Base case no inhibition -'s
Q: What is an inhibitor?
A: Enzymes are referred to as molecules of proteins that helps in increasing the metabolism rate and…
Q: (a) What are the Vmax and Km (including units) for Tigerzyme? b) What are the Vmax and Km…
A: According to Michaelis-Menten enzyme kinetics, when the rate or velocity of an enzyme catalyzed…
Q: The compound below is an affinity label for the enzyme trypsin. H2N. NH2 NH H3C Provide detailed…
A: Affinity labels are a class of enzyme inhibitors that covalently bind to their target causing its…
Q: Given the active site and reaction mechanism below, what is the mechanism of rreversible inhibition…
A: The mechanism followed by the given example is of Transition state analog Chemical compounds with a…
Q: Why can we say that having a pure noncompetitive inhibitor present is similar to just having less…
A: Enzyme inhibition refers to the decrease in enzyme related processes, enzyme production, or enzyme…
Q: An enzyme catalyzes a reaction in which substrate A is cleaved into two products, P and Q. In the…
A: As you have posted question with multiple subparts, we will answer the first question for you. If…
Q: In an enzyme kinetics study, three inhibitors resulted to the following results: Inhibitor ABC…
A: Enzymes are biological catalysts that help chemical processes occur faster. Enzymes are proteins…
Q: Given the active site and reaction mechanism, identify the mechanism of irreversible inhibition for…
A: Irreversible inhibition is a process in which inhibitors bind covalently or non-covalently to a…
Q: Write the chemical reaction catalyzed by chymotrypsin. How is its specificity determined? How is the…
A: Chymotrypsin is a type of protease enzyme. It acts as a digestive enzyme, primarily synthesized…
Q: In an enzyme kinetics study, three inhibitors resulted to the following results: Inhibitor ABC…
A: The rate of a enzyme catalyzed reaction increases with temperature until a point and once the…
Q: In the case of cyanide poisoning, what kind of inhibitor, including the inhibitor, plays an…
A: Ans: Cyanide is harmful to the body since it prevents the body's cells from using oxygen. Further…
Q: Which type of Enzymatic inhibition is commonly found in Pharma and why?
A: The inhibitors are the molecules that inhibit or decrease the activity of enzyme in catalyzing the…
Q: What are the cellular advantages to feedback inhibition?
A: Feedback inhibition is a cellular control mechanism in which an enzyme's activity is inhibited by…
Q: In an enzyme kinetics study, three inhibitors resulted to the following results: Inhibitor ABC…
A: Asked : Type of inhibitor PQR
Q: Why is the transition-state analog not necessarily the same as a competitive inhibitor
A: Competitive inhibitor is the inhibitor having the similar shape as the substrate of the enzyme and…
Q: Given the active site and reaction mechanism below, what is the mechanism of irreversible inhibition…
A: The given example is uncompetitive inhibition. Uncompetitive inhibition, also known as…
Q: In which reversible inhibitors, adding more substrates can revert the inhibition? Why?
A: Enzymes takes part in various types of metabolic activity . So , their activities must have to be…
Q: Given the active site and reaction mechanism below, what is the mechanism of irreversible inhibition…
A: Irreversible inhibition is a process in which inhibitors bind covalently or non-covalently to a…
Q: Is the inhibitor competitive or uncompetitive or non-competitive and why?
A: Inhibitors are broadly classified into 2; reversible and irreversible. Reversible inhibitors are…
Q: What is the impact of the lower value Vmax on the affinity for enzyme for substrate? And what is…
A: The relationship between Km and substrate concentration is that Km corresponds to the substrate…
Q: Why does the apparent KM decrease in the presence ofan uncompetitive inhibitor?
A: An enzyme inhibitor is a molecule that binds to enzyme and decreases its activity. By binding to the…
Q: What type of reversible inhibitor are sulfanomides? Competitive Uncompetitive…
A: Substrate molecules get converted to product molecules during a biochemical reaction. Enzymes are…
Q: Based on some preliminary measurements, you suspect that a sample of enzyme contains an irreversible…
A: Enzymes are the biocatalyst which is required for most of the process occurring inside the living…
Q: what type of inhibition is shown and where does the inhibitor bind to the enzyme?
A: Enzymes are usually protein molecules which catalyzes the biochemical reaction by decreasing the…
Q: In an enzyme experiment, when the enzyme is added only to the substrate, the Vmax value is 0.828 and…
A: Enzymes are proteins with catalytic function. They are sensitive to temperature, the potential of…
Q: Given below are five Km values for the binding of substrates to a particular enzyme. Which substrate…
A: Those proteins or biological catalysts which help to speed up the chemical reaction are termed…
Q: The graph shows the rate of nitrocefin hydrolysis at different concentrations of nitrocefin in the…
A: The Michaelis-Menton plot gives the equation which describes the rate of enzymatic reactions, by…
Q: A junior student performed an experiment to found Ki values for 3 different competitive inhibitors.…
A: Competitive inhibitor competes for the same site to which the substrate binds on enzyme. Competitive…
Q: Why does a pure noncompetitive inhibitor not changethe observed KM?
A: The non-competitive inhibitor is a molecule that does not compete with the substrate to get bind to…
Q: In concerted model, all subunits in the enzyme are either in the low affinity or high affinity…
A: There are two models of allosteric regulation of enzymes:- - concerted model -sequential model
Q: How can inhibitor binding to an enzyme be quantified?
A: Inhibitor binding is competitive with the enzyme when the substrate is also present in the sample.…
Q: How can enzyme inhibition be used in the treatment of certain diseases? Cite examples
A: Introduction: Inhibitors are substances that bind to an enzyme and inhibit its activity. It will…
Q: draw and label a Michaelis-Menten plot showing the effect of a compentitive inhibitor
A: Two scientists named Leonor Michaelis and Maud Leonora Menten, proposed a model which is known as…
Q: Under what conditions, a higher rate in the presence of the inhibitor observed? (how could the…
A: There is a gradual increase in reaction rate because competitive inhibitors are occupying only some…
Q: What is the difference between pure and mixed noncompetitive inhibition?
A: Pure competitive inhibition is the type of inhibition in which the inhibitor binds to the site other…
Q: In pure noncompetitive inhibition: a. Where on the enzyme does the inhibitor bind? b. Does…
A: Enzymes refer to the protein that acts as a catalyst for the biochemical reaction. It facilitates…
Q: Noncompetitive inhibition can often be explained by which of the following models? the induced fit…
A: The action of enzyme depends on different factors. Enzymes are biocatalysts in nature. All the…
Q: Write the difference between compititive and non compititive enzyme inhibitors.
A: Enzyme inhibitors binds with the active sites of enzymes and reduces the compatibility of substrate…
Q: A plot of 1/ versux 1151, called a Lineweaver Burk or double-reciprocal plol, is a useful tool for…
A: Enzymes are proteins that hasten biochemical reactions such that substrate molecules are converted…
Q: Which inhibitor binds to the enzyme with greater strength, 1, 2, or 3?
A: The structure of a competitive inhibitor closely resembles that of the enzyme's normal substrate.…
Q: What is feedback inhibition? Why is it a useful property?
A: Proteins serve numerous functions in the body including catalysis of biochemical reactions, immune…
Q: What type of inhibition ia observed from the ahift of the Lineweaver-Burke plot ahown in the graph…
A: Disclaimer: Since you have asked multiple question, we will solve the first question for you. If you…
Q: 5.5 Explain the effect of each type of inhibitor on the apparent kinetic parameters:
A:
Q: If the higher value of KM resulting in the new plot ( red curb ) is due to the presence of an enzyme…
A: The concentration of irreversible inhibitor is less than the concentration of enzyme, an…
Q: When we compare the binding of I and of S to the enzyme in a mixed noncompetitive inhibitor, we…
A: In non-competitive inhibition, the inhibitor binds to the enzyme at a site other than the active…
Q: What TYPE of inhibition is observed in the following: S E S
A: A chemical that binds to an enzyme and inhibits its activity is known as an enzyme inhibitor.…
Q: What is the difference between competitive and non-competitive inhibition? How can each be…
A: The substance that inhibit the activity of enzyme are called enzyme inhibitors and the mechanism by…
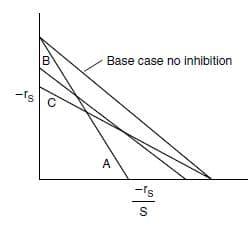
An Eadie-Hofstee plot is shown below for the different types of enzyme inhibition. Match the line with
the type of inhibition.
(a) Line A Inhibition Mechanism. Ans: ________
(b) Line B Inhibition Mechanism. Ans: ________
(c) Line C Inhibition Mechanism. Ans: ________
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- Modified TRUE or FALSE. Write the word TRUE if the statement is correct. If the statement is false, write the correct word to make the statement true. 1. The L-form is the naturally-occurring form of amino acids.Give me properly solution fastAttach an amino acid monomer below. Name of the amino acid: Abbreviated name: Letter symbol:

