b My Questions | bartleby 101 Chem101 app.101edu.co Submit Question 28 of 29 Using the provided table, determine the enthalpy for the reaction C3H8 (g) + 5 O2 (g) → 3 CO2 (g) + 4 H2O (g) AH? (kJ/mol) Substance СэНв (9) -108.4 O2 (g) CO2 (g) -393.5 H2O (g) -241.8 |kJ/mol х 1 3 4 8. 9. +/- х 100 7:49 PM O Type here to search 12/9/2019 LO
b My Questions | bartleby 101 Chem101 app.101edu.co Submit Question 28 of 29 Using the provided table, determine the enthalpy for the reaction C3H8 (g) + 5 O2 (g) → 3 CO2 (g) + 4 H2O (g) AH? (kJ/mol) Substance СэНв (9) -108.4 O2 (g) CO2 (g) -393.5 H2O (g) -241.8 |kJ/mol х 1 3 4 8. 9. +/- х 100 7:49 PM O Type here to search 12/9/2019 LO
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter8: Thermochemistry
Section: Chapter Questions
Problem 72QAP: On complete combustion at constant pressure, a 1.00-L sample of a gaseous mixture at 0C and 1.00 atm...
Related questions
Question
Transcribed Image Text:b My Questions | bartleby
101 Chem101
app.101edu.co
Submit
Question 28 of 29
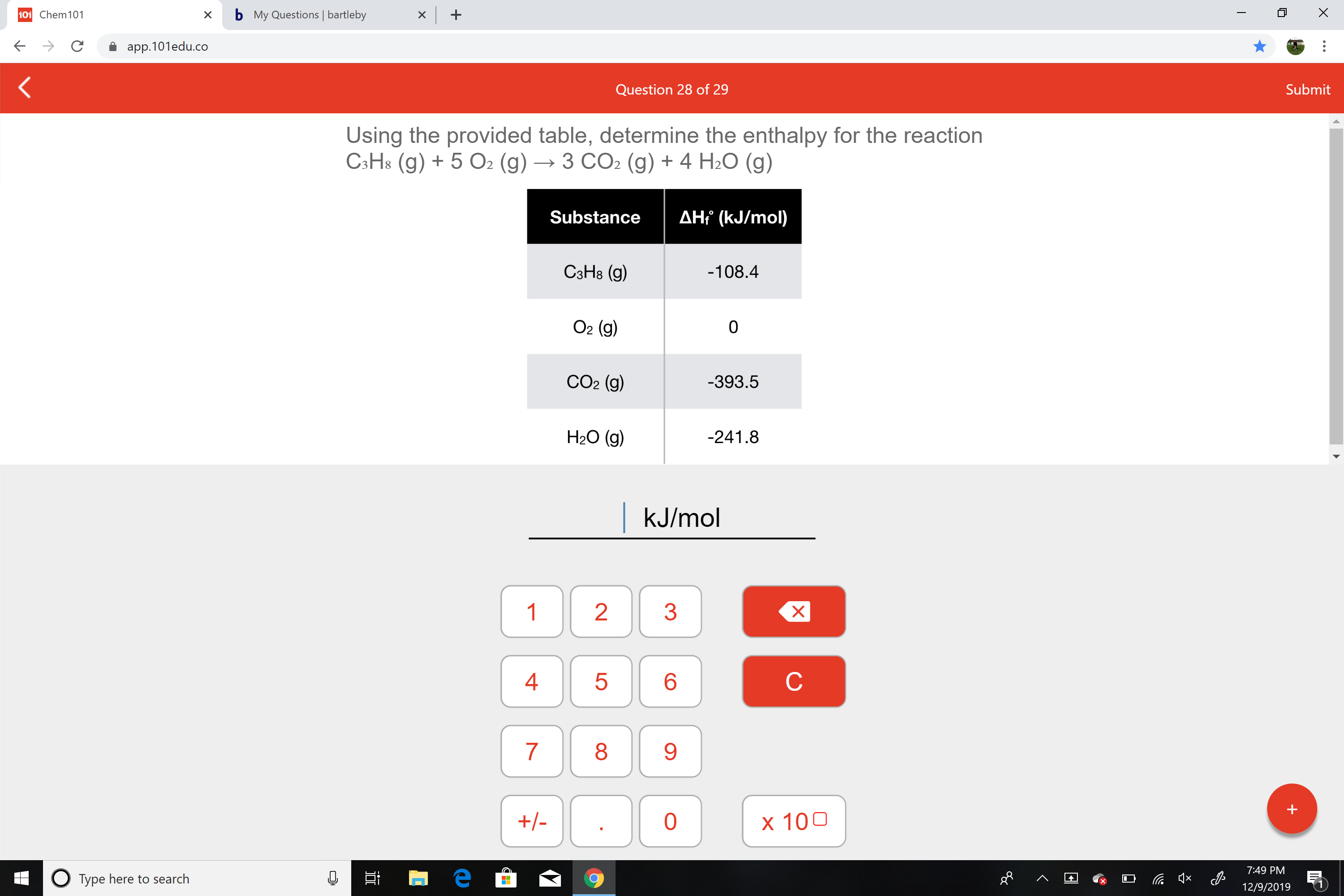
Using the provided table, determine the enthalpy for the reaction
C3H8 (g) + 5 O2 (g) → 3 CO2 (g) + 4 H2O (g)
AH? (kJ/mol)
Substance
СэНв (9)
-108.4
O2 (g)
CO2 (g)
-393.5
H2O (g)
-241.8
|kJ/mol
х
1
3
4
8.
9.
+/-
х 100
7:49 PM
O Type here to search
12/9/2019
LO
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 6 steps with 5 images

Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning