also given.
C2.
Subject:-Chemistry
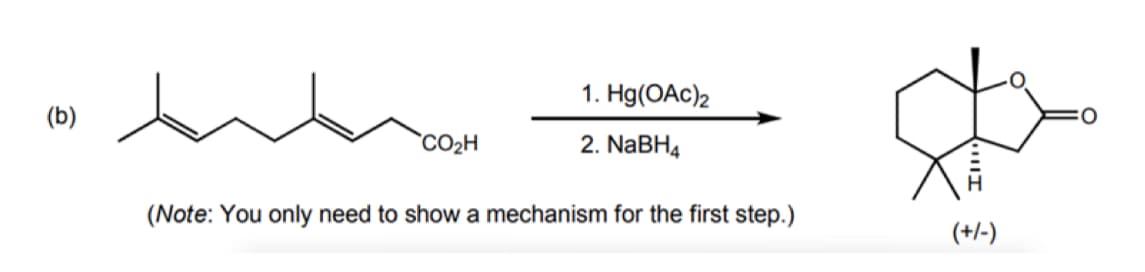
Step by step
Solved in 3 steps with 1 images

- Do not give handwriting solution. please draw out what compound A is and then provide aroow pushing mechanism for thisreaction! thanks01 Cive the ctrientiono of Comnand 4 2.2 Provide a reasonable arrowpushing mechanism for the reaction in 2.1When drawing organic chemistry mechanisms with NaOCH3, do we automatically dissociate it into Na+ and OCH3-, making it easier to go through SN2 and/or E2 reactions? Does NaOCH3 dissociate, and is this why it is a strong nucleophile?Below are two potential methods for preparing the same ether (Option A and B), but only one of them is actually successful. Identify the successful approach (A or B)

